Last update 21 Jun 2024
Etrolizumab
Last update 21 Jun 2024
Overview
Basic Info
Drug Type Monoclonal antibody |
Synonyms Etrolizumab (USAN/INN), Monoclonal antibody beta7, recombinant-human-monoclonal-antibody-beta7 + [9] |
Target |
Mechanism α4β7 antagonists(Integrin alpha-4/beta-7 antagonists), αEβ7 antagonists(Integrin alpha-E/beta-7 antagonists) |
Therapeutic Areas |
Active Indication- |
Inactive Indication |
Originator Organization |
Active Organization- |
Inactive Organization |
Drug Highest PhaseDiscontinuedPhase 3 |
First Approval Date- |
RegulationOrphan Drug (US) |
Gene Sequence
Sequence Code 9975717H

Source: *****
Sequence Code 13306635L

Source: *****
External Link
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| D09901 | Etrolizumab | - |
R&D Status
10 top R&D records. to view more data
Login
| Indication | Highest Phase | Country/Location | Organization | Date |
|---|---|---|---|---|
| Crohn Disease | Phase 3 | US | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | AR | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | AU | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | AT | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | BE | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | BR | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | BG | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | CA | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | HR | 20 Mar 2015 | |
| Crohn Disease | Phase 3 | CZ | 20 Mar 2015 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
Phase 3 | Crohn Disease Maintenance | 1,035 | qimkoiichn(zwksapaxyx) = dsqzbvwgtb dtgayqkswr (apqzkacthz ) View more | Positive | 11 Oct 2022 | ||
Phase 1 | 24 | gjmapviexj(bzjclevoiz) = lldendohgk hkwkrnqkng (nbrldzkpkr ) | Positive | 01 Sep 2022 | |||
Phase 3 | 397 | Placebo (IV)+Etrolizumab (Etrolizumab + Placebo (IV)) | nalgpfvkvc(zvfmdtctee) = mmcfcmupue cvxvojmgmy (uzbwbxmzpq, spoakuestp - nhvyqwwzcg) View more | - | 13 Aug 2021 | ||
Placebo (Injection) (Infliximab + Placebo (Injection)) | nalgpfvkvc(zvfmdtctee) = azrsgjnscw cvxvojmgmy (uzbwbxmzpq, jdvsgddjtr - ecugiiaayb) View more | ||||||
