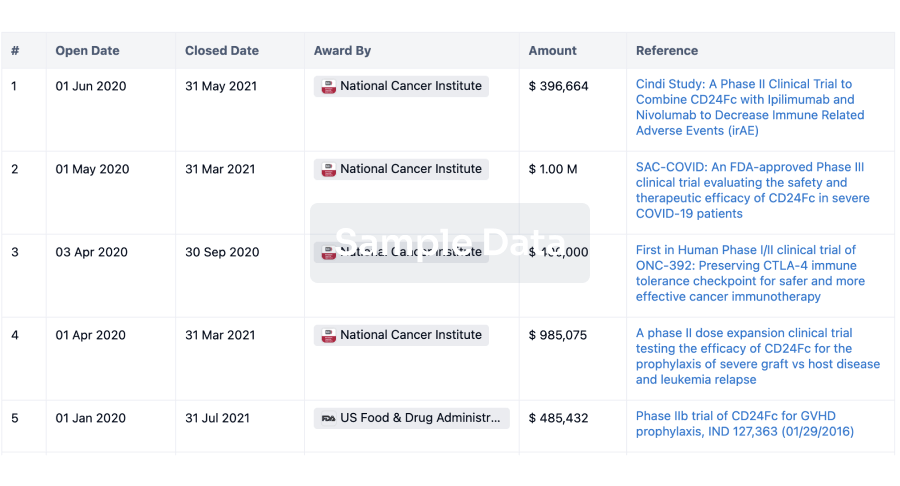
Under the deal, The Salzman Group provides, among other services, clinical study management services in relation to the Phase 2 study evaluating the use of cannabidiol in the prevention of graft versus host disease.
SAN FRANCISCO and TEL AVIV, Israel, Jan. 26, 2018 (GLOBE NEWSWIRE) --
Kalytera Therapeutics, Inc.
(TSX-V:
KALY
) (OTCQB:KALTF) (the “
Company
” or “
Kalytera
”) today announced that the Company has elected to issue 396,015 common shares of the Company (“
Common Shares
”) to The Salzman Group in payment of an invoice issued under the services agreement with The Salzman Group previously announced on December 7, 2017 (the “
Agreement
”). Under the Agreement, The Salzman Group provides, among other services, clinical study management services in relation to the Phase 2 study evaluating the use of cannabidiol in the prevention of graft versus host disease.
The invoice owing to The Salzman Group is in the amount of US$112,798 (or C$139,000.98 based on the daily average exchange rate for January 25, 2018 published by the Bank of Canada). The number of Common Shares to be issued is based on a deemed issue price of C$0.351 per Common Share, being 90% of the closing price of the Common Shares on the TSXV on January 25, 2018, the trading day prior to the Company’s election to pay the debt in Common Shares. The Common Shares are expected to be issued to The Salzman Group on or about January 31, 2018.
About Kalytera Therapeutics
Kalytera Therapeutics, Inc. (“
Kalytera
”) is pioneering the development of a next generation of cannabinoid therapeutics. Through its proven leadership, drug development expertise, and intellectual property portfolio, Kalytera seeks to establish a leading position in the development of novel cannabinoid medicines for a range of important unmet medical needs, with an initial focus on graft versus host disease (“
GVHD
”).
Kalytera also intends to develop a new class of proprietary cannabidiol (“
CBD
”) therapeutics. CBD is a versatile compound that has shown activity against a number of pharmacological targets. However, there are limitations associated with natural CBD, including its poor oral bioavailability. Kalytera will seek to develop innovative CBD formulations and prodrugs in an effort to overcome these limitations, and to target specific disease sites within the body. Kalytera intends to of matter and method of use patents covering its novel inventions, with the goal of limiting future competition.
Website Home:
News and Insights:
Investors:
Cautionary Statements
Neither TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
This press release may contain certain forward-looking information and statements (“forward-looking information”) within the meaning of applicable Canadian securities legislation, that are not based on historical fact, including without limitation in respect of its product candidate pipeline, planned clinical trials, regulatory approval prospects, intellectual property objectives and other statements containing the words “believes”, “anticipates”, “plans”, “intends”, “will”, “should”, “expects”, “continue”, “estimate”, “forecasts” and other similar expressions. Readers are cautioned to not place undue reliance on forward-looking information. Actual results and developments may differ materially from those contemplated by these statements depending on, among other things, the ris
k
that future clinical studies may not proceed as expected or may produce unfavourable results.
Kalytera undertakes no obligation to comment on analyses, expectations or statements made by third-parties, its securities, or financial or operating results (as applicable). Although Kalytera believes that the expectations reflected in forward-looking information in this press release are reasonable, such forward-looking information has been based on expectations, factors and assumptions concerning future events which may prove to be inaccurate and are subject to numerous risks and uncertainties, certain of which are beyond Kalytera’s control. The forward-looking information contained in this press release are expressly qualified by this cautionary statement and are made as of the date hereof. Kalytera disclaims any intention and has no obligation or responsibility, except as required by law, to update or revise any forward-looking information, whether as a result of new information, future events or otherwise.
Contact Information
Robert Farrell
President, CEO
(888) 861-2008
info@kalytera.co