New Treatment Option for Inflammatory Diseases - IRAK4 inhibitors
Interleukin-1 receptor-associated kinases (IRAKs) were initially identified as serine/threonine kinases that can bind to the interleukin-1 receptor (IL-1R) upon induction by the pro-inflammatory cytokine interleukin-1 (IL-1). Later, these proteins were found to have high homology with the pelle kinases vital for resisting microbial infection in Drosophila Toll-like receptor (TLR) pathways. There are four members in the IRAK family: IRAK1, IRAK2, IRAK 4, and IRAK–M. All four IRAKs have a kinase domain (KD) containing serine and threonine, and a conserved death domain (DD). Except for IRAK4, the other three IRAKs also have a long C-terminal domain (C-terminal Domain). Among them, IRAK1 and IRAK4 play a positive regulatory role in TLR-mediated signaling pathways, while IRAK2 and IRAK-M play a negative regulatory role.
As for the function of IRAK4 in TLRs/IL-1R mediated signaling pathways, aside from the classical activation of downstream NF-κB and AP-1 transcription factors, inducing the expression of inflammatory cytokines, chemokines, other studies have found that overexpression of IRAK4 can enhance LPS induced NADPH oxidase activity, and trigger the signal transduction of downstream p38 MAPK. This extends the biological role of the IRAK4 molecule even further, from simply functioning in TLRs /IL-1R mediated signaling pathways to actively participating in the widespread regulation of cellular response networks. With its signaling complex formation impeded by the absence of functional IRAK4 due to mutation, inflammation signal propagation becomes flawed. As a result, IRAK4 can serve as an effective potential therapeutic target for inflammatory-related disease, tackling them by inhibiting inflammatory signal transduction through specific targeting of IRAK4.
IRAK4 Competitive Landscape
According to the data provided by Patsnap Synapse-Global Drug Intelligence Database: the following figure shows that as of 20 Sep 2023, there are a total of 42 IRAK4 drugs worldwide, from 36 organizations, covering 38 indications, and conducting 49 clinical trials.
👇Please click on the picture link below for free registration or login directly if you have freemium accounts, you can browse the latest research progress on drugs , indications, organizations, clinical trials, clinical results, and drug patents related to this target.
Based on the analysis of the provided data, the current competitive landscape of target IRAK4 in the pharmaceutical industry is characterized by the involvement of multiple companies at different stages of development. Kymera Therapeutics, Inc., Bayer AG, and Gilead Sciences, Inc. are among the companies with the highest number of drugs in advanced stages of development.
The indications for drugs targeting IRAK4 are diverse, including rheumatoid arthritis, dermatitis, atopic, and hidradenitis suppurativa, among others. Small molecule drugs and PROTACs are the most rapidly progressing drug types under this target.
The United States, European Union, and China are the countries/locations with the highest development activity. China has shown progress in the development of drugs targeting IRAK4. Overall, the target IRAK4 presents a competitive landscape with potential for future development in various indications and drug types.
