/ RecruitingNot Applicable DAISY Uterine Drain: Device Evaluation With Standard Wall Suction
The goal of this study is to obtain user feedback while placing and observing the DAISY uterine drain with wall suction. This study defines the obstetrical surgeons as "users" and the patients in whom the drain is placed as "participants." Participants are pregnant women who are undergoing cesarean delivery (CD), who have not entered active labor, who have consented to drain placement and who have met all the inclusion/exclusion criteria. Users are staff or fellow obstetrical surgeons who will use the drain and provide the evaluation.
/ RecruitingEarly Phase 1 Trans-Abdominal Fetal Pulse Oximetry - An Early Feasibility Study
The Lumerah System, developed and manufactured by Raydiant Oximetry, Inc., is a non-invasive fetal pulse oximeter that measures fetal arterial oxygen saturation using safe, non-invasive, transabdominal near-infrared spectroscopy. The Lumerah System is intended as an adjunct to cardiotocography. In this study, women in labor will also be simultaneously monitored with a re-engineered version of the previously approved transvaginal oximeter sensor connected to a Nellcor N-400 fetal oximetry monitor for the purposes of device development. The data obtained from the transabdominal sensor and the transvaginal sensor will be used for research purposes only and will not be used to guide or alter patient management.
/ TerminatedNot Applicable Trans-abdominal Fetal Pulse Oximetry: PILOT 1
The Raydiant Oximetry Sensing System (Lumerah) is a non-invasive fetal pulse oximeter that measures fetal arterial oxygen saturation using safe, non-invasive, transabdominal near-infrared spectroscopy. Lumerah is intended as an adjunct to cardiotocography by detecting decreases in fetal oxygenation.
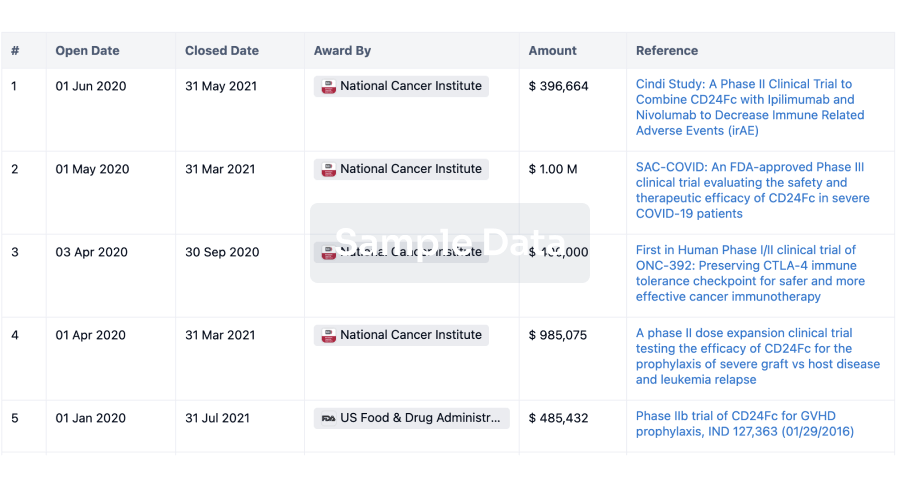
100 Clinical Results associated with Raydiant Oximetry, Inc.
0 Patents (Medical) associated with Raydiant Oximetry, Inc.
01 Sep 2024·Biomedical Optics Express
Sensitivity analysis of transabdominal fetal pulse oximetry using MRI-based simulations
Article
Author: Ruesch, Alexander ; Kainerstorfer, Jana M ; Debreczeny, Martin P ; Komolibus, Katarzyna ; Kainerstorfer, Jana M. ; Satish, Gopika ; Wu, Jingyi ; Jayet, Baptiste ; Andersson-Engels, Stefan
Transabdominal fetal pulse oximetry offers a promising approach to improve fetal monitoring and reduce unnecessary interventions. Utilizing realistic 3D geometries derived from MRI scans of pregnant women, we conducted photon simulations to determine optimal source-detector configurations for detecting fetal heart rate and oxygenation. Our findings demonstrate the theoretical feasibility of measuring fetal signals at depths up to 30 mm using source-detector (SD) distances greater than 100 mm and wavelengths between 730 and 850 nm. Furthermore, we highlight the importance of customizing SD configurations based on fetal position and maternal anatomy. These insights pave the way for enhanced non-invasive fetal monitoring in clinical application.
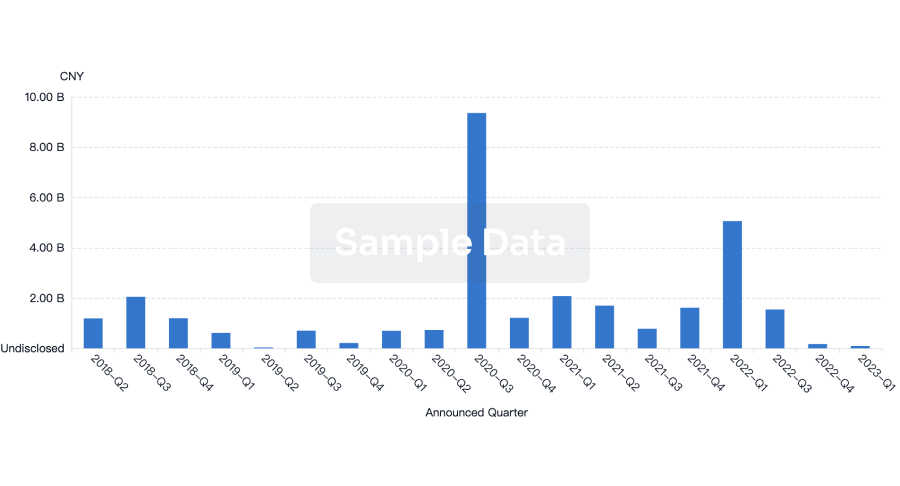
100 Deals associated with Raydiant Oximetry, Inc.
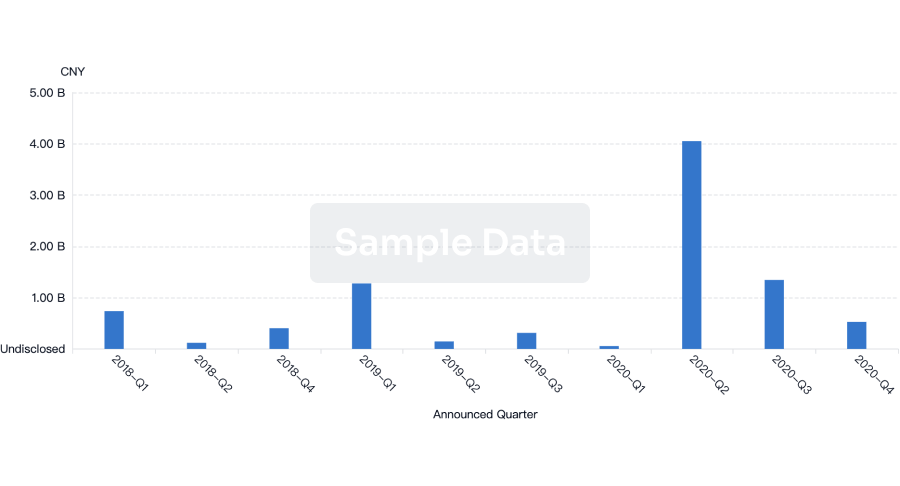
100 Translational Medicine associated with Raydiant Oximetry, Inc.