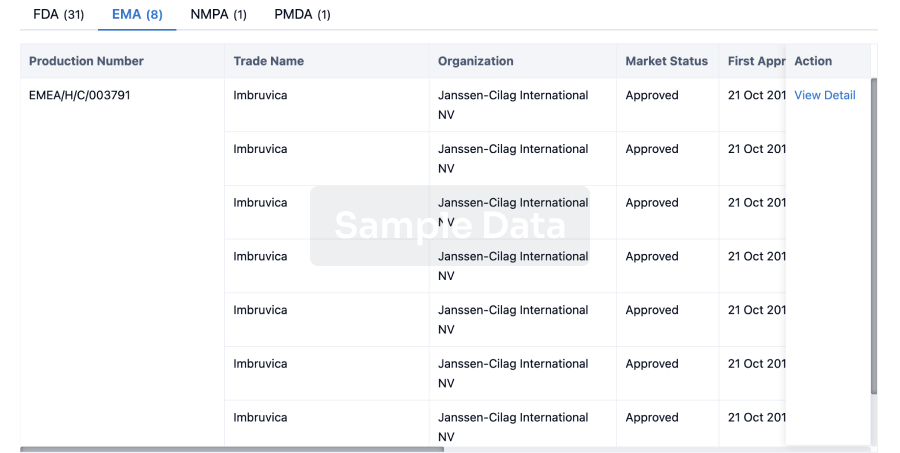
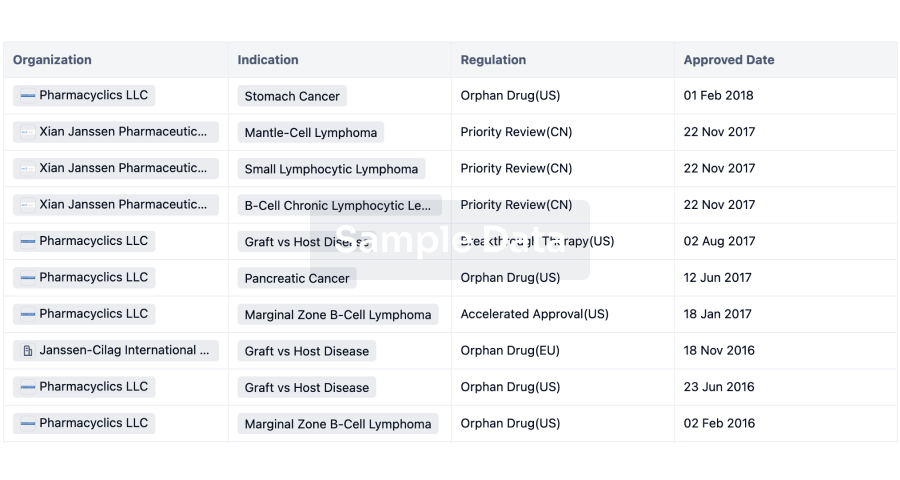
3
Clinical Trials associated with Anti-CD19 CAR-T cell therapy(Xuzhou Medical University)Prospective, Multicenter, Open, One-arm Clinical Study of the Safety and Efficacy of the R/R B-NHL Regimen With BTK Inhibitor+Anti-CD19 CAR-T Cells
To evaluate the ORR (CR+PR) of R/R B-NHL subjects treated with BTKi+Anti-CD19 CAR T cells.
A Phase I/II Study of Competitive Transfer of αCD19-TCRz-CD28 and αCD19-TCRz-CD137 Chimeric Antigen Receptor T-Cells in Patients With Refractory CD19+ B-lineage Leukemia/Lymphoma
This is a single-arm open-label phase I/II study to determine the relative superiority of αCD19-TCRζ-CD28 and αCD19-TCRζ-CD137 CAR-T Cells in safety, efficacy and engraftment potential in patients with CD19+ B-lineage leukemia and lymphoma. Recently, cancer immunotherapy, treatments aiming to arm patients with immunity specifically against cancer cells, has emerged as a promising therapeutic strategy. Clinical trials utilizing CARs against B cell malignancies have demonstrated remarkable potential. In this trial, all subjects will be competitively infused with αCD19-TCRz-CD28 and αCD19-TCRz-CD137 CAR-T cells in equal number to test a hypothesis that CD137-costimulation can promote the persistence and engraftment of CAR-T cells and this superiority can lead to improved progression-free survival.
