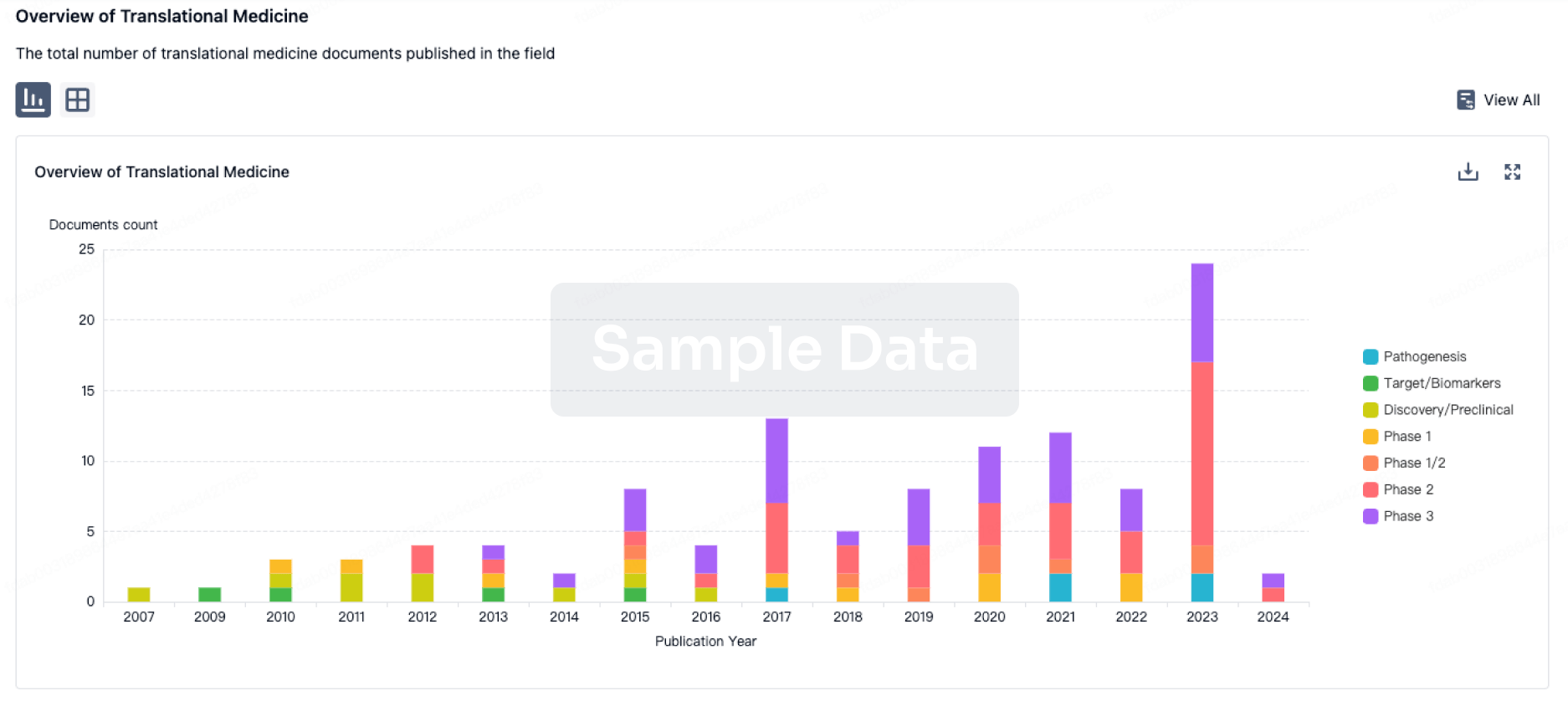
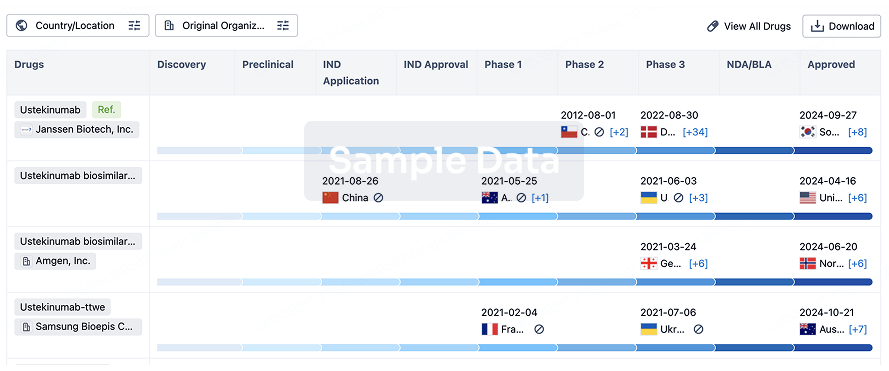
Fewer participants treated with SNG001 compared to placebo progressed to hospitalisation (1/110 vs 7/110, P=0.07)
SNG001 was well-tolerated with a favourable safety pro with previous studies
High level of adherence to SNG001 treatment at-home was observed, with 90% of patients completing all doses of the once-daily regimen
SOUTHAMPTON, England--(BUSINESS WIRE)-- Synairgen plc (LSE: SNG), the respiratory company developing SNG001, an investigational formulation for inhalation containing the broad-spectrum antiviral protein interferon beta to treat or prevent severe viral lung infections, today announces topline data for participants treated with SNG001 in the US National Institutes of Health (NIH)/ AIDS Clinical Trials Group (ACTG)-led ACTIV-2 Phase 2 adaptive platform trial for COVID-19.
Between February and August 2021, 221 symptomatic, non-hospitalised adult participants with COVID-19 were enrolled in the Phase 2 SNG001 evaluation and randomised between active drug (N=110 treated) and placebo (N=110 treated1), across 25 US sites.
There were no statistically significant differences between SNG001 and placebo with respect to the primary outcomes of safety (SNG001 was well-tolerated), symptom resolution, or virology.
Fewer participants required hospitalisation following treatment with SNG001 (N=1/110, [1%]) compared to placebo (N=7/110, [6%]), an 86% relative risk reduction, that was not statistically significant (P=0.07; two-sided Fisher’s exact test). There were no deaths in either arm.
There were 13 participants with treatment-emergent Grade 3 or higher Adverse Events (AE) with four on SNG001 [3.6%] and nine on placebo [8.2%].
