3
Clinical Trials associated with Furosemide/Amiloride ChlorideResistant Hypertension On Treatment - Sequential Nephron Blockade Compared to Dual Blockade of the Renin-angiotensin-aldosterone System Plus Bisoprolol in the Treatment of Resistant Arterial Hypertension: A Randomized Trial (ResHypOT)
A randomized clinical trial comparing sequential nephron blockage (SNB) with dual blockade of the renin-angiotensin system (RAAS) plus bisoprolol (DBB) in the treatment of resistant arterial hypertension (RH) was designed to investigate the importance of the SNB and the contribution of its volume component versus DBB and the importance of the serum renin in maintaining BP levels. This randomized trial with two treatment arms could help tailor therapy by identifying a more effective choice to control hypertension whether by acting on the control of volume or sodium balance, or by acting on the effects of the RAAS on the kidney.
Methods - Participants: 80 patients undergoing treatment for RH with losartan (100-200 mg), chlorthalidone (25 mg), and amlodipine (5 mg) will be randomly divided into two groups after applying inclusion and exclusion criteria.
Group 1: Sequential nephron blockade (SNB Group) n = 40 Group 2: Dual blockade of the RAAS plus bisoprolol (DBB Group) n = 40 Intervention: SNB consists in a progressive increase in sodium depletion. After the administration of a thiazide diuretic (chlorthalidone) and aldosterone receptor blocker, low doses of furosemide are administered and subsequently amiloride is prescribed to enhance the natriuretic effect.
The dual blockade of the RAAS plus bisoprolol is used to increase the effect of angiotensin receptor 1 blockers (ARBs). Therapy then requires sequentially adding an angiotensin converting enzyme (ACE) inhibitor to reduce the levels of angiotensin (Ang) II resulting from blockage of the Ang II receptor and then to administer a beta-blocker to decrease the elevated renin secretion due to both the ACE inhibitors and ARBs Objective: This study, which compares two antihypertensive treatment regimens in patients with RH, has the following objectives: to demonstrate the therapeutic efficacy of SNB against DBB in RH patients, and to assess the side effects and adherence to treatment over 20 weeks of treatment.
Enrollment: The eligibility criteria will follow those shown in the flowchart for the diagnosis of RH of the First Brazilian Position on RH.
Patients will be excluded if they have: chronic renal failure, atrial fibrillation/atrioventricular block, contraindication to the drugs that will be used, refusal or failure to follow the regimen and secondary hypertension.
Follow-up: Patients will be analyzed in five visits at intervals of 28 days for 20 weeks
Start Date01 Sep 2015 |
Sponsor / Collaborator- |
The Effect of Antihypertensive Agents Over Sleep Apnea: a Randomized Controlled Trial
Obstructive sleep apnea and hypertension are well-known cardiovascular risk factors. Their control could reduce the burden of heart disease across populations. There are several drugs to control hypertension, but the only consistently beneficial treatment to reduce apneas is continuous positive airway pressure. The demonstration that one drug could improve sleep apnea and hypertension would support a novel approach in the treatment of both diseases. The role of fluid retention in sleep apnea is known for several decades. The role of diuretics is well established in hypertension but was never appropriately tested in sleep apnea. Besides to test the efficacy of these drugs, this study will help to understand the mechanisms that link hypertension and sleep apnea and its treatment.