A Two-Arm, Single-Center, Open-Label Pilot Study of IL-2 Programmed or IL-7/IL-15 Programmed Anti-CD19:TCRz:CD28 T-cells in Patient With CD19-Positive Lymphoma That is Resistant or Refractory to Chemotherapy
The goal of this clinical trial is to study how approaches for manufacturing chimeric antigen receptor (CAR)-modified T (CAR-T) cells affect their in vivo persistence and therapeutic efficacy against B lymphoma. Recently, cancer immunotherapy, treatments aiming to arm patients with immunity specifically against cancer cells, has emerged as a promising therapeutic strategy. Among the many emerging immunotherapeutic approaches, clinical trials utilizing CARs against B cell malignancies have demonstrated remarkable potential. CARs combine the variable region of an antibody with T-cell signaling moieties to confer T-cell activation with the targeting specificity of an antibody. Thus, CARs are not MHC-restricted so they are not vulnerable to MHC down regulation by tumors. However, defined by the activation and contraction program of their mother cells, the persistency and function of CAR-T cells are also restricted by the protocol of manufacturing. Previous clinical studies largely utilized interleukin-2 (IL-2) for the ex vivo expansion of CAR-T cells, which preferentially generate CAR-T cells with characteristics of terminally differentiated effector cells. Our preliminary data indicated that two common gamma chain cytokines, IL-7 and IL-15, can help to selectively expand CAR-T cells with various memory phenotypes. CAR-T Cells prepared under this condition resulted in improved therapeutic efficacy in preclinical animal models. This clinical investigation is to test a hypothesis whether IL-7/IL-15-programmed anti-CD19 CAR-T cells persist longer in lymphoma patients after infusion and whether the persistency of CAR-T cells can lead to improved anti-lymphoma efficacy.
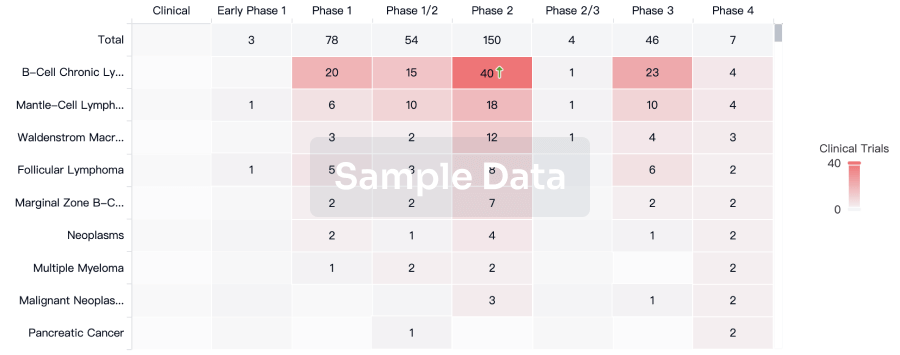
100 Clinical Results associated with Anti-CD19 CAR-T cell therapy(Xuzhou Medical University)
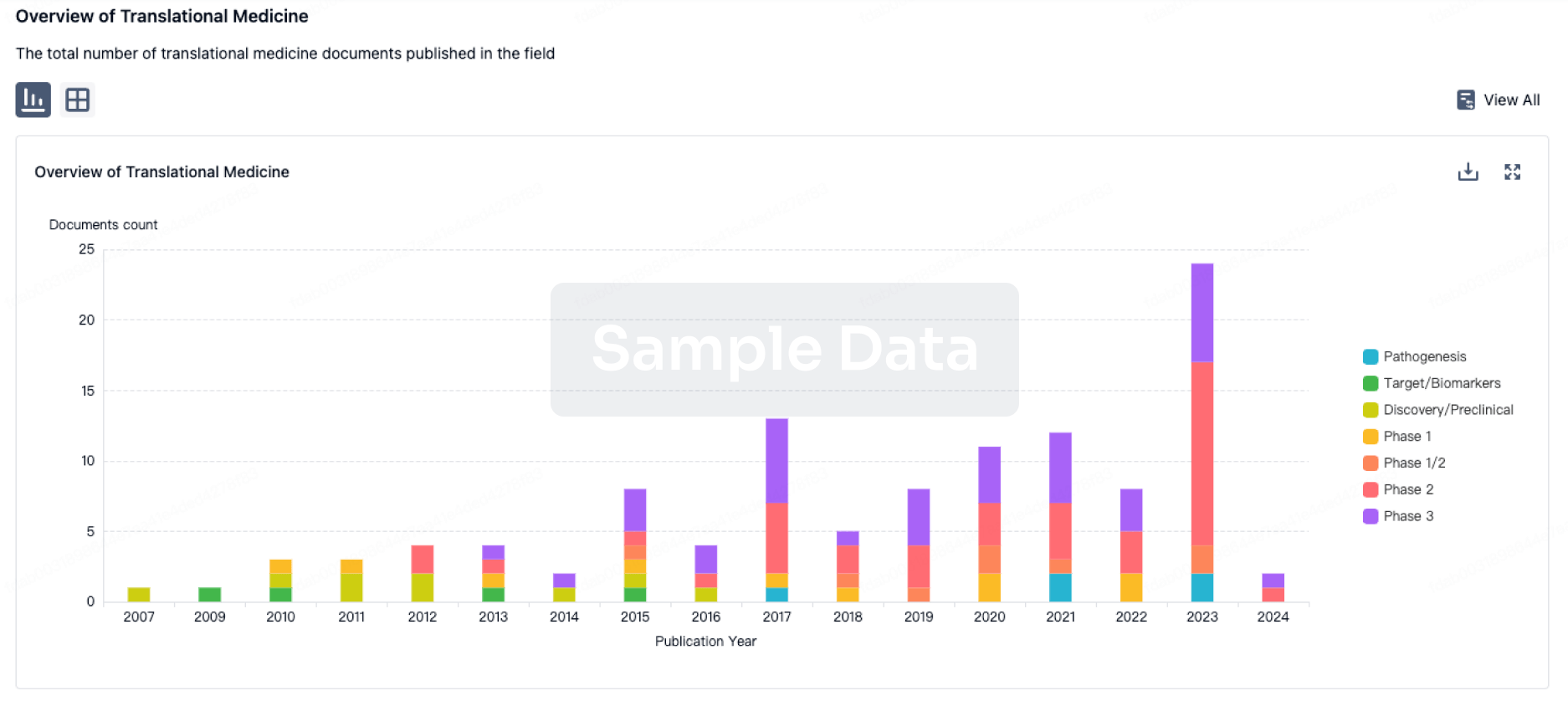
100 Translational Medicine associated with Anti-CD19 CAR-T cell therapy(Xuzhou Medical University)
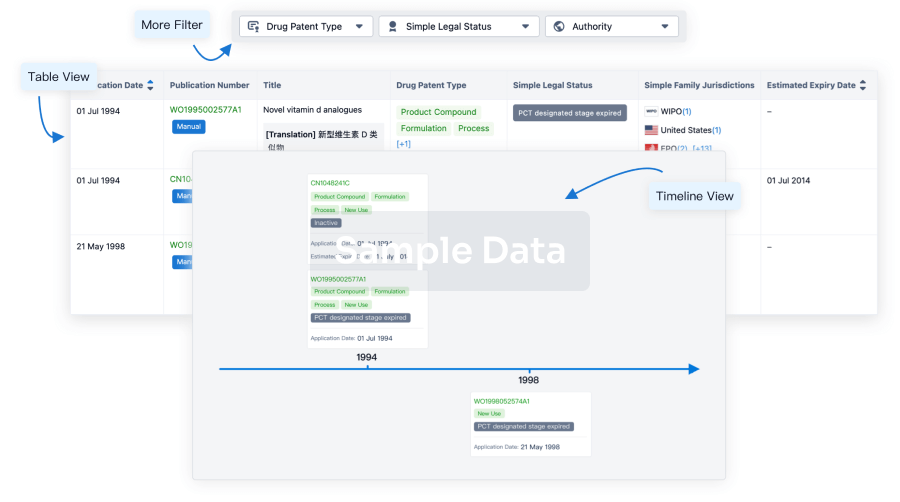
100 Patents (Medical) associated with Anti-CD19 CAR-T cell therapy(Xuzhou Medical University)
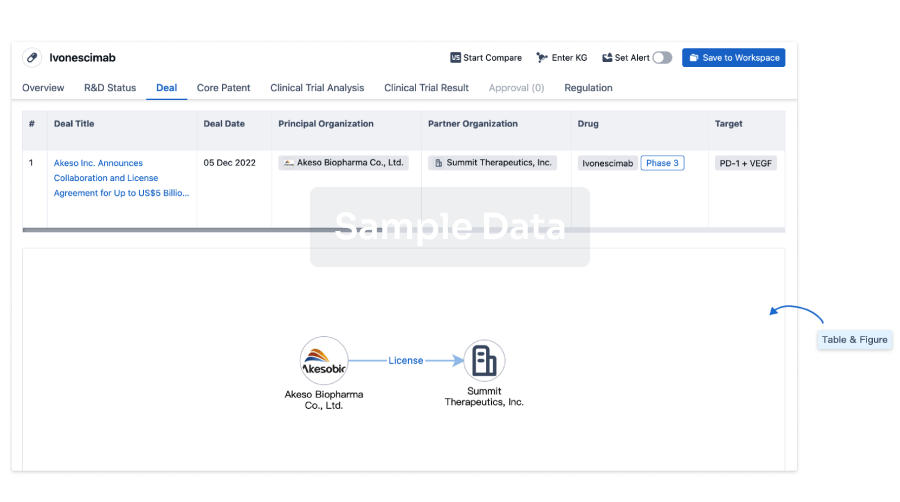
100 Deals associated with Anti-CD19 CAR-T cell therapy(Xuzhou Medical University)