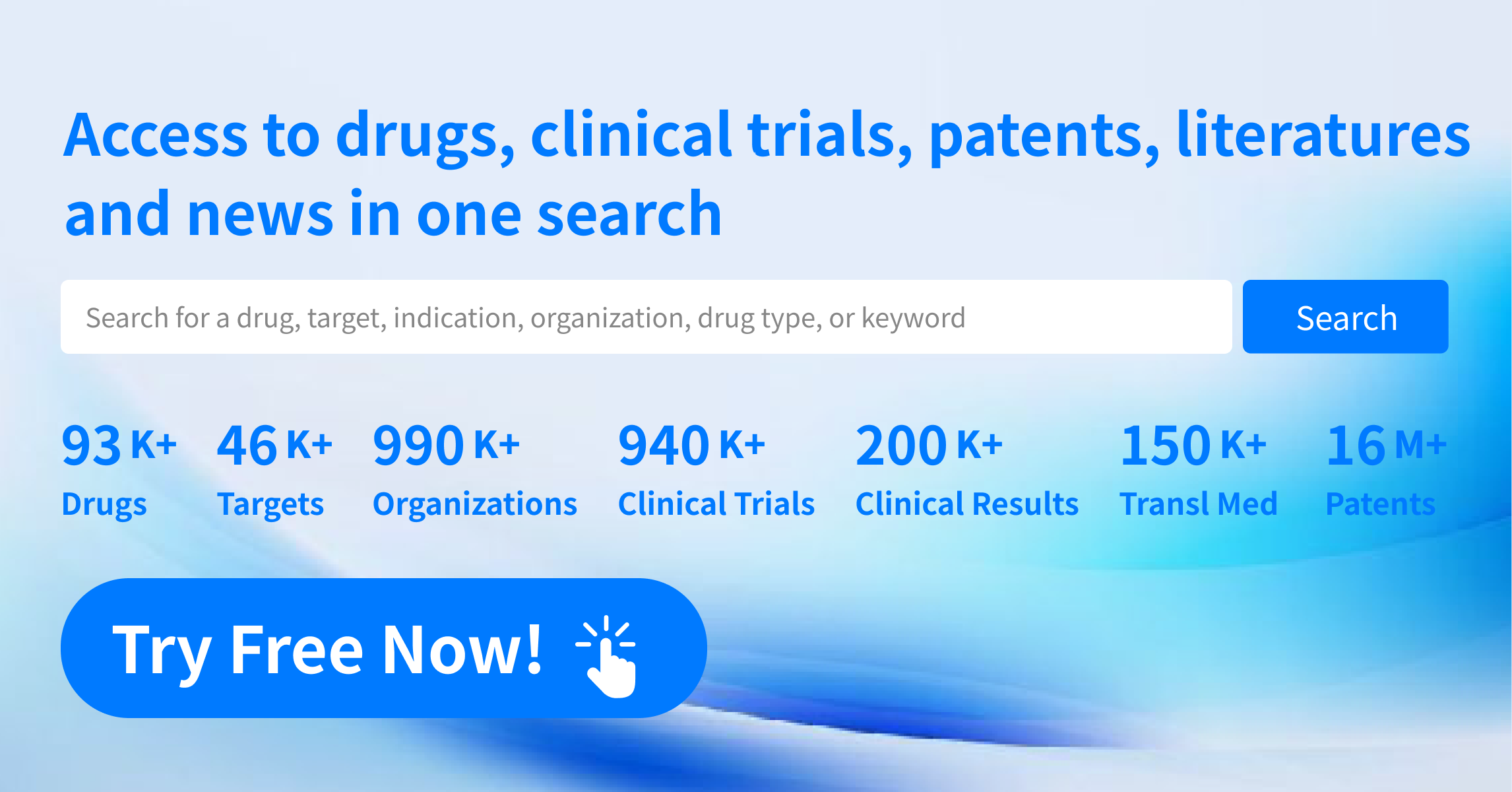
Global New Drug Research and Development Progress Weekly Report(8.26-9.1)
Global Pharmaceutical Research and Development Progress
1.Wantai Biological Files for Market Authorization of Its Nine-Valent HPV Vaccine
On August 26, the CDE (Center for Drug Evaluation) website revealed that Wantai Biological's application for the market approval of its nine-valent human papillomavirus (HPV) vaccine (produced in E. coli) has been accepted. In April of this year, Wantai Biological announced, in collaboration with Xiamen University, the completion and preliminary data analysis of the blinding phase of the "Phase III Main Clinical Trial V8 Visit (out of 12 visits set) of the Recombinant HPV Types 6/11/16/18/31/33/45/52/58 Nine-Valent HPV Vaccine (E. coli)." This study is a multicenter, randomized, double-blind, bivalent HPV vaccine-controlled, phase III clinical trial. The preliminary data analysis showed that the primary endpoint results, which accumulated to the planned 12-month persistent infection event numbers, met the expected outcomes. The data obtained from this Phase III clinical trial unblinding covers only primary outcomes up to the V8 visit, with comprehensive and intensive analyses planned as per the study protocol and statistical analysis plan.