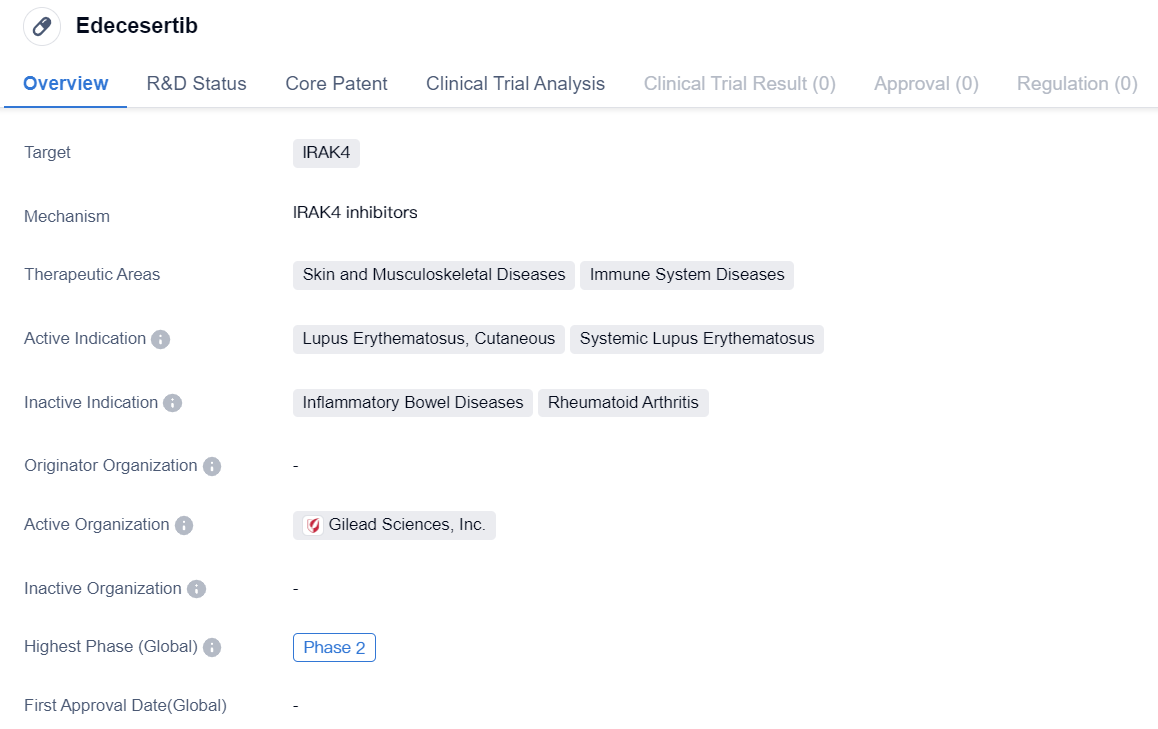
IRAK4 inhibitors entering Phase II clinical research: Edecesertib
Edecesertib is a small molecule drug that targets IRAK4, a protein involved in immune system signaling. It is being developed for the treatment of skin and musculoskeletal diseases as well as immune system diseases. The active indications for Edecesertib include lupus erythematosus, cutaneous, and systemic lupus erythematosus.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
Currently, Edecesertib is in Phase 2 of clinical development. This means that the drug has already undergone initial testing for safety and efficacy in a small group of patients and is now being evaluated in a larger population to further assess its effectiveness and side effects.
Lupus erythematosus is a chronic autoimmune disease that affects various parts of the body, including the skin, joints, kidneys, heart, and lungs. Cutaneous lupus erythematosus specifically refers to the form of the disease that primarily affects the skin. Systemic lupus erythematosus, on the other hand, affects multiple organs and can cause more severe symptoms.
The development of Edecesertib as a potential treatment for lupus erythematosus, both cutaneous and systemic, is significant as there is currently no cure for this condition. Current treatment options mainly focus on managing symptoms and preventing flare-ups. Therefore, the introduction of a new drug that targets IRAK4 and potentially modulates the immune system response could provide a novel approach to managing lupus erythematosus.
Additionally, Edecesertib's therapeutic areas also include skin and musculoskeletal diseases, which further expands its potential applications. Skin diseases encompass a wide range of conditions, such as psoriasis, eczema, and dermatitis, which can significantly impact a patient's quality of life. Musculoskeletal diseases, on the other hand, involve the bones, muscles, joints, and connective tissues, and include conditions like rheumatoid arthritis and osteoporosis.
In summary, Edecesertib is a small molecule drug that targets IRAK4 and is being developed for the treatment of skin and musculoskeletal diseases, as well as immune system diseases such as lupus erythematosus. With its current status in Phase 2 clinical trials, Edecesertib shows promise as a potential therapeutic option for patients suffering from these conditions. Further research and development will be necessary to determine its safety and efficacy in larger patient populations.