Based on the study results, the Independent Data Safety Monitoring Board for ACTIV-2 recommended SNG001 advance from Phase 2 into Phase 3 in the ACTIV-2 trial in October 2021. Later, in March 2022, the NIH decided to halt all participant recruitment in the trial due to the significant shift in the nature of the pandemic which would necessitate a substantial modification of the study design which was not feasible in a multiple treatment-arm platform trial of this size.
Richard Marsden, CEO of Synairgen, commented: “The data from the ACTIV-2 trial in the US showing a non-significant but encouraging 86% relative risk reduction in hospitalisation following treatment with SNG001 in home-based patients are very interesting when considered in the context of therapeutic options for the management of non-hospitalised patients during a pandemic.
“These results are really encouraging and build upon the strong body of non-clinical and clinical evidence, which includes Synairgen’s home-based trial (SG016) in high-risk patients,2 supporting SNG001’s potential as a broad-spectrum antiviral for a wide range of severe lung infections including COVID-19. We will now begin to evaluate the full data set and factor these findings into our development plans.”
William Fischer, M.D., University of North Carolina (UNC), a lead investigator of SNG001 said: “These results, including a decrease in hospitalisations and adverse events among participants who received SNG001, are promising and warrant further investigation in a larger Phase 3 clinical trial. SNG001 is the first nebulized inhaled therapeutic to demonstrate promising results and, if confirmed in larger studies, could represent a potential treatment option for people who have COVID-19 but are not hospitalised.”