2.Semaglutide Receives Market Approval Application Acceptance in China for New Indication
On August 26, the website of the National Medical Products Administration's Drug Evaluation Center showed that Novo Nordisk's semaglutide injection for a new indication has been accepted. On August 7, local time, Novo Nordisk released its mid-year report for 2024, mentioning in the investor information published on its website that it would be filing for a chronic kidney disease (CKD) indication for semaglutide in China during the third quarter of this year. This acceptance likely pertains to CKD. Semaglutide is among the leading GLP-1 medications globally, already approved for diabetes and weight loss indications, with Novo Nordisk actively expanding its indications. In March this year, Novo Nordisk announced that in the FLOW double-blind clinical trial, patients with type 2 diabetes and CKD treated with the glucose-lowering version of semaglutide (1.0mg) injection showed a 24% reduction in risk for worsening kidney disease, cardiovascular diseases, and death from kidney disease compared to placebo, thereby meeting the trial's primary endpoint. According to Novo Nordisk, approximately 40% of individuals with type 2 diabetes suffer from CKD, potentiating semaglutide as a pioneering GLP-1 therapy for such patients.
3.The EU has approved Roche's new generation of monthly subcutaneous injection therapy for PNH
On August 27th, according to a notification from Roche, the European Commission has approved the new generation C5 complement antibody, PiaSky® (crovalimab), for the treatment of adults and adolescents (aged 12 and above, weighing at least 40 kg) with paroxysmal nocturnal hemoglobinuria (PNH), regardless of whether they have previously received C5 inhibitor treatment. This makes PiaSky the first monthly subcutaneous (SC) injection therapy for PNH in the EU, allowing patients to self-administer the injections after adequate training. Piasky, a humanized monoclonal antibody that targets the complement protein C5, is engineered to block the complement system in the human immune system. This product is engineered through monoclonal antibody recycling technology, which induces C5 protein degradation upon binding to C5, subsequently allowing the antibody to be released extracellularly through its interaction with the FcRn receptor, and then bind to other C5 proteins. Therefore, PiaSky can achieve rapid and sustained inhibition of the complement pathway at a lower dosage. The approval is based on Phase 3 COMMODORE 2 study results, which demonstrate that PiaSky, given once every 4 weeks subcutaneously, provides good disease control and tolerability in PNH patients who have not previously received C5 inhibitor treatment. PiaSky's efficacy was comparable to that of the C5 inhibitor Eculizumab, which requires bi-weekly intravenous injections. The incidence of adverse events in patients treated with PiaSky was similar to that of the active comparator (78% and 80%, respectively).
