/ CompletedNot Applicable 瑞巴派特片(100mg)在中国健康受试者中空腹及餐后给药条件下随机、开放、单剂量、两序列、两周期、双交叉生物等效性试验
[Translation] A randomized, open-label, single-dose, two-sequence, two-period, double-crossover bioequivalence study of rebamipide tablets (100 mg) in Chinese healthy subjects under fasting and fed conditions
主要研究目的:
按有关生物等效性试验的规定,选择大塚制薬株式会社 (OTSUKA PHARMACEUTICAL Co.,Ltd.)生产的瑞巴派特片(商品名: Mucosta®,规格:100mg/片)为参比制剂,对浙江金华康恩贝生物制药有限公司生产、杭州默银医药技术有限公司提供的受试制剂瑞巴派特片(规格:100mg/片)进行空腹及餐后给药人体生物等效性试验,比较受试制剂中药物的吸收速度和吸收程度与参比制剂的差异是否在可接受的范围内,比较两种制剂在空腹及餐后给药条件下的生物等效性。
次要研究目的:
观察健康志愿受试者口服受试制剂瑞巴派特片(规格: 100mg/片)和参比制剂瑞巴派特片(商品名:Mucosta®,规格:100mg/片)的安全性。
[Translation] Main research purposes:
According to the regulations on bioequivalence testing, rebamipide tablets (trade name: Mucosta®, specifications: 100 mg/tablet) produced by OTSUKA PHARMACEUTICAL Co., Ltd. were selected as the reference preparation. , conduct human bioequivalence tests on fasting and postprandial administration of the test preparation rebamipide tablets (specification: 100mg/tablet) produced by Zhejiang Jinhua Conba Biopharmaceutical Co., Ltd. and provided by Hangzhou Moyin Pharmaceutical Technology Co., Ltd. , compare whether the difference in absorption speed and degree of drug in the test preparation and the reference preparation is within the acceptable range, and compare the bioequivalence of the two preparations under fasting and postprandial administration conditions.
Secondary research purpose:
Observe the safety of the test preparation rebamipide tablets (specification: 100mg/tablet) and the reference preparation rebamipide tablets (trade name: Mucosta®, specification: 100mg/tablet) taken orally by healthy volunteer subjects.
/ CompletedNot Applicable 瑞巴派特片(100mg)在中国健康受试者中空腹及餐后给药条件下随机、开放、单剂量、两序列、两周期、双交叉生物等效性试验
[Translation] A randomized, open-label, single-dose, two-sequence, two-period, double-crossover bioequivalence study of rebamipide tablets (100 mg) in Chinese healthy subjects under fasting and fed conditions
主要研究目的:按有关生物等效性试验的规定,选择大塚制薬株式会社(OTSUKA PHARMACEUTICAL Co.,Ltd.)生产的瑞巴派特片(商品名:Mucosta,规格:100mg/片)为参比制剂,对浙江金华康恩贝生物制药有限公司生产、杭州默银医药技术有限公司提供的受试制剂瑞巴派特片(规格:100mg/片)进行空腹及餐后给药人体生物等效性试验,比较受试制剂中药物的吸收速度和吸收程度与参比制剂的差异是否在可接受的范围内,比较两种制剂在空腹和餐后给药条件下的生物等效性。
次要研究目的:观察健康志愿受试者口服受试制剂瑞巴派特片(规格:100mg/片)和参比制剂瑞巴派特片(商品名:Mucosta,规格:100mg/片)的安全性
[Translation] Main research purpose: According to the regulations on bioequivalence testing, rebamipide tablets (trade name: Mucosta, specification: 100mg/tablet) produced by OTSUKA PHARMACEUTICAL Co., Ltd. are selected as As a reference preparation, the test preparation rebamipide tablets (specification: 100mg/tablet) produced by Zhejiang Jinhua Conba Biopharmaceutical Co., Ltd. and provided by Hangzhou Moyin Pharmaceutical Technology Co., Ltd. were administered to human organisms on an empty stomach and after meals. Efficacy test, compare whether the difference in absorption speed and extent of drug in the test preparation and the reference preparation is within the acceptable range, and compare the bioequivalence of the two preparations under fasting and postprandial administration conditions.
