Is Daxxify approved by the FDA?
Daxxify, with the generic name daxibotulinumtoxinA-lanm, is an FDA-approved medication used for both cosmetic and therapeutic purposes. It belongs to the drug class of skeletal muscle relaxants and is administered as a lyophilized powder for injection.
Indications and Usage: Daxxify works as a longer-lasting botox injection due to its unique stabilizer (cell-penetrating peptide). It acts by inhibiting acetylcholine release and blocking neuromuscular transmission, thereby relaxing muscles. This mechanism helps reduce frown lines and alleviate symptoms of cervical dystonia, such as spasms and stiffness.
Dosage and Administration:
- For Glabellar Lines: Typically administered as 0.1 mL (8 Units) per injection site into five specific points, totaling 40 Units.
- For Cervical Dystonia: Dosage ranges from 125 Units to 250 Units, divided among affected muscles.
Side Effects: Common side effects of Daxxify include headaches, eyelid drooping, and temporary facial muscle weakness. Serious side effects may include heart problems, eye issues, allergic reactions, and potential spread of toxin effects to other body areas.
Warnings: Daxxify carries warnings about potential severe side effects like breathing difficulties, swallowing problems, and spread of toxin effects, which could affect distant muscles and cause symptoms of botulism.
Precautions: Before receiving Daxxify, patients should inform their healthcare providers about existing medical conditions, especially muscle or nerve disorders, prior botulinum toxin treatments, and current medications.
Conclusion: In summary, Daxxify is FDA approved for treating both glabellar lines and cervical dystonia in adults. Its unique formulation offers a longer duration of action compared to earlier botulinum toxins, making it a viable option for cosmetic enhancement and therapeutic relief.
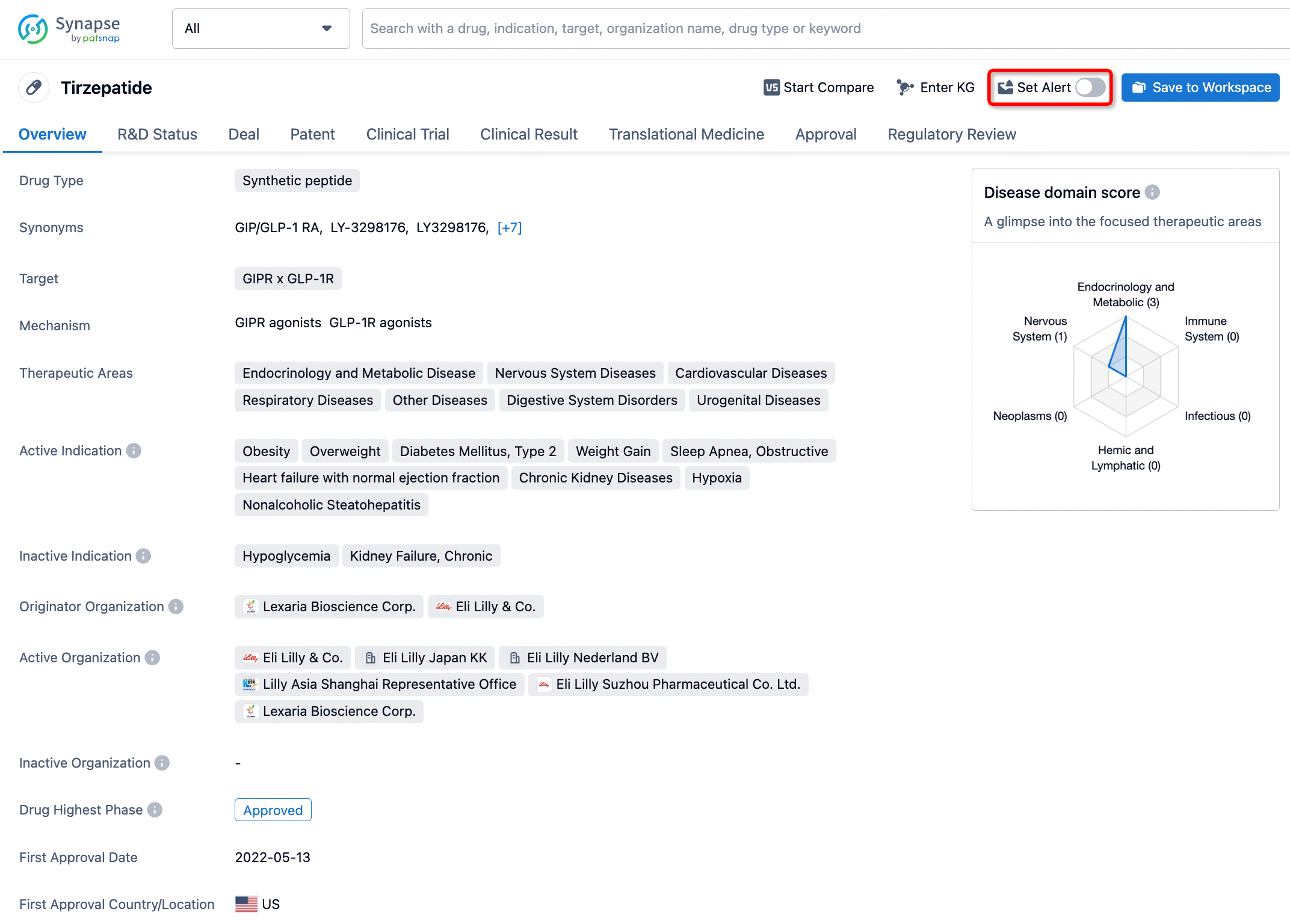
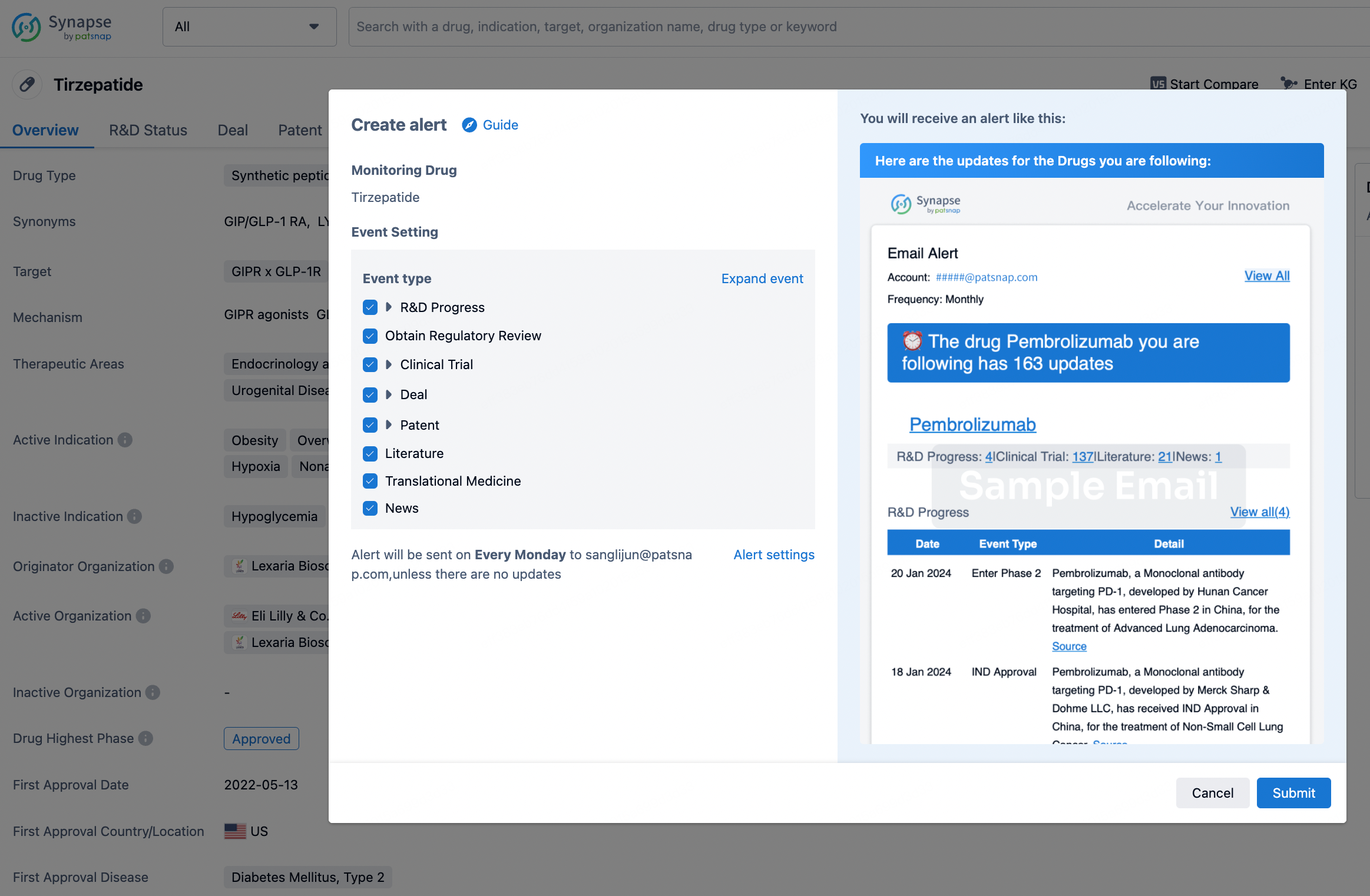
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!