4.Johnson & Johnson's significant therapy Rybrevant receives EU approval
On August 27th, Johnson & Johnson announced that the European Commission (EC) has approved its bispecific antibody, Rybrevant (amivantamab), for use in combination with chemotherapy (Carboplatin and Pemetrexed) for the treatment of adult patients with advanced non-small cell lung cancer (NSCLC) harboring EGFR exon 19 deletions (ex19del) or exon 21 L858R substitution mutations. These patients had previously undergone treatment including EGFR tyrosine kinase inhibitors (TKIs) and experienced disease progression. A press release noted that the combination of amivantamab and chemotherapy has shown a significant improvement in progression-free survival (PFS) for the first time in this patient group compared to chemotherapy alone. The approval was chiefly based on the results from the Phase 3 clinical study MARIPOSA-2, which evaluated the efficacy and safety of amivantamab in combination with chemotherapy in patients with locally advanced or metastatic NSCLC harboring EGFR ex19del or L858R substitution mutations, following or during disease progression after treatment with Osimertinib. Analysis indicated that the trial achieved its primary endpoint, significantly reducing the risk of disease progression or death by 52% in patients receiving the combination therapy compared to those receiving chemotherapy alone, with a PFS of 6.3 months versus 4.2 months for the control group (HR=0.48; 95% CI: 0.36–0.64; P<0.001). Additionally, the objective response rate (ORR) in the combination therapy group was 64%, compared to 36% in the control group.
5.Genrix Biopharmaceutical Announces Approval of Xeligekimab for Marketing
On August 27, Genrix Biopharmaceutical announced that the National Medical Products Administration (NMPA) had approved the marketing of their independently developed Class 1 new drug, Xeligekimab injection, intended for the treatment of adult patients with moderate to severe plaque psoriasis who are candidates for systemic therapy or phototherapy. Xeligekimab (GR1501) is a recombinant fully human monoclonal antibody targeting IL-17A. It binds specifically to the IL-17A protein in the serum, blocking the interaction between IL-17A and IL-17RA, thus inhibiting the initiation and progression of inflammation and achieving therapeutic effects in autoimmune diseases such as plaque psoriasis and axial spondyloarthritis, which overexpress IL-17A. Besides the indication for moderate to severe plaque psoriasis, the company has filed a New Drug Application (NDA) in January 2024 for radiographic axial spondyloarthritis, which was accepted. According to data published in February this year in the British Journal of Dermatology (BJD), at week 12, patients treated with Xeligekimab showed significantly higher percentages achieving PGA 0/1 (74.4% vs 3.6%, P<0.001) and PASI 75 (90.7% vs 8.6%, P<0.001) compared to the placebo group. Furthermore, response rates for PGA 0/1 and PASI 75/90 were maintained through week 52. Notably, 30.2% of patients in the Xeligekimab group achieved complete remission (PASI 100) by week 12, and 59.7% by week 52. Most adverse reactions were mild to moderate, the most common being upper respiratory tract infections (14.0%), hyperlipidemia (12.1%), and injection site reactions (10.2%). The rate of treatment-emergent adverse events (TEAEs) was similar during the induction and maintenance phases.
6.MEK Inhibitor Mirdametinib Receives FDA Priority Review Designation
On August 28, SpringWorks Therapeutics announced that the U.S. FDA had accepted the New Drug Application (NDA) for its investigational MEK inhibitor, mirdametinib, for the treatment of adults and children with neurofibromatosis type 1-associated plexiform neurofibromas (NF1-PN). The FDA has also granted the application priority review status, with a PDUFA target date of February 28, 2025. In addition, the European Medicines Agency (EMA) has completed the validation of the marketing authorization application (MAA) for mirdametinib. According to the press release, mirdametinib has the potential to be the first approved medication for adult patients with NF1-PN and could become a best-in-class therapy for children with NF1-PN. Mirdametinib is an oral small molecule MEK inhibitor targeting MEK1 and MEK2. Both the U.S. FDA and the European Commission have granted orphan drug designation to mirdametinib for the treatment of NF1. Furthermore, the FDA has awarded Fast Track designation for the treatment of NF1-PN in patients aged 2 years and older.
