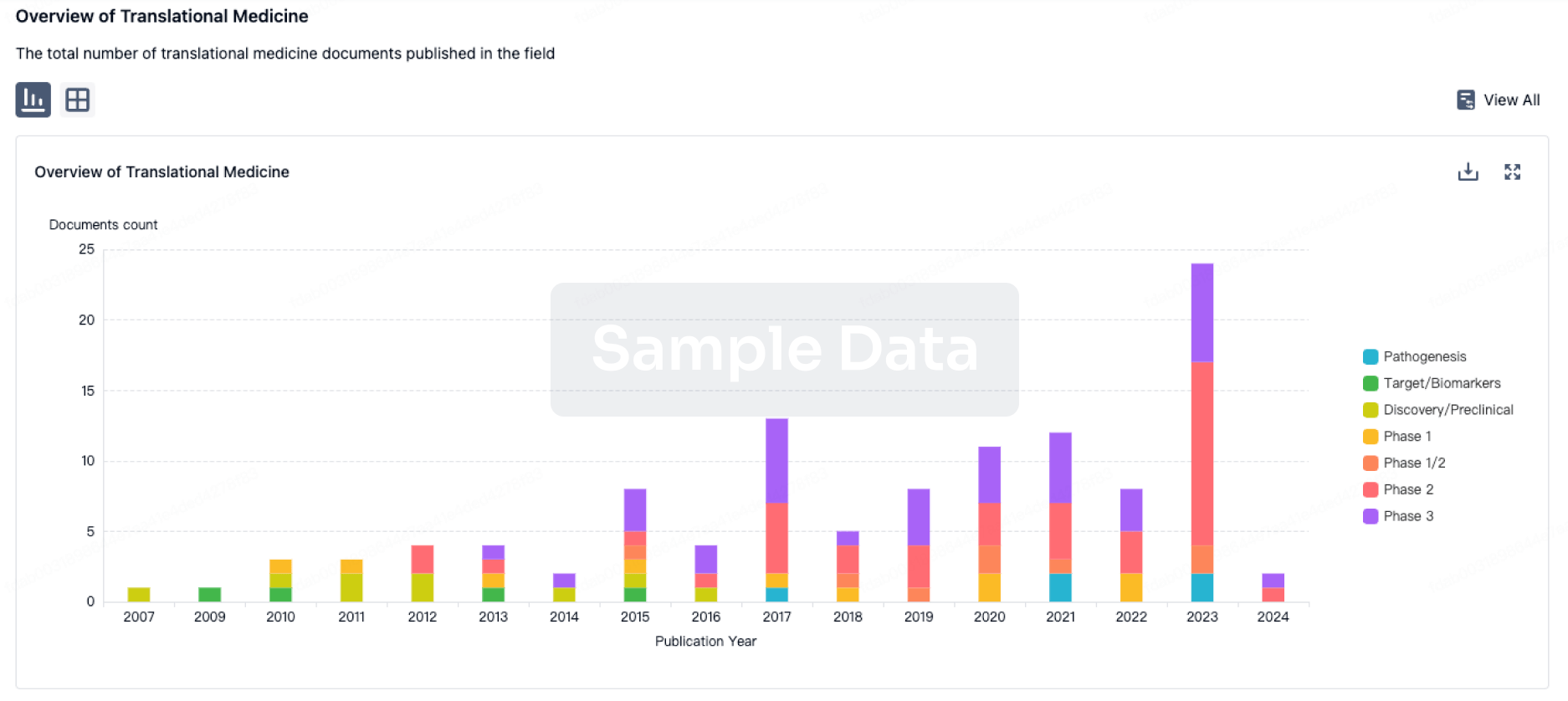
A team of neuroscientists and research integrity experts assembled a 300-page dossier supporting their claims of misconduct in 132 papers published by Eliezer Masliah, M.D. Separately, the NIH flagged concerns with two papers.