Safety and Efficacy of the Combination of Furosemide 40 mg + Amiloride Hydrochloride 10 mg in the Reduction of Edema of Cardiac Origin in Patients With Congestive Heart Failure Functional Class II (NYHA)
The study consists in two treatment groups, one group will receive Diurisa® (furosemide 40 mg + amiloride chloride 10 mg) and the other one will receive furosemide 40 mg (Lasix®)
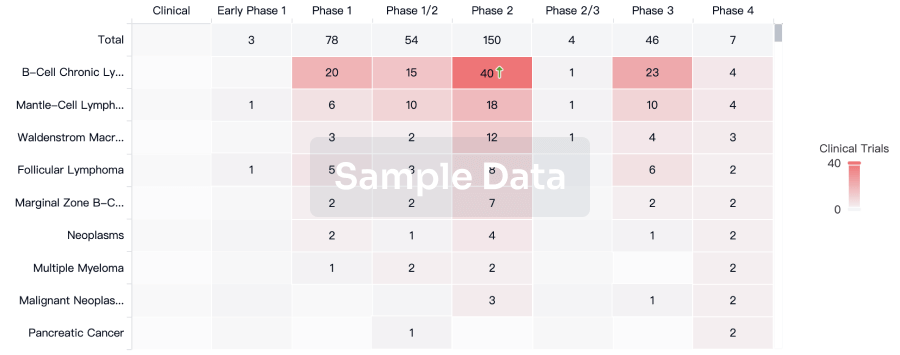