7.Novartis's siRNA Therapy for Low/Moderate-Risk Atherosclerotic Cardiovascular Disease Succeeds in Phase III Study
On August 28, Novartis announced positive results from its Phase III V-MONO study, aimed at evaluating the efficacy and safety of biannual administration of Leqvio (inclisiran) in comparison with placebo and ezetimibe for patients with low to moderate-risk atherosclerotic cardiovascular disease (ASCVD) who have not received lipid-lowering treatment previously. The results demonstrated that the study met its primary endpoint, with Leqvio significantly reducing levels of low-density lipoprotein cholesterol (LDL-C). The V-MONO trial was a six-month, randomized, double-blind, placebo-controlled, and active-comparator study involving 350 patients, divided into three groups: Leqvio alone (n=174), ezetimibe alone (n=89), and placebo (n=87). The primary endpoint was the percentage change in LDL-C levels from baseline at day 150 in the Leqvio group compared to both the placebo and ezetimibe groups. Results indicated a clinically and statistically significant reduction in LDL-C levels in the Leqvio group compared to the placebo and ezetimibe groups. Novartis acquired global rights for Leqvio from Alnylam Pharmaceuticals in 2013. Leqvio has now been approved in nearly 100 countries and regions, including the US, EU, Japan, and China.
8.Tonghua Golden-Horse Submits New Oral Alzheimer's Drug for Market Approval
On August 29, the CDE website revealed that Tonghua Golden-Horse has applied for market approval of Octahydroaminoacridine succinate for the treatment of Alzheimer's disease. Octahydroaminoacridine tablets are a small molecule acetylcholinesterase inhibitor, developed by Tonghua Golden-Horse with full intellectual property rights, which concurrently inhibits both acetylcholinesterase and butyrylcholinesterase, primarily for the treatment of mild to moderate Alzheimer's disease. In vitro tests have shown that the inhibitory effect of Octahydroaminoacridine tablets on these cholinesterases is several times greater than that of conventional first-line medications, thus offering better clinical effects. Phase I and II clinical trials have also validated its clinical efficacy; furthermore, the safety profile of Octahydroaminoacridine tablets has proven to be relatively more reliable and safer. In September 2023, Tonghua Golden-Horse announced the successful completion of its Phase III study for the treatment of mild to moderate Alzheimer's disease, which began in January 2017 and completed patient enrollment in August 2021. The results indicated a significant clinical improvement in the Alzheimer's Disease Assessment Scale-Cognitive Subscale (ADAS-cog). The incidence of adverse events in the Octahydroaminoacridine group was lower than that in the two control groups (placebo and donepezil hydrochloride tablets), with results reaching statistical significance (P<0.001).
9.Johnson & Johnson Submits New Myasthenia Gravis Drug for Approval
On August 29, Johnson & Johnson announced that it has submitted a Biologics License Application (BLA) to the U.S. FDA, seeking the first-ever global approval for its investigational neonatal Fc receptor (FcRn)-targeted antibody therapy, nipocalimab, for the treatment of generalized myasthenia gravis (gMG). According to a previous press release, this therapy is the first FcRn inhibitor that, when used in combination with standard of care (SOC) in antibody-positive gMG patients (including anti-AChR+, anti-MuSK+, and anti-LRP4+ patients), significantly outperformed placebo in improving patients' daily living activities (MG-ADL) scores. The press release states that nipocalimab is the first FcRn inhibitor to demonstrate sustained disease control over a six-month treatment period (administered biweekly) by improving MG-ADL scores compared to placebo plus SOC. Nipocalimab is a potentially "best-in-class" antibody therapy targeting the neonatal Fc receptor. It works by binding to FcRn, preventing autoantibodies taken up by monocytes and endothelial cells from being released back into the bloodstream, where they are instead degraded within the cells. Studies have shown that it can reduce circulating IgG levels, including pathogenic autoantibodies, by more than 75%. This antibody therapy holds promise for treating multiple autoimmune diseases mediated by autoantibodies.
