Analysis on the Clinical Research Progress of Nitric Oxide Synthase
Nitric oxide (NO) is a signaling molecule and a free radical, involved in many important physiological processes in mammals, and it plays a key role in inflammation and tumor diseases. Endogenous NO is produced by the oxidation of L-arginine catalyzed by nitric oxide synthase (NOSs).
Nitric oxide synthase (NOS) is an essential enzyme in the human body that plays a crucial role in various physiological processes. NOS is responsible for the production of nitric oxide (NO), a signaling molecule involved in regulating blood flow, immune response, and neurotransmission. NO acts as a vasodilator, relaxing blood vessels and improving blood circulation. It also plays a role in immune defense by enhancing the activity of white blood cells. Additionally, NO acts as a neurotransmitter, facilitating communication between nerve cells. The proper functioning of NOS and the production of NO are vital for maintaining cardiovascular health, immune function, and neuronal signaling.
Nitric Oxide Synthase Competitive Landscape
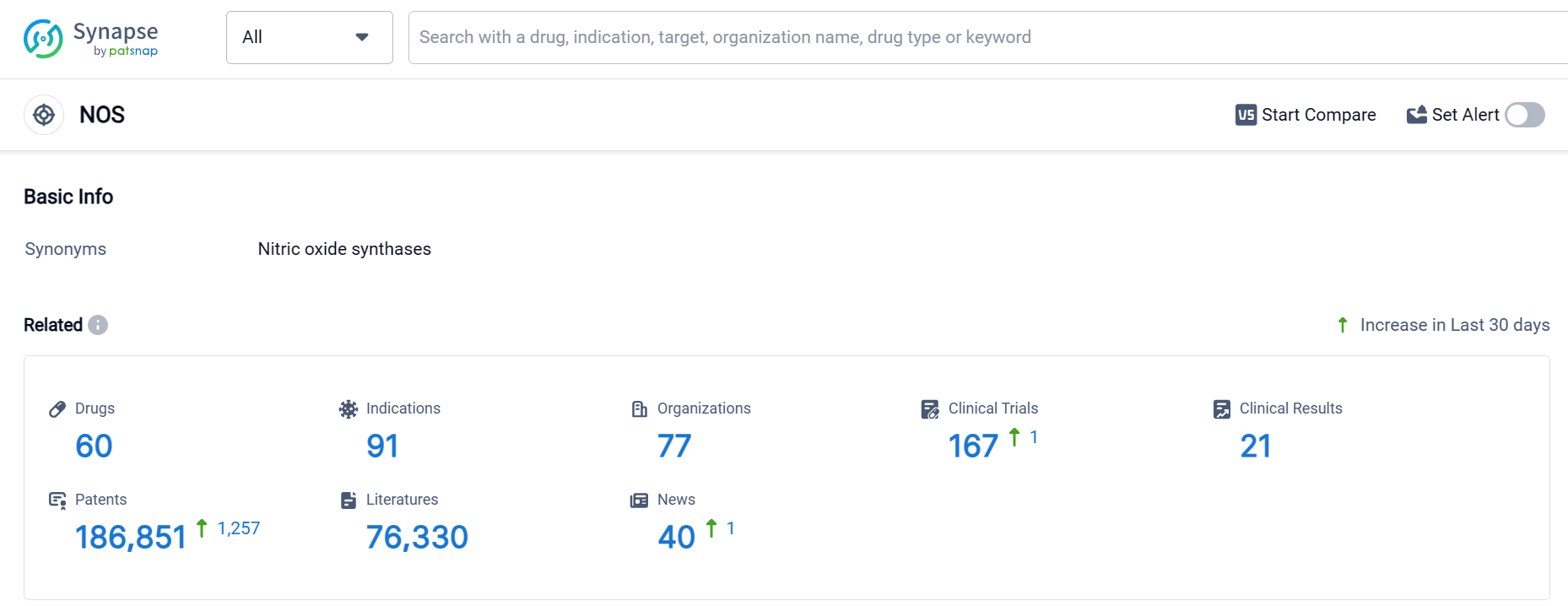
According to Patsnap Synapse, as of 6 Oct 2023, there are a total of 60 NOS drugs worldwide, from 77 organizations, covering 91 indications, and conducting 167 clinical trials.
👇Please click on the picture link below for free registration or login directly if you have freemium accounts, you can browse the latest research progress on drugs , indications, organizations, clinical trials, clinical results, and drug patents related to this target.
Based on the analysis of the data provided, the current competitive landscape of the target NOS in the pharmaceutical industry is characterized by the presence of multiple companies with drugs in different phases of development. Ipsen SA, vasopharm GmbH, Mallinckrodt Plc, and PTC Therapeutics, Inc. are some of the companies with drugs in advanced stages of development. The approved indications for drugs under the target NOS include Migraine Disorders, Hypotension, Alzheimer Disease, Nervous System Diseases, Cluster Headache, and others.
The drug types progressing most rapidly under the target NOS include Small molecule drugs, Stem cell therapy, Gene therapy, Synthetic peptide, and Biological products. The presence of biosimilars indicates intense competition around innovative drugs.
Countries/locations such as China, European Union, United States, and Canada are actively involved in the development of drugs under the target NOS.
Overall, the target NOS presents opportunities for further research and development in the pharmaceutical industry. The competitive landscape is dynamic, with multiple companies and countries/locations contributing to the progress in this area. Further analysis and research are recommended to identify potential collaborations and partnerships for future development.