100 Clinical Results associated with Furosemide/Amiloride Chloride
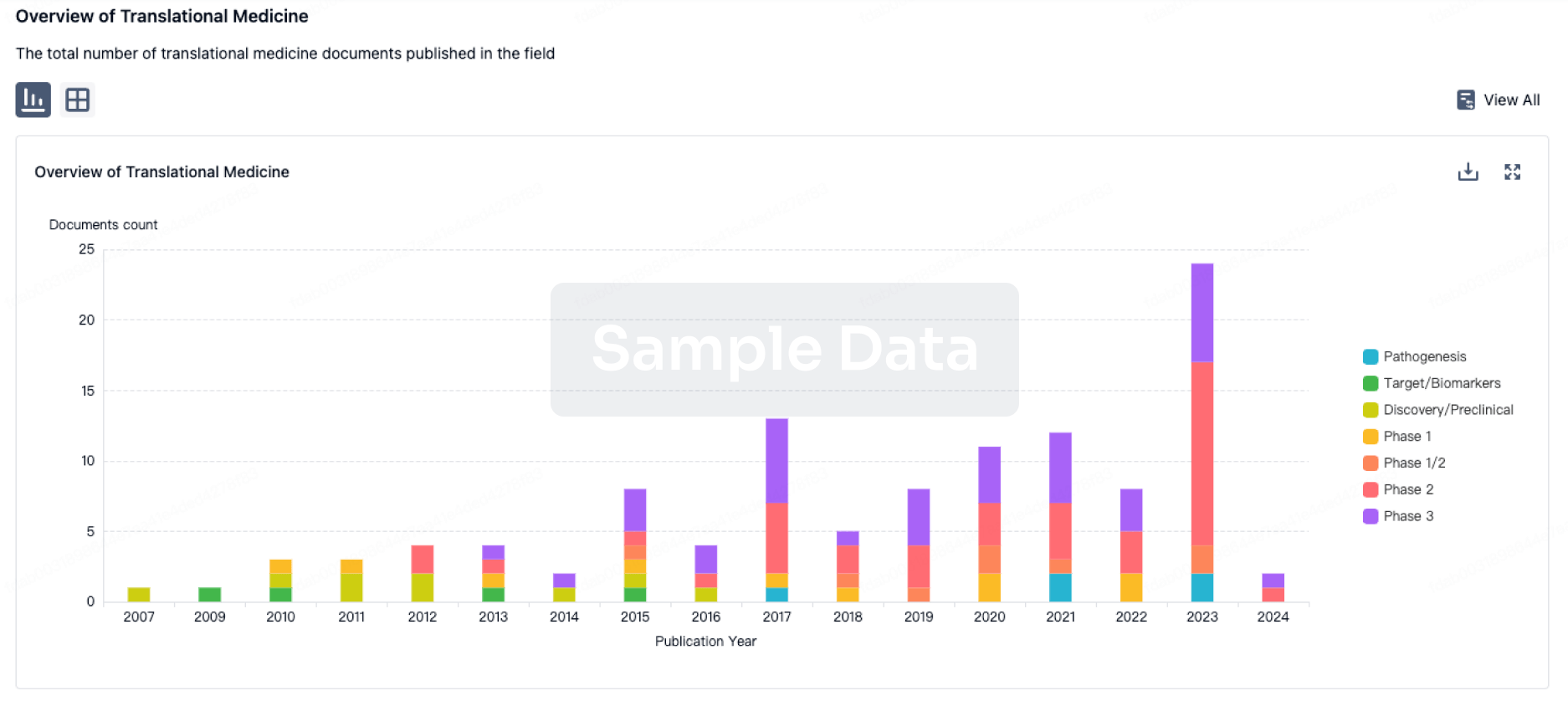
100 Translational Medicine associated with Furosemide/Amiloride Chloride
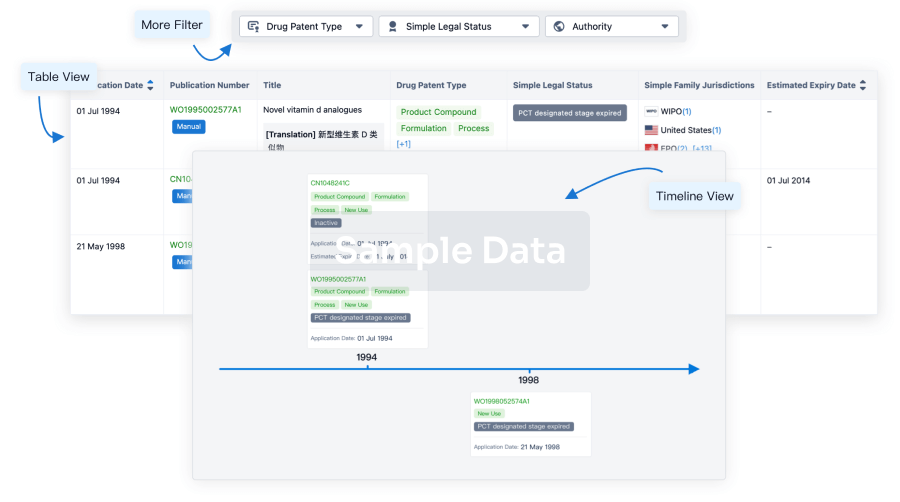
100 Patents (Medical) associated with Furosemide/Amiloride Chloride
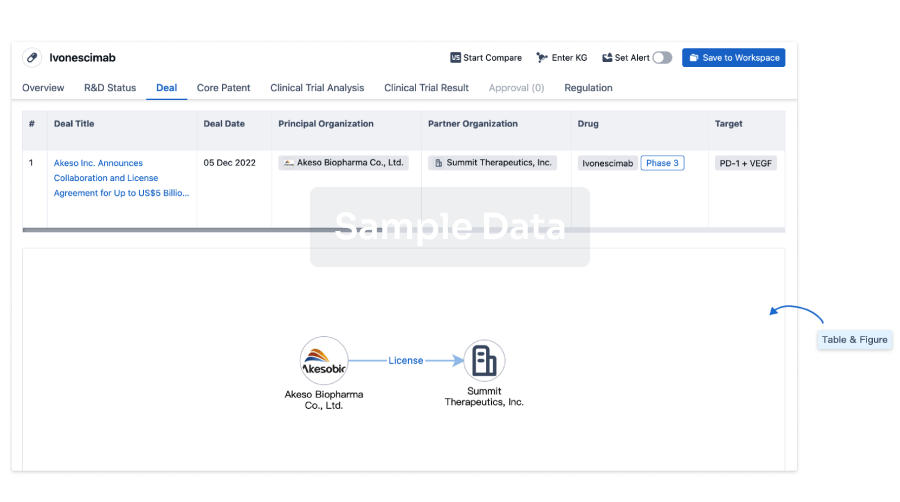
100 Deals associated with Furosemide/Amiloride Chloride