10.BridgeBio Releases Phase 3 Clinical Results of Potential Breakthrough Small Molecule Therapy
On August 30, BridgeBio Pharma presented further data on its investigational therapy, acoramidis, in patients with transthyretin amyloid cardiomyopathy (ATTR-CM) complicated by cardiomyopathy, during the Phase 3 ATTRibute-CM trial and its open-label extension (OLE) study at the 2024 European Society of Cardiology (ESC) Congress. Acoramidis is a next-generation, potent oral small molecule stabilizer of transthyretin (TTR) protein. The latest data showed that among participants who switched from the active control group and placebo group in the ATTRibute-CM trial to receive acoramidis in the OLE study, the mean serum TTR levels increased by 3.0 mg/dL (n=21, p=0.01) at Month 1 of the switch and by 3.4 mg/dL (n=18, p=0.01) at Month 6. Based on the positive results of the ATTRibute-CM trial, BridgeBio has submitted a New Drug Application (NDA) to the FDA, with a review decision expected by November 29, 2024. BridgeBio has granted Bayer exclusive rights to commercialize acoramidis in Europe for the treatment of ATTR-CM.
For more information on the progress of drug development, please follow the Synapse database.
Dynamics of Global Pharmaceutical Trade Cooperation
1.UCB Announces Sale of Neurology and Allergy Business in China
On August 26, UCB announced a strategic divestiture agreement in China, deciding to adjust its strategic focus in the region by selling its mature neurology and anti-allergy product businesses to CBC Group and Mubadala Investment Company. The new partnership will form a company to scale up operations. The sale includes mature neurology products such as Levetiracetam, Lacosamide, and Rotigotine, as well as anti-allergy products Cetirizine Hydrochloride and Levocetirizine Hydrochloride, along with a production facility located in Zhuhai. The transaction, expected to be completed by the fourth quarter of 2024, values at $680 million. Until then, all assets and operations will continue to be owned by UCB and business will proceed as usual. Concurrently, UCB disclosed progress in innovation, actively preparing to introduce a series of innovative drugs in the fields of immunology, neurology, and rare diseases in the Chinese market.
2.Navigator Medicines Completes $100 Million Series A Funding Round
On August 28th, Navigator Medicines announced the completion of its $100 million Series A financing, along with the acquisition of licensing rights from IMBiologics for NAV-240 and the OX40L-targeted research and development pipeline. NAV-240, formerly known as IMB101, is a clinical-stage bispecific antibody targeting OX40L and TNFα, both of which play critical roles in the pathogenesis of various inflammatory diseases. Targeting both the OX40L and TNFα signaling pathways could potentially be more efficacious than single-agent therapies, making it a potential choice for treating complex and heterogeneous diseases with unmet medical needs. IMBiologics has indicated that this therapy has the potential to become a "best-in-class" medication for treating multiple autoimmune diseases. The therapy is currently being assessed in a Phase I clinical trial in healthy volunteers and is expected to commence a Phase Ib clinical trial in patients with rheumatoid arthritis later this year. Per the licensing agreement, Navigator will obtain global rights to develop NAV-240 outside of Asia. IMBiologics will receive an upfront payment of $20 million and is eligible for up to $925 million in development and commercial milestone payments. Related to this transaction, Navigator has completed its $100 million Series A funding round co-led by RA Capital Management and Forbion. The funds raised will be used to support the development of NAV-240, as well as to advance a series of monoclonal or bispecific antibodies targeting OX40L.
3.Salubris Pharmaceuticals Secures Exclusive License for Yoltech Therapeutics’ Base Editing Drug YOLT-101
On August 26, Salubris Pharmaceuticals announced its plans to sign an agreement with Yoltech Therapeutics to acquire exclusive licensing rights for the API and formulation related intellectual property and technical information of Yoltech’s investigational PCSK9-targeted base editing drug "YOLT-101" within Mainland China. This includes rights for research and development, registration, production, and commercial sales among others. The agreement stipulates an upfront payment and research and development milestone payments totaling no more than RMB 205 million, with cumulative sales milestones that could reach up to RMB 830 million. YOLT-101, a PCSK9 base editing drug independently developed by Yoltech Therapeutics, is currently intended for conditions such as familial hypercholesterolemia (FH). YOLT-101 is in the pre-clinical research stage, and the first administration to a subject has been completed in an Investigator Initiated Trial (IIT). Pre-clinical data has shown that in a nonhuman primate model, YOLT-101 achieved a significant reduction in LDL-C (low-density lipoprotein cholesterol) lasting nearly two years with just one administration.