Participants self-administered SNG001 or placebo via a nebuliser at home to complete 14 daily doses, with the first dose taken either at the study site or at home, with instructions provided by study staff and through an instructional video available online on how to take the study medication. There was a high level of adherence to the once-daily treatment in patients taking SNG001, with 90% of patients receiving full treatment. The ability to self-administer is important in the context of a pandemic when the ability of infected patients to travel to a clinic may be very challenging.
The primary goals of the trial were to evaluate safety, to see if the investigational therapeutic could reduce the duration of symptoms through study day 28, and to see if the investigational therapeutic could increase the proportion of patients with undetectable virus in nasopharyngeal swabs at specific time points.
Synairgen continues to actively pursue and refine multiple clinical approaches including company- and investigator-sponsored studies and possibly platform studies (as and when appropriate), to confirm the most expeditious clinical development path for SNG001 in patients hospitalised due to a severe viral lung infection including SARS-CoV-2, influenza, RSV, adenovirus, parainfluenza and rhinoviruses.
This announcement reports on the reduction in hospitalisations, a key endpoint for determining efficacy of antivirals for COVID-19. Full results, including other key endpoints, will be presented and/or published by the NIH in due course.
This announcement contains inside information for the purposes of Article 7 of Regulation (EU) 596/2014.
Notes for Editors
About Synairgen
Synairgen is a UK-based respiratory company focused on drug discovery, development and commercialisation. The Company’s primary focus is developing SNG001 (inhaled interferon beta) for the treatment of severe viral lung infections, including COVID-19, as potentially the first host-targeted, broad-spectrum antiviral treatment delivered directly into the lungs. SNG001 has been granted Fast Track status from the US Food and Drug Administration (FDA). Founded by University of Southampton Professors Sir Stephen Holgate, Donna Davies and Ratko Djukanovic in 2003, Synairgen is quoted on AIM (LSE: SNG). For more information about Synairgen, please see .
About SNG001
SNG001 is a pH-neutral formulation of interferon-beta (IFN-beta) for inhalation that is delivered directly into the lungs using a mesh nebuliser, which the Company is developing as a potential host-directed antiviral treatment for patients hospitalised with severe viral lung infections, including COVID-19. SNG001 has broad potential applicability for patients hospitalised with respiratory symptoms due to viral infections such as SARS-CoV-2, influenza, RSV, adenovirus, parainfluenza and rhinoviruses.
Viruses have been shown to suppress the production of IFN-beta, a naturally occurring protein that orchestrates the body's antiviral defences, with the aim of evading host immune responses. By administering IFN-beta into the lungs, the aim is to correct this deficiency, potentially switching back on the lungs' antiviral pathways to clear the virus. SNG001 has been shown to demonstrate potent in vitro antiviral activity against a broad range of viruses at concentrations that are achievable following inhaled delivery of SNG001 including RSV, rhinovirus, various influenza strains including H5N1, MERS-CoV and SARS-CoV-2 including Alpha, Beta, Gamma, Delta and Omicron variants of concern.
About ACTIV-2
Accelerating COVID-19 Therapeutic Interventions and Vaccines (ACTIV) was a public-private partnership to develop a coordinated research strategy to accelerate the development of the most promising COVID-19 treatments and vaccines (Protocol ACTIV-2/A5401; "Adaptive Platform Treatment Trial for Outpatients with COVID-19 (Adapt out COVID)"). ACTIV-2 is an adaptive Phase 2/3 trial testing agents in non-hospitalised adults experiencing mild to moderate COVID-19 sponsored by NIAID, part of the National Institutes of Health, and is led by the NIAID-funded AIDS Clinical Trials Group (ACTG).
1 One participant randomized to placebo did not receive study intervention and was excluded from all analyses. The treated population therefore included 220 participants (110 SNG001, 110 placebo).
2 Synairgen conducted a trial of SNG001 in high-risk, home-based COVID-19 patients (SG016) in which fewer patients treated with SNG001 were hospitalised (0/56) compared to placebo (2/58). In total across SG016 and ACTIV-2, 1/165 SNG001-treated home-based COVID-19 patients have been hospitalised.