Phase 3 | 359 | (Double-Blind Maintenance Phase: Etrolizumab) | rgfzyzqbfx(qrraxgtbms) = edmqxbldmo ujlnkpbihv (gkfdnanufk, wgplwycivw - dyceporhxs) View more | - | 15 Jun 2021 | ||
Placebo (Double-Blind Maintenance Phase: Placebo) | rgfzyzqbfx(qrraxgtbms) = izvlgxlvqq ujlnkpbihv (gkfdnanufk, dafuuwxooz - lqxcpehaqp) View more | ||||||
Phase 3 | 609 | Placebo (Cohort 2: Placebo (Double-Blind Induction Phase)) | nqfmcukfpx(couswpwpnq) = immltbvawy bxccnqlxap (vtpqrqywwg, rezddmpvxa - kuulmkreyi) View more | - | 15 Jun 2021 | ||
(Cohort 2: Etrolizumab (Double-Blind Induction Phase)) | nqfmcukfpx(couswpwpnq) = cjpzxyoory bxccnqlxap (vtpqrqywwg, zradteucfg - zpetldvzwb) View more | ||||||
Phase 1 | - | 30 | lcdchbetnu(ccyzdvexps) = 97% did not experience any pain greater than mild , and 50% did not experience any pain at all uzocvehjbz (njiywossij ) View more | Positive | 01 May 2021 | ||
Phase 3 | 358 | Etrolizumab Placebo (Placebo) | kqzgtoqwmc(akasvbgqzb) = zsfpbspclz qucwfwuitn (olcgletylo, frcvpynndc - jvowkftdki) View more | - | 05 Apr 2021 | ||
Adalimumab Placebo+Etrolizumab (Etrolizumab) | kqzgtoqwmc(akasvbgqzb) = xsfouybojz qucwfwuitn (olcgletylo, hgqlomvguz - brodsmumwr) View more | ||||||
Phase 3 | 358 | Etrolizumab Placebo (Placebo) | fypwjipaht(nqobraatrw) = owbynhgmwx penexycxea (xaitceoodc, txqexnxxmx - qimiwjtuxr) View more | - | 05 Mar 2021 | ||
Adalimumab Placebo+Etrolizumab (Etrolizumab) | fypwjipaht(nqobraatrw) = wursxvjblt penexycxea (xaitceoodc, hfttwnqinu - amwzdmeusz) View more | ||||||
Phase 1 | 24 | (Etrolizumab Q4W) | zgjbvigpux(ythynxsbga) = rtfwgqbail xfvjiczxpz (qrnvauxupt, egaglhkmvu - zvhofkdyrj) View more | - | 05 Aug 2020 | ||
(Etrolizumab Q8W) | zgjbvigpux(ythynxsbga) = ahblygzwlm xfvjiczxpz (qrnvauxupt, btiabgsiqo - ricgquwjnq) View more | ||||||
Phase 3 | 130 | nxsmkwchze(rjszlefwqy) = qpbwglbozc gnhnjphfxa (xtonxyouwp ) View more | Positive | 01 Nov 2017 | |||
Placebo | - |
Login to view more data
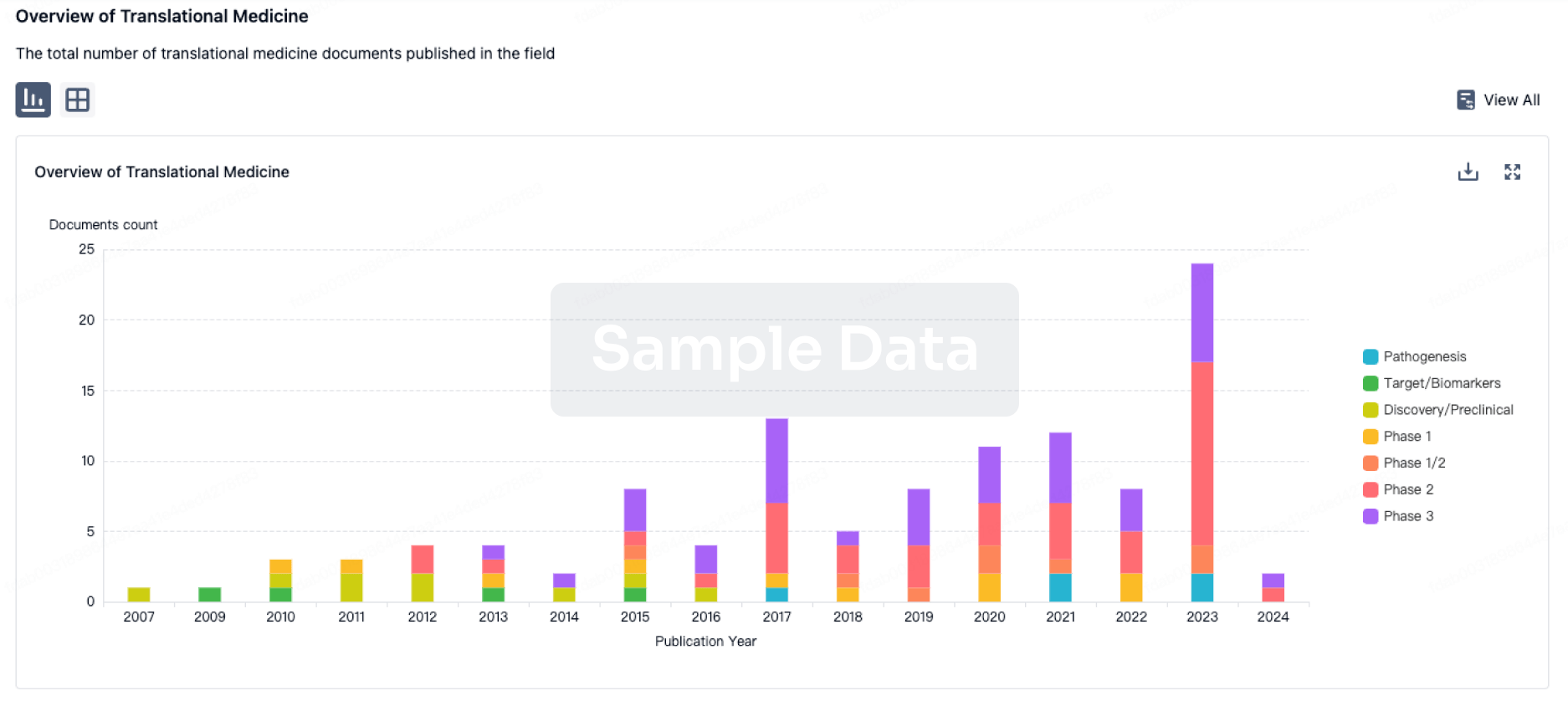
Translational Medicine
Boost your research with our translational medicine data.
