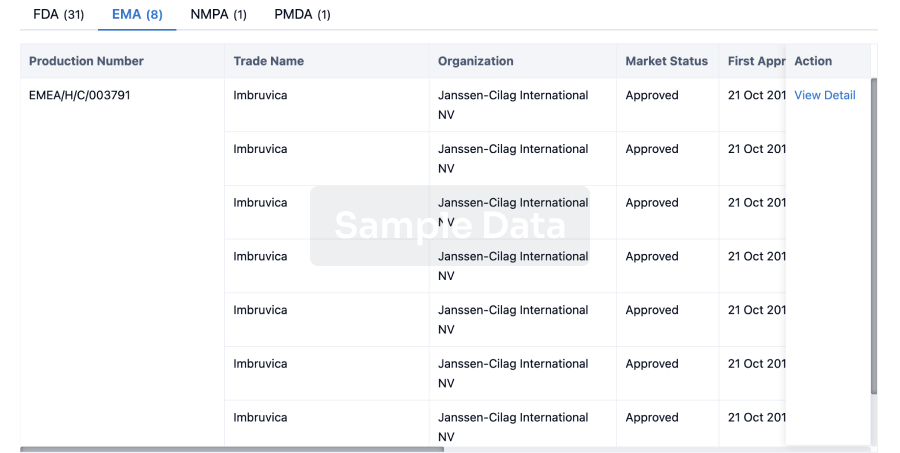
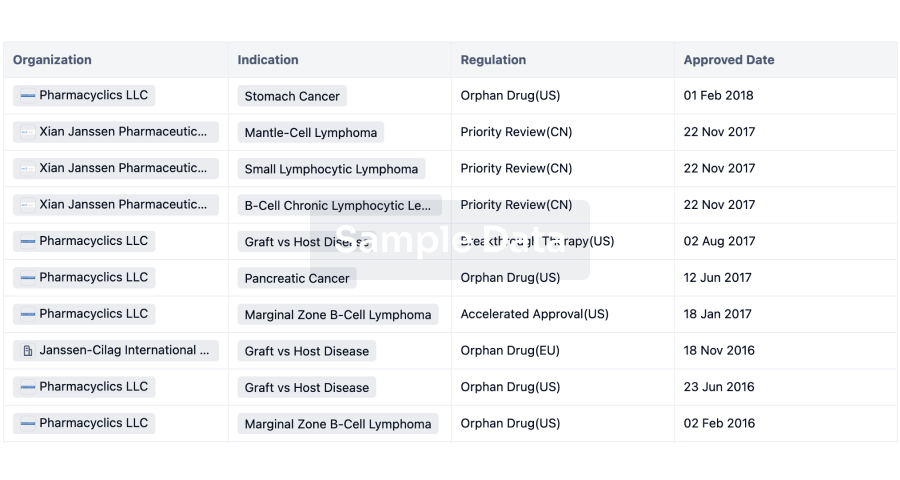
4
Clinical Trials associated with CMV pp65 Peptide-loaded Alpha-type-1 Polarized Dendritic Cell Vaccine(Roswell Park Cancer Institute)A Phase II Study of Intratumorally Injected Autologous Dendritic Cells (DCs) in PD-L1-Negative Treatment Naïve and in Refractory Metastatic Triple Negative Breast Cancer Patients
This phase II trial tests the safety, side effects, and whether dendritic cell-based treatment and pembrolizumab work in treating patients with triple negative breast cancer that has spread to other places in the body (metastatic) or cannot be removed by surgery (unresectable). The term triple-negative breast cancer refers to the fact that the cancer cells don't have estrogen or progesterone receptors (ER or PR) and also don't make any or too much of the protein called HER2 (the cells test "negative" on all 3 tests). Dendritic cell-based treatment works by boosting the immune system (a system in our bodies that protects us against infection) to recognize and destroy the cancer cells. Pembrolizumab, is an immune checkpoint inhibitor drug, that works by targeting molecules that act as a check and balance system for immune responses. Immune checkpoint inhibitor drugs are designed to either "unleash" or "enhance" the cancer immune responses that already exist by either blocking inhibitory molecules or by activating stimulatory molecules. Giving dendritic cell-based therapy and pembrolizumab may decrease symptoms and improve quality of life in patients with triple negative breast cancer.
A Phase II Study of Type-1 Polarized Dendritic Cell (aDC1) -Based Treatment in Combination With Tumor-Selective Chemokine Modulation (CKM: Interferon Alpha 2b, Rintatolimod and Celecoxib) in Melanoma Patients With Primary PD-1/PD-L1 Resistance
This phase II trial studies how well polarized dendritic cell (aDC1) based therapy, interferon alpha-2, rintatolimod, and celecoxib work together in treating patients with HLA-A2 positive (+) melanoma that has not responded to previous treatment (refractory). The aDC1 cell-based treatment contains white blood cells (dendritic cells or DCs) that stimulates the immune system. Interferon alpha-2 can improve the body's natural response to infections and other diseases. It can also interfere with the division of cancer cells and slow tumor growth. Rintalolimid may stimulate the immune system. Celecoxib is a drug that reduces pain. This study is being done to find out if the combination of the study cell-based treatment (aDC1 dendritic cells) and interferon alpha-2, rintatolimod, and celecoxib can prevent the growth and/or progression of melanoma.
