|
Mechanism5-HT1A receptor agonists [+3] |
|
|
|
|
Drug Highest PhaseApproved |
First Approval Ctry. / Loc.US |
First Approval Date15 Nov 2002 |
注射用醋酸奥曲肽微球在中国健康成年受试者空腹状态下的随机、开放、单次给药、平行对照的生物等效性研究
[Translation] A randomized, open-label, single-dose, parallel-controlled bioequivalence study of octreotide acetate microspheres for injection in Chinese healthy adult subjects under fasting conditions
主要目的:研究受试制剂注射用醋酸奥曲肽微球和参比制剂善龙(注射用醋酸奥曲肽微球)在健康受试者体内单次给药后的药代动力学,评价受试制剂和参比制剂的生物等效性。
次要目的:比较受试制剂注射用醋酸奥曲肽微球和参比制剂善龙(注射用醋酸奥曲肽微球)在健康受试者中单次给药后的药效动力学和安全性。
[Translation] Main purpose: To study the pharmacokinetics of the test preparation Octreotide Acetate Microspheres for Injection and the reference preparation Centron (Octreotide Acetate Microspheres for Injection) after a single administration in healthy subjects, and to evaluate the test preparations and reference preparations. Bioequivalence of preparations.
Secondary purpose: To compare the pharmacodynamics and safety of the test formulation Octreotide Acetate Microspheres for Injection and the reference formulation Centron (Octreotide Acetate Microspheres for Injection) after a single administration in healthy subjects.
注射用阿立哌唑微球与ABILIFY MAINTENA®在中国精神分裂症患者中多剂量、不同给药间隔、多次给药药代动力学对比临床研究
[Translation] A comparative clinical study on the pharmacokinetics of aripiprazole microspheres for injection and ABILIFY MAINTENA® in Chinese schizophrenia patients with multiple doses, different dosing intervals and multiple doses
1.评价注射用阿立哌唑微球 350 mg/4周多次给药达稳态时的PK参数(阿立哌唑)AUC0-28d,ss是否能与ABILIFY MAINTENA® 400 mg/4周达到生物等效;
2.评价注射用阿立哌唑微球 350 mg/4周多次给药达稳态后给药间隔内(0-28天)阿立哌唑血药浓度的Cmin,ss 和 Cmax,ss实测值在 94~534 ng/mL 目标浓度区间内(即在口服片剂阿立哌唑 10 mg 稳态 Cmin 的 50th 至 30 mg 稳态 Cmax 的 75th 内)的受试者达标率。
[Translation] 1. To evaluate whether the PK parameter (aripiprazole) AUC0-28d,ss of aripiprazole microspheres for injection 350 mg/4 weeks at steady state can be compared with that of ABILIFY MAINTENA® 400 mg/4 weeks. bioequivalent;
2. To evaluate the Cmin,ss and Cmax,ss of aripiprazole plasma concentration within the dosing interval (0-28 days) after multiple administration of aripiprazole microspheres for injection 350 mg/4 weeks to a steady state The rate of compliance in subjects with values within the target concentration range of 94 to 534 ng/mL (ie, within the 50th of the oral tablet aripiprazole 10 mg steady-state Cmin to within 75th of the 30 mg steady-state Cmax).
一项评估注射用阿立哌唑微球在中国精神分裂症患者中单次给药的药代 动力学特征及安全性、耐受性的多中心、开放性临床研究
[Translation] A multicenter, open-label clinical study evaluating the pharmacokinetic characteristics, safety and tolerability of single-dose aripiprazole microspheres for injection in Chinese patients with schizophrenia
主要目的
1.评价注射用阿立哌唑微球在中国精神分裂症患者中不同剂量、单次给药的药代动力学特征。
次要目的
1.评价注射用阿立哌唑微球在中国精神分裂症患者中不同剂量、单次给药的安全性和耐受性;
2.评价注射用阿立哌唑微球单次给药在中国精神分裂症患者中的初步疗效;
3.评价注射用阿立哌唑微球单次给药与阿立哌唑长效肌内注射剂(ABILIFY MAINTEN,单次给药)在中国精神分裂症患者中的药代动力学特征差异。
探索性目的
1.评价注射用阿立哌唑微球单次给药在不同CYP2D6酶代谢型受试者中的药代动力学特征差异(若CYP2D6酶代谢型存在差异则对主要终点进行亚组分析, 否则不进行分析)。
[Translation] main purpose
1. To evaluate the pharmacokinetics of aripiprazole microspheres for injection in Chinese patients with schizophrenia at different doses and single administration.
secondary purpose
1. To evaluate the safety and tolerability of aripiprazole microspheres for injection in Chinese patients with schizophrenia at different doses and single administration;
2. To evaluate the preliminary efficacy of single administration of aripiprazole microspheres for injection in Chinese patients with schizophrenia;
3. To evaluate the difference in pharmacokinetics between single-dose aripiprazole microspheres for injection and aripiprazole long-acting intramuscular injection (ABILIFY MAINTEN, single-dose) in Chinese patients with schizophrenia.
exploratory purpose
1. To evaluate the differences in the pharmacokinetic characteristics of a single dose of aripiprazole microspheres for injection in subjects with different CYP2D6 enzyme metabolites (if there are differences in CYP2D6 enzyme metabolites, subgroup analysis of the primary endpoint will be performed, otherwise not analyzed).