4.Bayer and NextRNA Reach Nearly $550 Million Collaboration
On August 29th, Bayer and NextRNA Therapeutics announced that they have entered into a collaboration and licensing agreement valued at nearly $550 million. Together, they will develop two potential 'first-in-class' small molecule therapies targeting long non-coding RNAs (lncRNAs), which will enhance Bayer's precision oncology research and development pipeline. Bayer and NextRNA will collaborate on advancing two therapies aimed at tumor indications with significant unmet needs. The first project involves a small molecule therapy targeting lncRNA, currently in the early preclinical development stage. For the second project, NextRNA will continue to investigate lncRNA targets identified by its platform, with Bayer having the option to co-develop one of these targets.
According to the terms of the agreement, NextRNA will receive up to $547 million from the collaboration across the two projects, including upfront payments, near-term milestones, research funding, and other payments related to development and commercial milestones. NextRNA is a biotechnology company focused on developing transformative drugs for lncRNA-driven oncological and neurological diseases. Their approach primarily involves disrupting the interaction between lncRNA and RNA-binding proteins (RBPs) to inhibit the function of disease-related lncRNAs. NextRNA's proprietary platform combines its computational engine, NextMap, with extensive expertise in lncRNA biology, along with diverse biochemical, biophysical, and chemical functionalities.
5.Pfizer and Flagship Expand Collaboration
On August 29th, Pioneering announced a further acceleration in its strategic collaboration with Pfizer. Quotient Therapeutics, a company established by Flagship, is set to work with Pfizer on two research projects. The partners will analyze somatic mutations occurring in patient tissues to guide the discovery and development of potentially transformative therapies for cardiovascular and kidney diseases. Pioneering Medicines, a drug discovery and development division under Flagship, is leading the strategic collaboration with Pfizer. This includes propelling the exploration process and swiftly identifying potential drug development projects based on Flagship's diverse biological platforms and models. The agreement with Quotient marks the latest initiative following Flagship's announcement of a strategic partnership with Pfizer in July 2023.
Quotient Therapeutics specializes in systemic research into the genetic variations and evolution of trillions of cells within the human body. The company's technology platform performs high-precision and high-resolution genetic sequencing on tissues obtained from patient lesions, revealing the relationships between somatic genetic mutations and diseases. This drives the discovery of transformative drugs aimed at curing, preventing, or reversing diseases. Quotient was founded by Flagship Pioneering in 2022 and is supported by experts in the field of somatic genetics.
6.Allist Secures Rights to Jiakesi's KRAS G12C Inhibitor and SHP2 Inhibitor in China
On August 30, Allist signed a strategic collaboration agreement with Jiakesi Pharmaceuticals, granting Allist exclusive rights in China (including Mainland China, Hong Kong, Macau, and Taiwan) for the research, development, manufacturing, registration, and commercialization of the KRAS G12C inhibitor Glecirasib and the SHP2 inhibitor JAB-3312. Jiakesi retains all rights to Glecirasib and JAB-3312 outside of this region and may continue the development of these drugs. According to the terms of the agreement, Allist will pay Jiakesi an upfront payment of 150 million RMB, development and sales milestone payments of up to 700 million RMB, and double-digit percentage royalties on sales, with the stated amounts including VAT. The New Drug Application (NDA) for Glecirasib, intended for second-line treatment of KRAS G12C-mutated non-small cell lung cancer, received priority review status on May 21, 2024. JAB-3312, a selective allosteric inhibitor of SHP2, a key node in the RAS signaling pathway, is the only SHP2 inhibitor globally to have entered Phase III clinical trials and has been granted orphan drug designation by the U.S. FDA for esophageal cancer, including esophageal squamous cell carcinoma.