A Phase I/IIa Safety and Immunologic Efficacy Trial of Intraperitoneal Induction of CTLs Combined With Alpha-Dendritic Cell Vaccine for Primary Ovarian Cancer
This phase I/IIa trial studies the side effects and best dose of a type of specialized immune cell (natural killer cell-like cytotoxic T-lymphocytes (CTLs) (nCTLs) and how well they work when given with a vaccine (alpha-type-1 polarized dendritic cells) in treating patients with stage II-IV ovarian, fallopian tube, or primary peritoneal cancer. nCTLs are immune cells that are isolated from each patient?s blood and "taught" in the laboratory how to recognize and eliminate tumor cells. These "educated" immune cells are then given back to the patient. An alpha-type-1 polarized dendritic cell vaccine is another population of "educated" immune cells that work to support the infused nCTLs. Giving nCTLS with a dendritic cell vaccine may work better in treating patients with ovarian, fallopian tube, or primary peritoneal cancer.
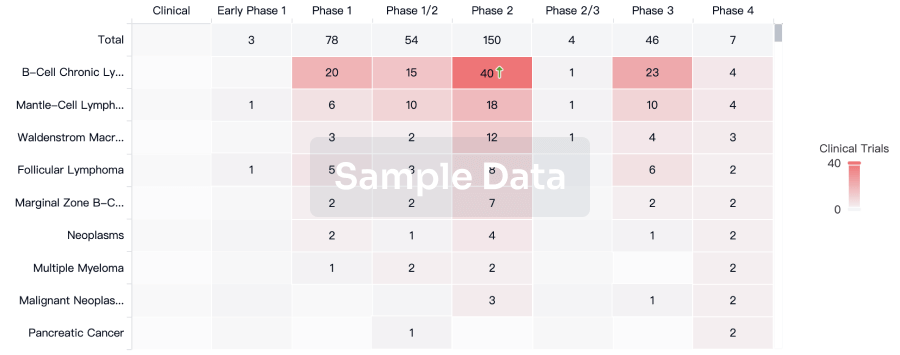
100 Clinical Results associated with CMV pp65 Peptide-loaded Alpha-type-1 Polarized Dendritic Cell Vaccine(Roswell Park Cancer Institute)
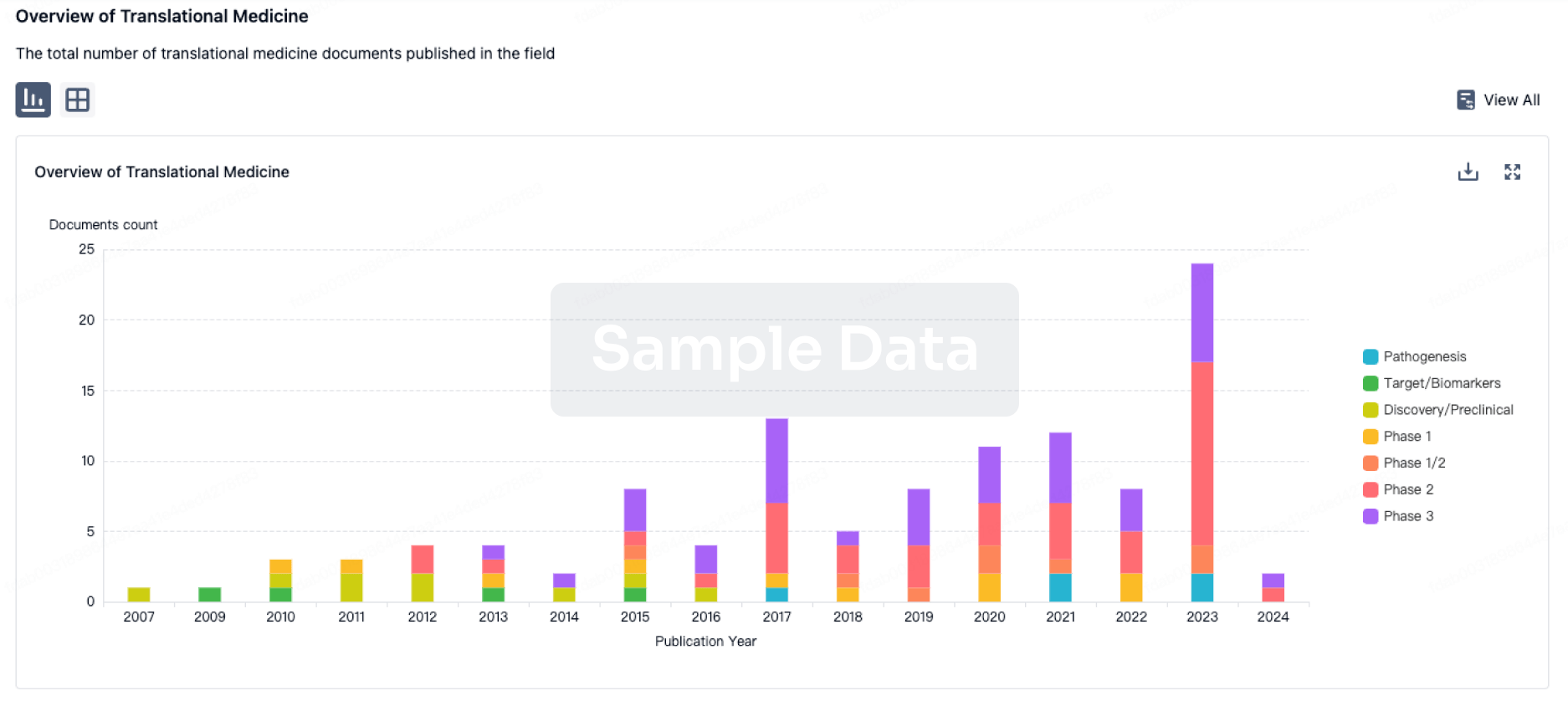
100 Translational Medicine associated with CMV pp65 Peptide-loaded Alpha-type-1 Polarized Dendritic Cell Vaccine(Roswell Park Cancer Institute)
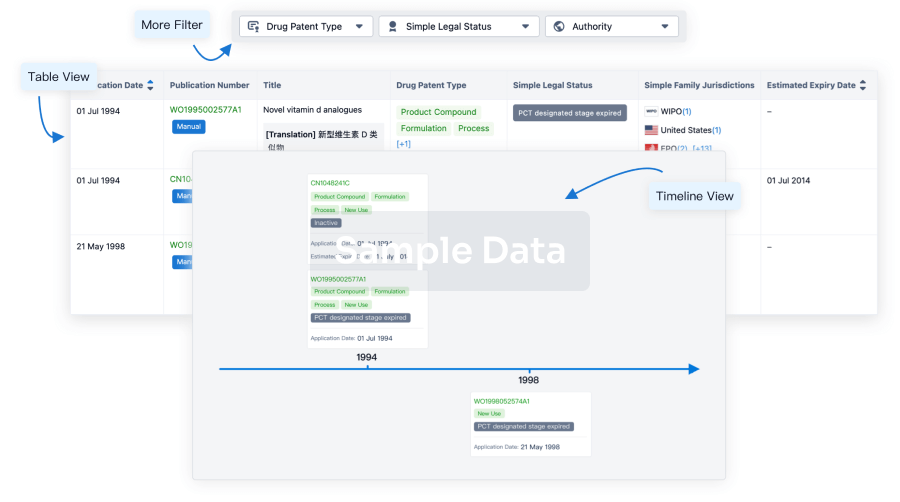
100 Patents (Medical) associated with CMV pp65 Peptide-loaded Alpha-type-1 Polarized Dendritic Cell Vaccine(Roswell Park Cancer Institute)
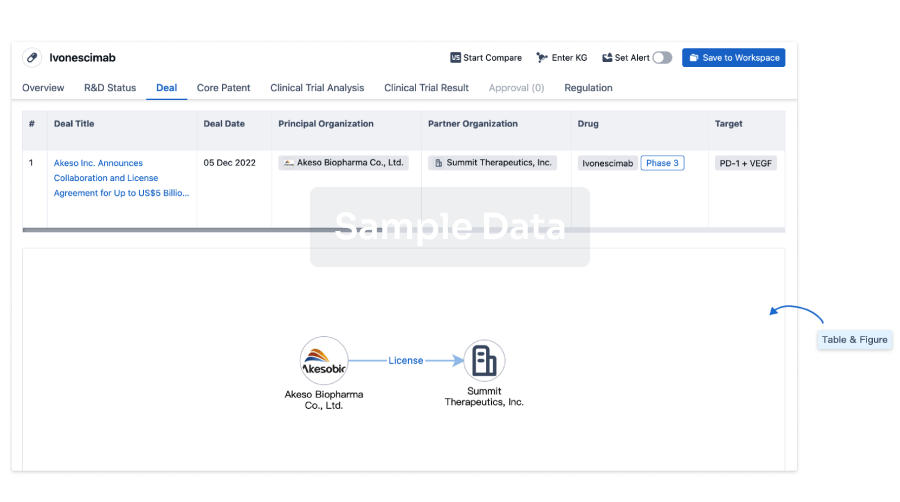
100 Deals associated with CMV pp65 Peptide-loaded Alpha-type-1 Polarized Dendritic Cell Vaccine(Roswell Park Cancer Institute)