100 Clinical Results associated with Zhuhai Livzon Weiqiu Technology Co., Ltd.
0 Patents (Medical) associated with Zhuhai Livzon Weiqiu Technology Co., Ltd.
100 Deals associated with Zhuhai Livzon Weiqiu Technology Co., Ltd.
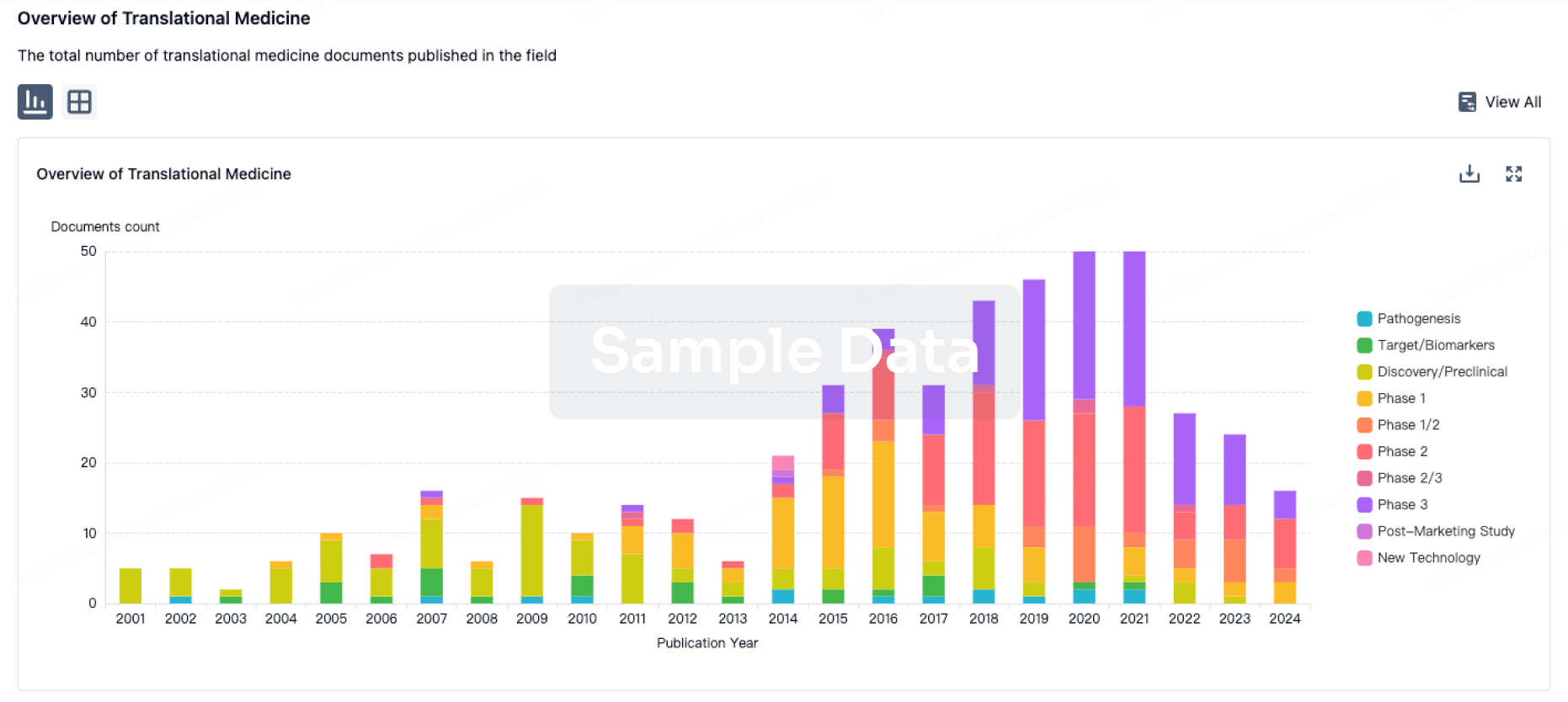
100 Translational Medicine associated with Zhuhai Livzon Weiqiu Technology Co., Ltd.