Secondary research purpose: To observe the safety of oral administration of test preparation rebamipide tablets (specification: 100mg/tablet) and reference preparation rebamipide tablets (trade name: Mucosta, specification: 100mg/tablet) to healthy volunteers sex
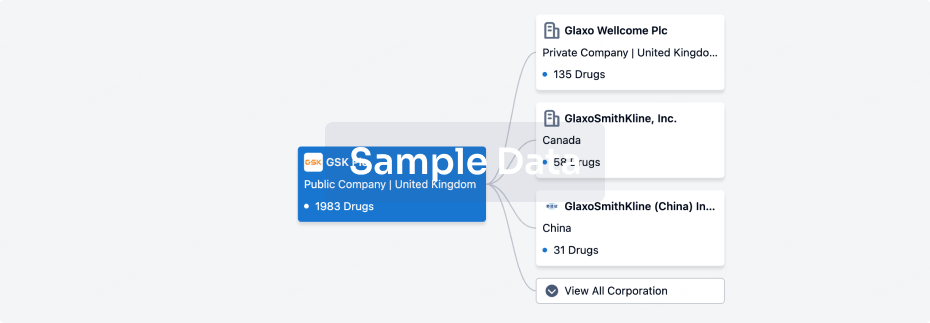
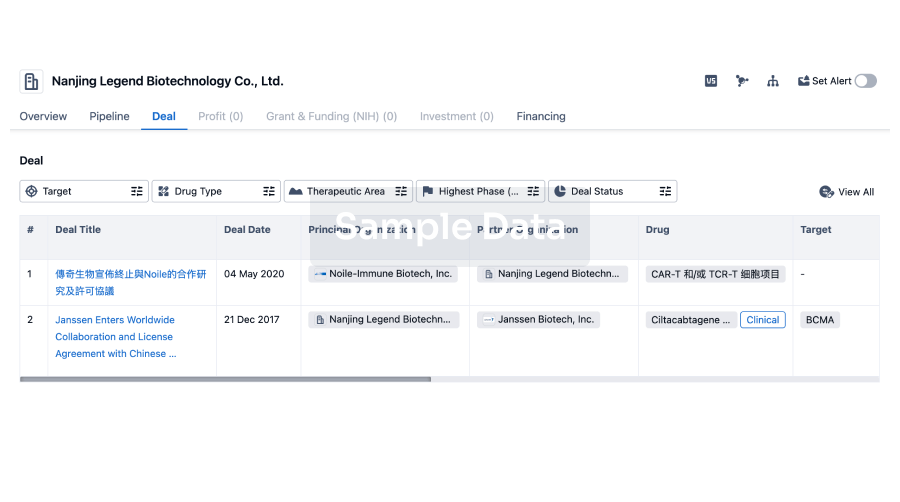
100 Clinical Results associated with Hangzhou Moyin Pharmaceutical Technology Co., Ltd.
0 Patents (Medical) associated with Hangzhou Moyin Pharmaceutical Technology Co., Ltd.
100 Deals associated with Hangzhou Moyin Pharmaceutical Technology Co., Ltd.
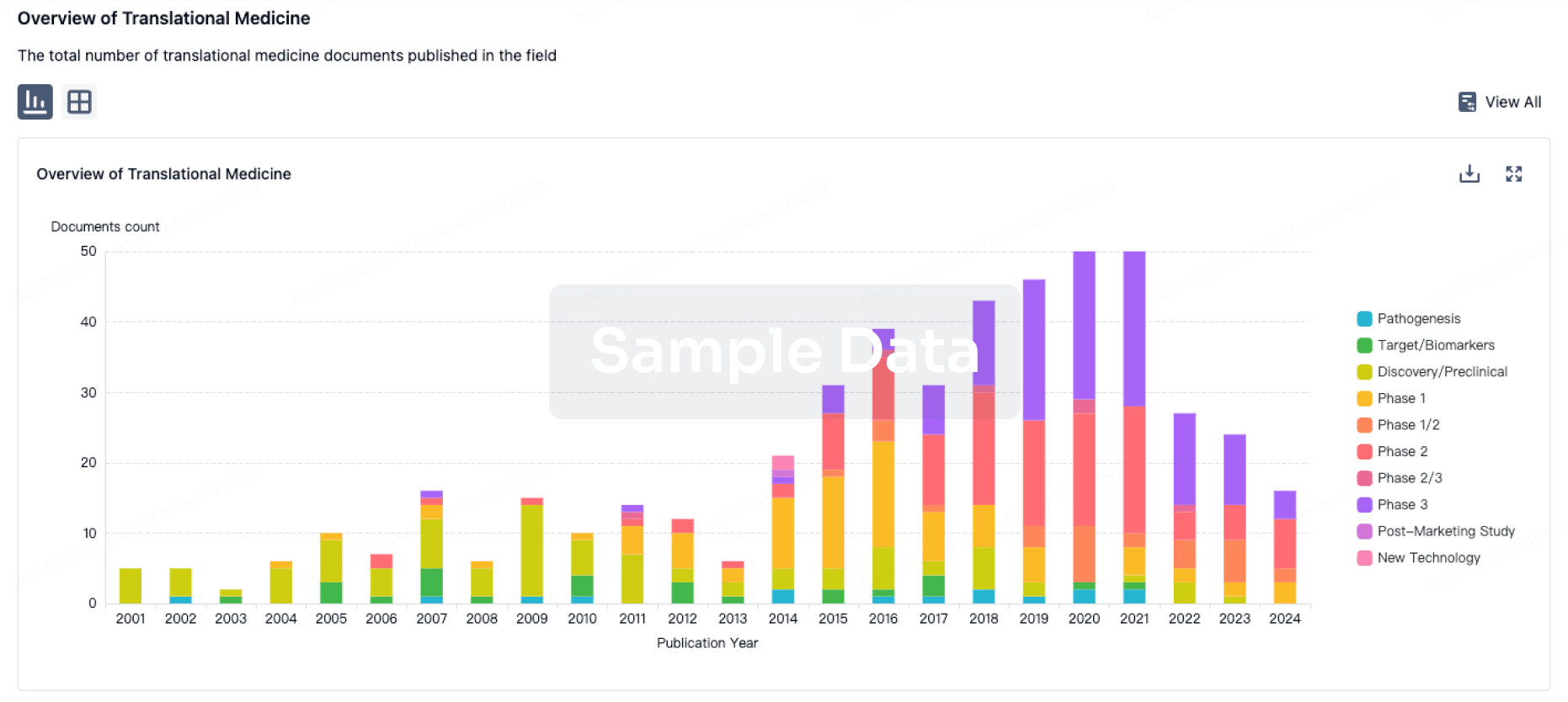
100 Translational Medicine associated with Hangzhou Moyin Pharmaceutical Technology Co., Ltd.