A Comprehensive Review of nitrendipine's R&D Innovations and Drug Target Mechanism
Nitrendipine's R&D Progress
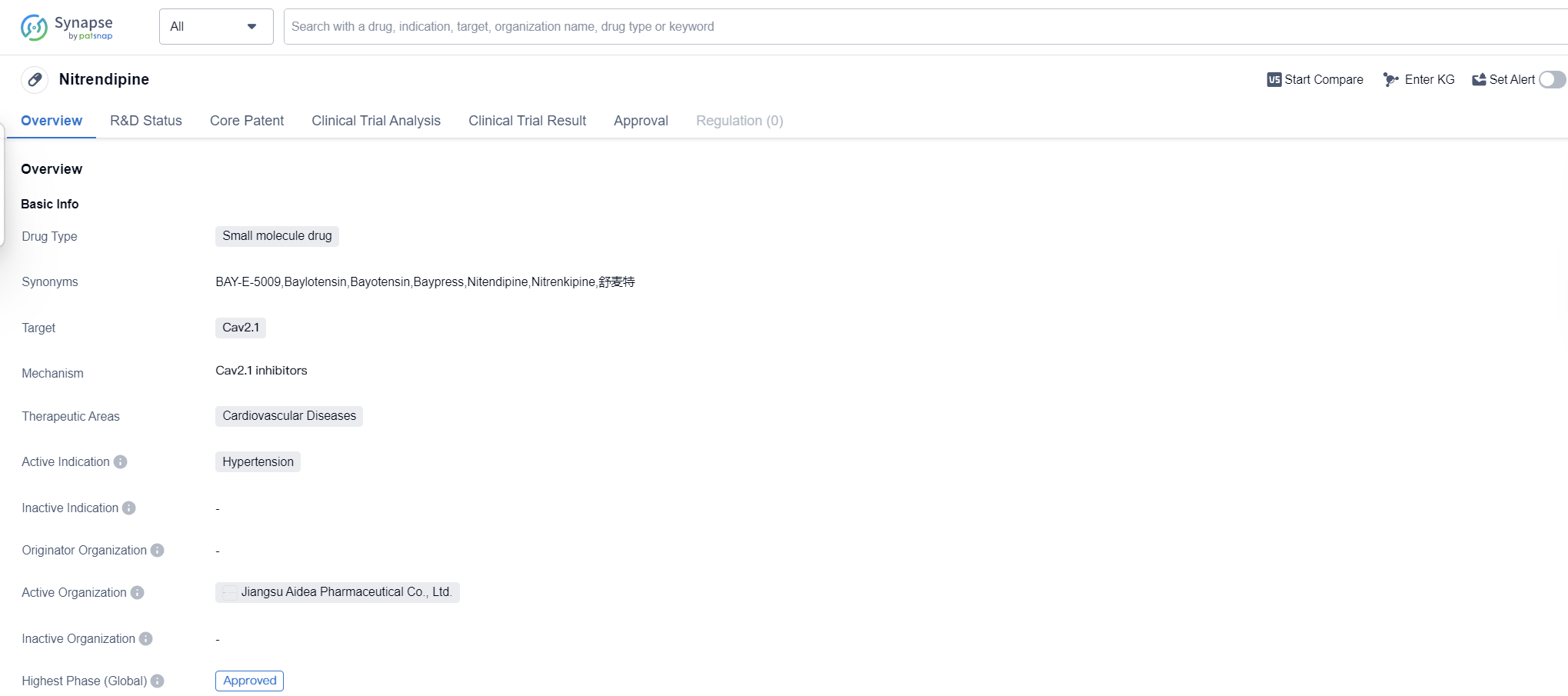
Nitrendipine is a small molecule drug that falls under the therapeutic area of cardiovascular diseases. It specifically targets Cav2.1, making it a potential treatment option for hypertension. The drug has reached the highest phase of development which is approved globally. This approval status also suggests that the drug has demonstrated positive results in clinical trials and has met the necessary regulatory requirements.
As a small molecule drug, Nitrendipine is designed to interact with specific molecular targets in the body, in this case, Cav2.1. Cav2.1 is a calcium channel that plays a crucial role in regulating blood pressure. By targeting this channel, Nitrendipine aims to reduce hypertension, a condition characterized by high blood pressure.
Since its first approval in 1985, Nitrendipine has likely been used as a treatment option for hypertension in patients worldwide. Its long-standing presence in the market indicates that it has established a track record of effectiveness and safety over the years.
The therapeutic area of cardiovascular diseases encompasses a wide range of conditions related to the heart and blood vessels. Hypertension, or high blood pressure, is one of the most prevalent cardiovascular diseases globally. It is a chronic condition that can lead to serious health complications if left untreated. Nitrendipine's approval for the treatment of hypertension suggests that it has shown promising results in lowering blood pressure and managing this condition.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
Mechanism of Action for nitrendipine: Cav2.1 inhibitors
Cav2.1 inhibitors are a type of drugs that target and inhibit the activity of the Cav2.1 calcium channels. Cav2.1, also known as P/Q-type calcium channels, are voltage-gated calcium channels found in the central nervous system. These channels play a crucial role in regulating neurotransmitter release in neurons.
