CARsgen’s CAR-T therapy for multiple myeloma gains NMPA approval
04 Mar 2024
Cell TherapyOrphan DrugPhase 2ImmunotherapyBreakthrough Therapy
Preview
Source: Pharmaceutical Technology
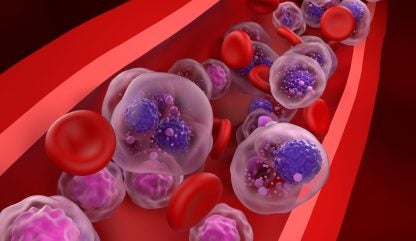
The CAR-T cell therapy involves modifying patients’ T-cells to express a CAR that targets BCMA, a protein found on multiple myeloma cell surface. Credit: Nemes Laszlo / Shutterstock.com.

Preview
Source: Pharmaceutical Technology
The National Medical Products Administration (NMPA) of China has approved CARsgen Therapeutics’ new drug application for zevorcabtagene autoleucel to treat multiple myeloma.
The B cell maturation antigen (BCMA)-targeted autologous chimeric antigen receptor (CAR)-T product candidate is indicated to treat adults with relapsed or refractory multiple myeloma who have not responded to three lines of previous therapies.
The therapy involves modifying patients’ T-cells to express a CAR that targets BCMA, a protein commonly found on the surface of multiple myeloma cells.
The cells are then infused back into the patient to attack and kill the cancer cells.
The approval is based on results from the LUMMICAR STUDY 1, a Phase II clinical trial conducted in China.
See Also:Oncology emerges as dominant therapy area for CRISPR technology

Preview
Source: Pharmaceutical Technology
Nanobiotix files patent for porous, high-z, carbon-free particles for oncology therapy

Preview
Source: Pharmaceutical Technology
The multicentre open-label, single-arm trial demonstrated that zevorcabtagene autoleucel has “encouraging” efficacy and a favourable safety profile.
CARsgen is also conducting a separate Phase Ib/II LUMMICAR STUDY 2 in North America. This trial aims to assess the efficacy and safety of CAR-T therapy in the same patient population.
In 2019, the therapy received multiple designations from international regulatory agencies, including regenerative medicine advanced therapy and orphan drug designations from the US Food and Drug Administration.
The European Medicines Agency granted priority medicines and orphan medicinal product designations in 2019 and 2020 respectively.
The NMPA awarded zevorcabtagene autoleucel breakthrough therapy designation in 2020.
In a strategic move to facilitate the marketing of zevorcabtagene autoleucel in mainland China, CARsgen entered into a partnership with Huadong Medicine in January 2023.
Huadong Medicine gained sole rights to commercialise the CAR-T therapy within the territory.
CARsgen Therapeutics founder, board chairman, CEO and chief scientific officer Dr Zonghai Li stated: “We look forward to zevorcabtagene autoleucel bringing renewed hope to adult patients with relapsed or refractory multiple myeloma, thereby improving their survival.
Cell & Gene Therapy coverage on Pharmaceutical Technology is supported by Cytiva.
Editorial content is independently produced and follows the highest standards of journalistic integrity. Topic sponsors are not involved in the creation of editorial content.
Free WhitepaperCell and gene therapies: Pipe dream to pipeline
The cell and gene industry is gaining momentum, with a new wave of therapies promising to transform the way doctors treat, and even cure, disease. In this report, Cytiva and GlobalData have collaborated to explore the rise of the cell and gene therapy industries, the current state of the market, present and future opportunities for advancement, and the challenges that lie ahead.

Preview
Source: Pharmaceutical Technology
-->
By downloading this case study, you acknowledge that GlobalData may share your information with Cytiva Thematic and that your personal data will be used as described in their Privacy Policy
For more details,please visit the original website
The content of the article does not represent any opinions of Synapse and its affiliated companies. If there is any copyright infringement or error, please contact us, and we will deal with it within 24 hours.
Targets
Hot reports
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Leverages most recent intelligence information, enabling fullest potential.