login
or
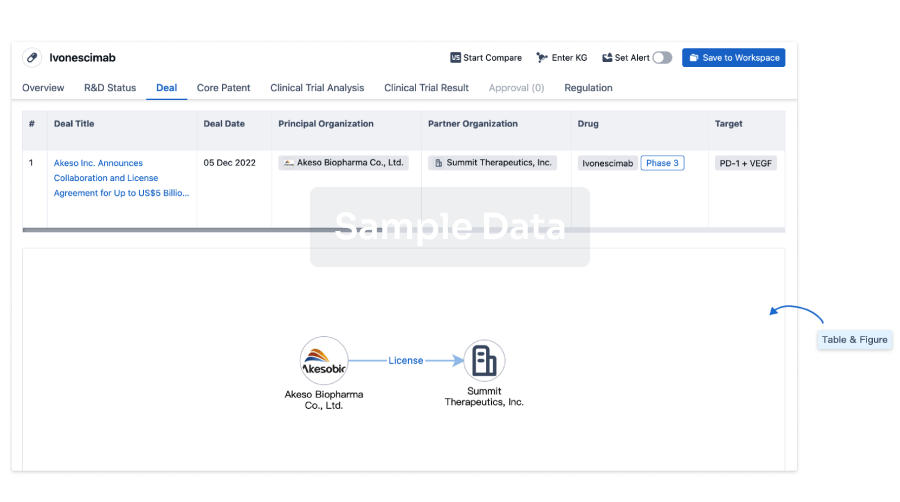
Deal
Boost your decision using our deal data.
login
or
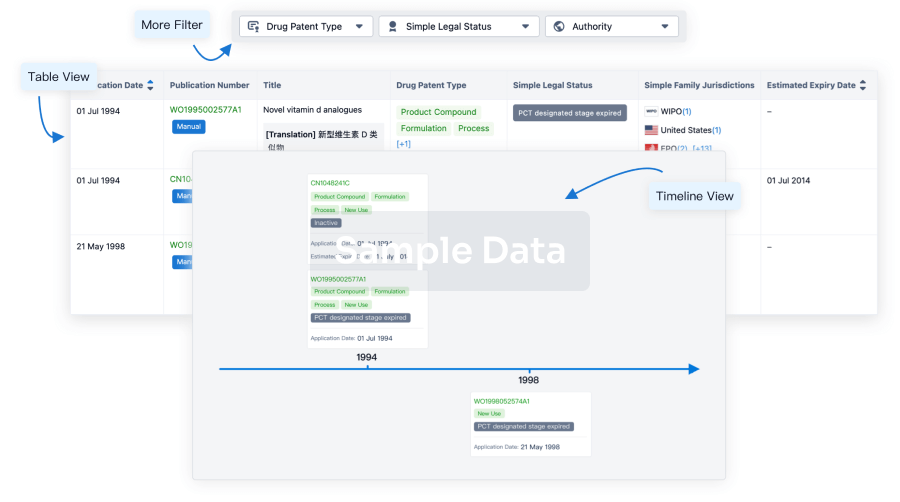
Core Patent
Boost your research with our Core Patent data.
login
or
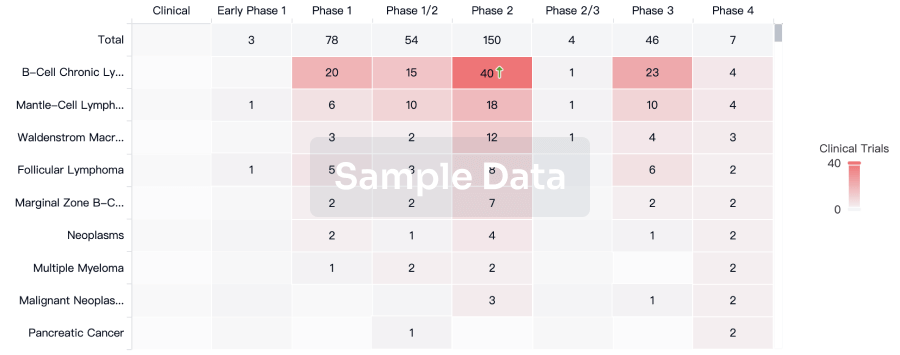
Clinical Trial
Identify the latest clinical trials across global registries.
login
or
Approval
Accelerate your research with the latest regulatory approval information.
login
or
Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Leverages most recent intelligence information, enabling fullest potential.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free