The NIH has found evidence of research misconduct against Eliezer Masliah, M.D., the longtime head of the National Institute on Aging's (NIA) neuroscience division, the NIH said in a Sept. 26 statement. The misconduct findings—made after a 9-month investigation by the agency—involve figures being reused and relabeled between two papers despite them representing different experimental conditions, according to the NIH.“NIH will notify the two journals of its findings so that appropriate action can be taken,” the agency said in the statement. Masliah is not currently serving as the NIA’s neuroscience director, the agency added. Deputy director Amy Kelley, M.D., is serving as the acting director of the NIA’s neuroscience division. Masliah took up the NIA neuroscience division post in the summer of 2016, according to his biography on the NIH website.NIH didn't return Fierce Biotech's request for comment, but said in the release that beyond the information already provided, they don't comment on personnel matters. It's unclear whether Masliah is still employed by the agency. NIH first began reviewing misconduct allegations against Masliah in May 2023, the agency said, beginning an investigation phase in Dec. 2023 that just concluded on Sept. 15. The NIH announcement coincides with the Sept. 26 publication of an investigation by Science finding that 132 papers published by Masliah contain apparently manipulated images. Some of the challenged research includes work on drug candidates that are currently in clinical trials or are already in use, including treatments for Parkinson’s and Alzheimer’s disease, according to Science's publication.For the Science investigation, a team of neuroscientists and research integrity experts assembled a 300-page dossier supporting their claims of misconduct in papers published by Masliah. Eleven other neuroscientists then reviewed the dossier, Science says, and all agreed that the findings warranted investigations by the NIH, academic journals, funders and the University of California, San Diego, where Masliah previously ran a research lab.“These fields, both Alzheimer's and Parkinson's, need a reset,” Matthew Schrag, M.D., Ph.D., a neuroscientist at Vanderbilt University Medical Center who contributed to the dossier, told Fierce Biotech in an interview. “We need to look at this with an understanding that research integrity is a significant problem, and we need to come in and make sure that we are chasing the right targets and that we are universally committed to the well-being of our patients.” Schrag’s investigative work is independent of his job at Vanderbilt University Medical Center.One of the drugs implicated in the Science investigation is prasinezumab, an antibody treatment for Parkinson’s disease. Prasinezumab targets alpha-synuclein, a protein that aggregates pathologically in Parkinson’s. The drug is under development by Prothena Biosciences in collaboration with Roche; Prothena was co-founded by the late Dale Schenk, Ph.D., a neuroscientist who co-authored four foundational papers on alpha-synuclein with Masliah that are implicated in the dossier, according to Science.In phase 2 trial data reported in Aug. 2022, prasinezumab showed no difference to placebo on measures of Parkinson’s disease progression. Another phase 2 trial is ongoing.“Let me not put too fine a point on this: the ongoing phase 2 trial should be shut down,” Schrag said.Another implicated med is dementia treatment cerebrolysin from Austria-based Ever Pharma, which is not approved in the United States but is available in dozens of other countries, according to Science. In addition, another alpha-synuclein drug for Parkinson’s called minzasolmin, from a company Masliah co-founded named Neuropore Therapies, is also held up by suspect research, according to Science’s findings.Prothena, Ever Pharma and Neuropore all told Science that the allegations are not related to the current clinical programs of the respective drugs. Antibodies that target alpha-synuclein are based on basic science by Masliah that's now being called into question, Schrag said. “I think we're seeing a number of attempts on this particular target through this mechanism that appear to be not performing as well as we'd like,” he said. Evidence that the antibodies can reach alpha-synuclein clumped inside of neurons “came from these animal studies that appear to have been heavily manipulated," the neuroscientist said.Numerous biotechs have been rocked recently by misconduct claims. Flagship Pioneering’s Laronde had its research on a new class of genetic medicine scrutinized in a Stat and Boston Globe investigation, revealing a “bad assay” and poor note-taking. Flagship then merged Laronde with another of its companies, Senda Biosciences, to form Sail Biomedicines.Lykos Therapeutics, a company researching MDMA-assisted therapies, has also been besieged by controversy lately. After facing a rejection by the FDA, the psychedelic firm had three papers retracted from scientific journals due to misconduct. The tumult continued from there, with Lykos laying off 75% of its staff and eventually saying goodbye to CEO Amy Emerson.And in the Alzheimer’s space, Cassava Biosciences has long been dogged by accusations of faking data. Schrag played a pivotal role in investigating publications underlying Cassava’s science, identifying dozens with problematic images. The company’s CEO and senior vice president of neuroscience both departed in July.On Sept. 12, the United States Department of Health and Human Services finalized new guidelines for research misconduct investigations conducted by recipients of Public Health Services funding. The new rules clarify the requirements of misconduct investigations, give institutions 90 days to investigate claims instead of 60 and outline an appeals process and potential administrative remedies to misconduct, according to a release.“We have to acknowledge the seriousness of the problem, and we need to get serious about addressing known, obvious cases of research misconduct in a way where there's meaningful consequences,” Schrag said. “We need to focus on the next generation of scientists and ingraining the importance of doing this right.”Editor's note: This article was updated to provide further context on Masliah's current position at NIH.