By inhibiting Cav2.1 channels, Cav2.1 inhibitors can modulate calcium signaling and neurotransmitter release. This can have therapeutic implications in various neurological conditions, including epilepsy, chronic pain, and movement disorders. By reducing the excessive release of neurotransmitters, Cav2.1 inhibitors can help alleviate symptoms associated with these conditions.
From a biomedical perspective, Cav2.1 inhibitors are being studied and developed as potential pharmacological interventions for neurological disorders. Researchers are investigating their efficacy, safety, and potential side effects to determine their clinical utility. These inhibitors may offer a targeted approach to modulating calcium signaling and neurotransmitter release, providing a new avenue for therapeutic interventions in the field of biomedicine.
Drug Target R&D Trends for nitrendipine
Cav2.1, also known as P/Q-type calcium channels, plays a crucial role in the human body. These channels are primarily found in the central nervous system and are responsible for regulating the release of neurotransmitters. By controlling the influx of calcium ions into neurons, Cav2.1 channels contribute to the transmission of signals between nerve cells, ultimately influencing various physiological processes such as muscle contraction, hormone secretion, and synaptic plasticity. Dysregulation or dysfunction of Cav2.1 channels has been associated with neurological disorders like epilepsy, ataxia, and migraine. Understanding the role of Cav2.1 channels provides valuable insights for the development of therapeutic interventions targeting these channels to treat related disorders.
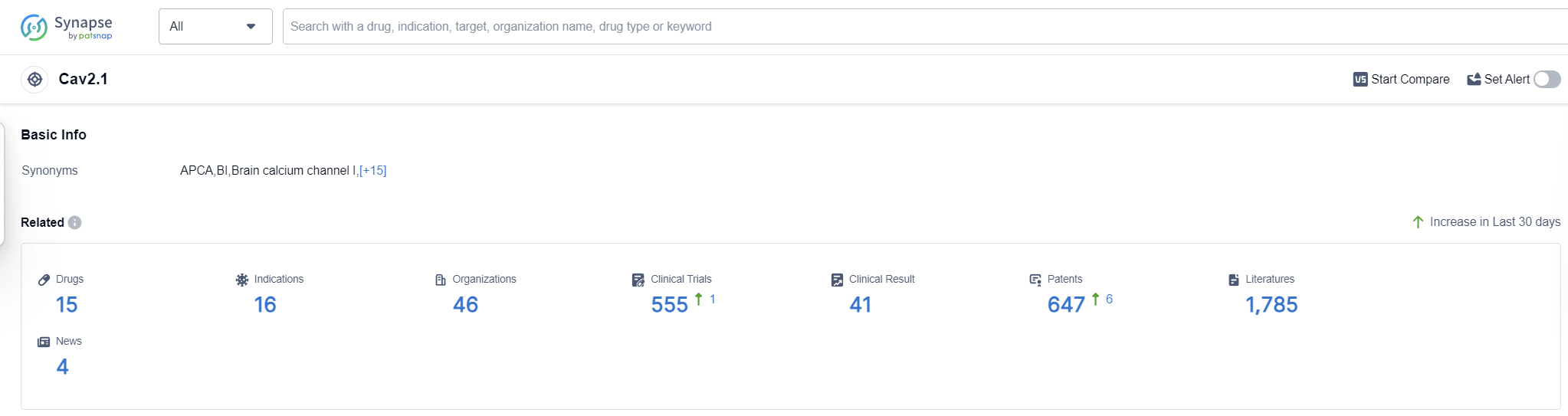
According to Patsnap Synapse, as of 16 Sep 2023, there are a total of 15 Cav2.1 drugs worldwide, from 46 organizations, covering 16 indications, and conducting 555 clinical trials.
Based on the analysis of the provided data, the current competitive landscape of target Cav2.1 in the pharmaceutical industry is characterized by the presence of multiple companies with drugs in the "Approved" phase. The most common indications for these drugs are hypertension and angina pectoris. Small molecule drugs are progressing rapidly, indicating a focus on this drug type for Cav2.1. China, the European Union, Japan, and the United States are the leading countries/locations in terms of drug development for Cav2.1, with China showing significant progress. Further analysis is required to determine the specific R&D progress, efficacy, and safety profiles of the drugs under target Cav2.1.
👇Please click on the picture link below for free registration or log in directly if you have a freemium account, you can browse the latest research progress on drugs, indications, organizations, clinical trials, clinical results, and drug patents related to this target
Conclusion
In summary, Nitrendipine is a small molecule drug that targets Cav2.1 and is approved for the treatment of hypertension. Its approval status in the global markets, along with its long history since 1985, indicates its effectiveness and safety as a treatment option for patients with high blood pressure.