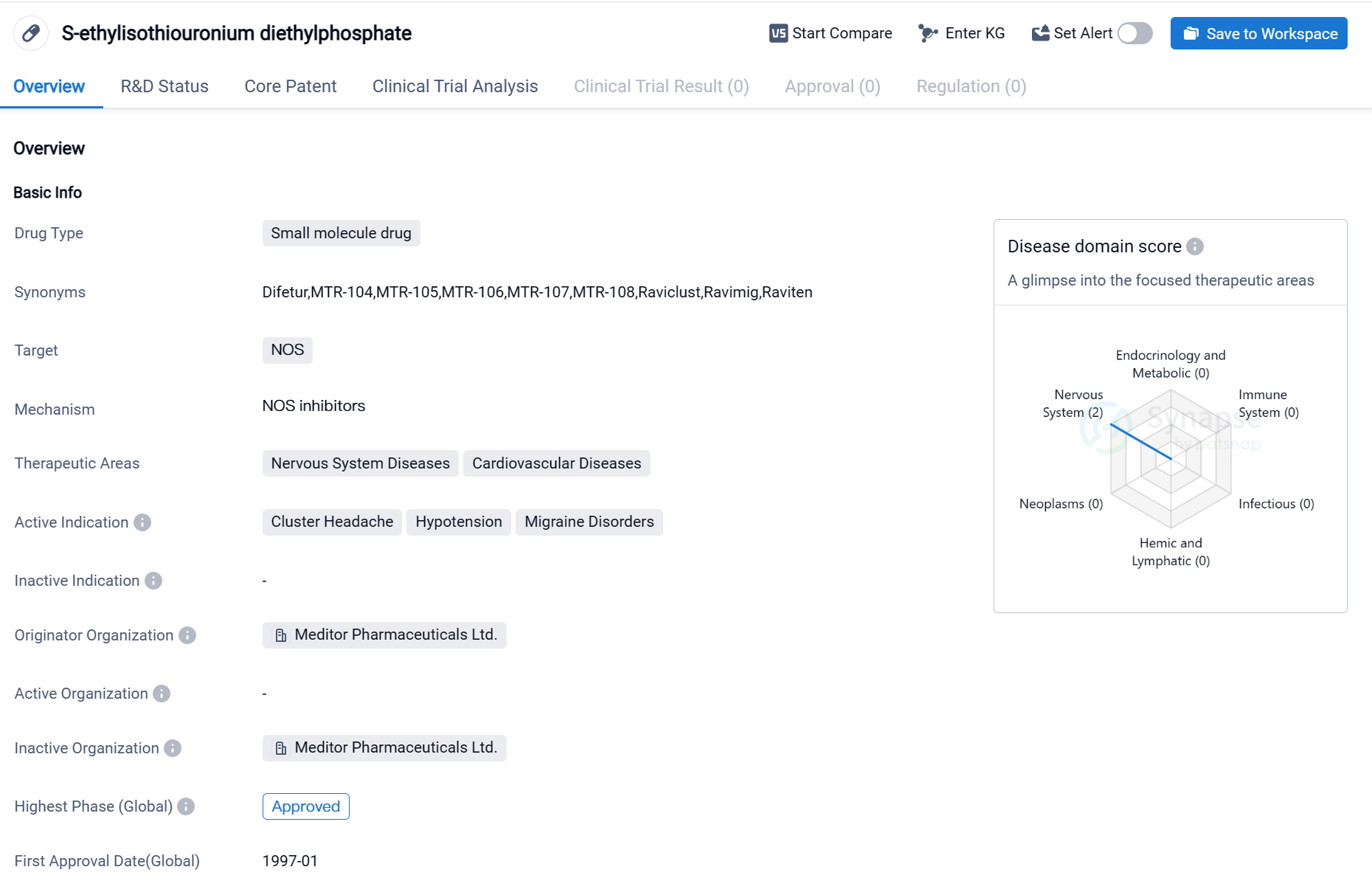
Key drug:S-ethylisothiouronium diethylphosphate
The drug S-ethylisothiouronium diethylphosphate is a small molecule drug that targets NOS (nitric oxide synthase). It is primarily used in the treatment of nervous system diseases and cardiovascular diseases. The active indications for this drug include cluster headache, hypotension, and migraine disorders.
The drug was developed by Meditor Pharmaceuticals Ltd., an originator organization in the pharmaceutical industry. It has reached the highest phase of development, which is approved.
The first approval date for this drug on a global scale was in January 1997. This indicates that it has been available for use in the market for over two decades. The drug has likely undergone extensive testing and evaluation to ensure its safety and efficacy before receiving approval.
👇Please click on the image below to directly access the latest data (R&D Status | Core Patent | Clinical Trial | Approval status in Global countries) of this drug.
S-ethylisothiouronium diethylphosphate is particularly relevant in the treatment of nervous system diseases and cardiovascular diseases. Cluster headache, a severe form of headache that occurs in clusters or cycles, can be debilitating for patients. Hypotension, or low blood pressure, can lead to symptoms such as dizziness and fainting. Migraine disorders are characterized by recurrent headaches, often accompanied by other symptoms such as nausea and sensitivity to light and sound.
The drug's mechanism of action, targeting NOS, suggests that it may work by regulating the production of nitric oxide, a signaling molecule involved in various physiological processes. By modulating NOS activity, the drug may help alleviate the symptoms associated with the aforementioned conditions.
Overall, S-ethylisothiouronium diethylphosphate is an approved small molecule drug developed by Meditor Pharmaceuticals Ltd. It targets NOS and is used in the treatment of nervous system diseases and cardiovascular diseases, specifically cluster headache, hypotension, and migraine disorders. Its first approval date on a global scale was in January 1997, indicating its long-standing presence in the market.