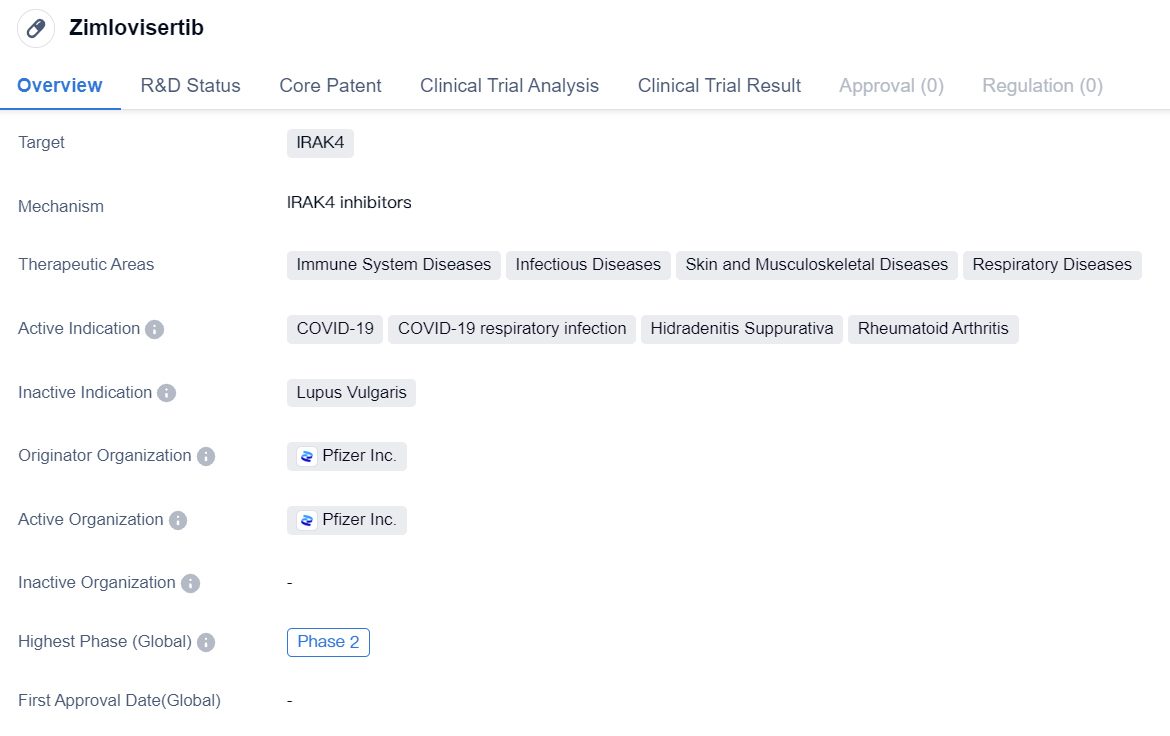
IRAK4 inhibitors entering Phase II clinical research: Zimlovisertib
Zimlovisertib is a small molecule drug that falls under the category of biomedicine. It specifically targets IRAK4, which is a protein involved in the immune response. The drug has shown potential in treating various therapeutic areas, including immune system diseases, infectious diseases, skin and musculoskeletal diseases, and respiratory diseases.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
In terms of active indications, Zimlovisertib has been identified as a potential treatment for COVID-19 and COVID-19 respiratory infections. This suggests that the drug may have antiviral properties and could be effective in combating the respiratory symptoms associated with the disease. Additionally, Zimlovisertib is being explored as a treatment for Hidradenitis Suppurativa, a chronic skin condition characterized by painful nodules and abscesses, as well as Rheumatoid Arthritis, an autoimmune disorder affecting the joints.
The originator organization of Zimlovisertib is Pfizer Inc., a renowned pharmaceutical company known for its contributions to the development of innovative drugs. This association adds credibility to the drug's potential efficacy and safety.
In terms of clinical development, Zimlovisertib has reached Phase 2 of clinical development globally. This indicates that the drug has undergone extensive preclinical testing and has shown promising results in early-stage clinical trials. However, it is important to note that the highest phase in China is still pending, suggesting that further regulatory approvals may be required before the drug can progress to advanced stages of development in the Chinese market.
In summary, Zimlovisertib is a small molecule drug developed by Pfizer Inc. that targets IRAK4. It has shown potential in treating various therapeutic areas, including immune system diseases, infectious diseases, skin and musculoskeletal diseases, and respiratory diseases. The drug is actively being investigated for its efficacy in treating COVID-19, COVID-19 respiratory infections, Hidradenitis Suppurativa, and Rheumatoid Arthritis. With its current highest phase being Phase 2 globally, Zimlovisertib holds promise as a potential treatment option, pending further regulatory approvals in China.