Exploring Sintilimab's R&D successes and its clinical results at the 2023 ESMO_ASIA
The combination of ICIs and anti-angiogenic therapy has a synergistic anti-tumor effect and can improve the prognosis of advanced NSCLC patients. On 2 Dec 2023, the latest clinical trial about Sintilimab in combination with anlotinib in 2023 ESMO_ASIA further demonstrated the therapeutic benefits.
Sintilimab's R&D Progress
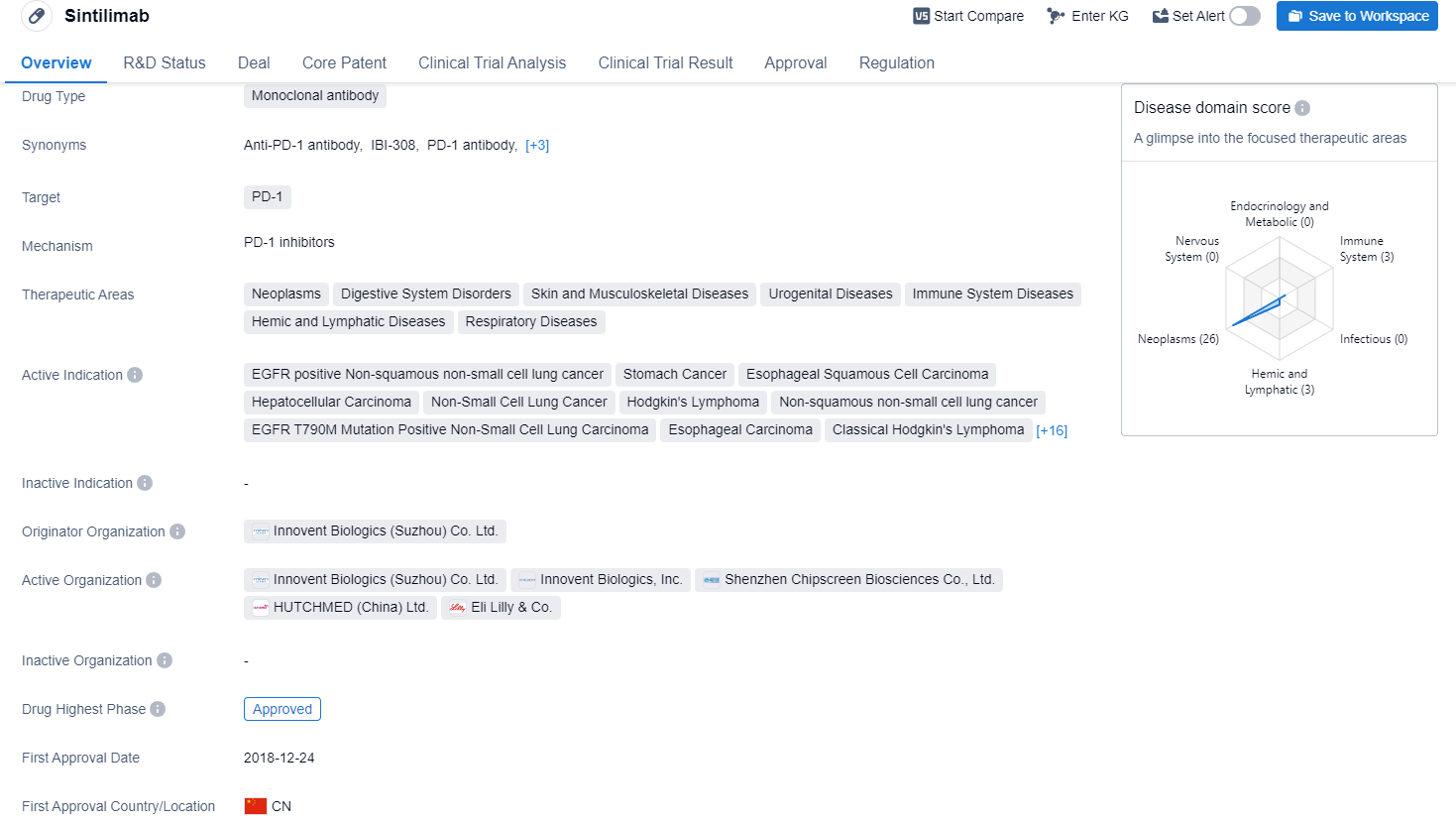
Sintilimab is a monoclonal antibody drug that targets PD-1, a protein involved in regulating the immune system. The drug has shown promise in treating a wide range of therapeutic areas, including neoplasms, digestive system disorders, skin and musculoskeletal diseases, urogenital diseases, immune system diseases, hemic and lymphatic diseases, and respiratory diseases.
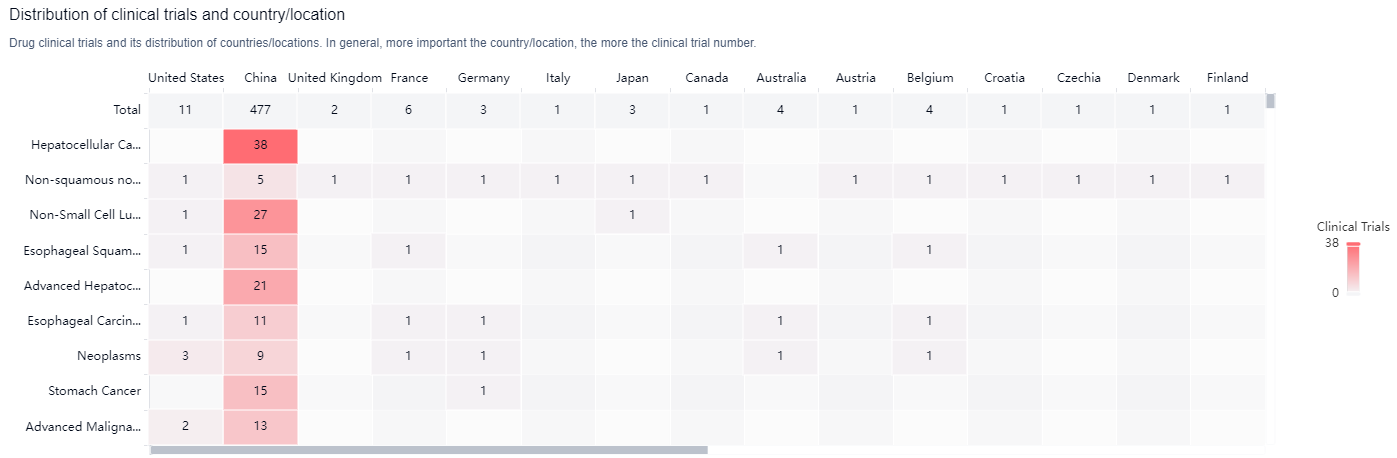
According to the Patsnap Synapse, Sintilimab has been developed by Innovent Biologics (Suzhou) Co. Ltd. and has received approvals globally. And the clinical trial distributions for Sintilimab are primarily in the United States, China and United Kingdom. The key indication is Hepatocellular Carcinoma.
Detailed Clinical Result of Sintilimab
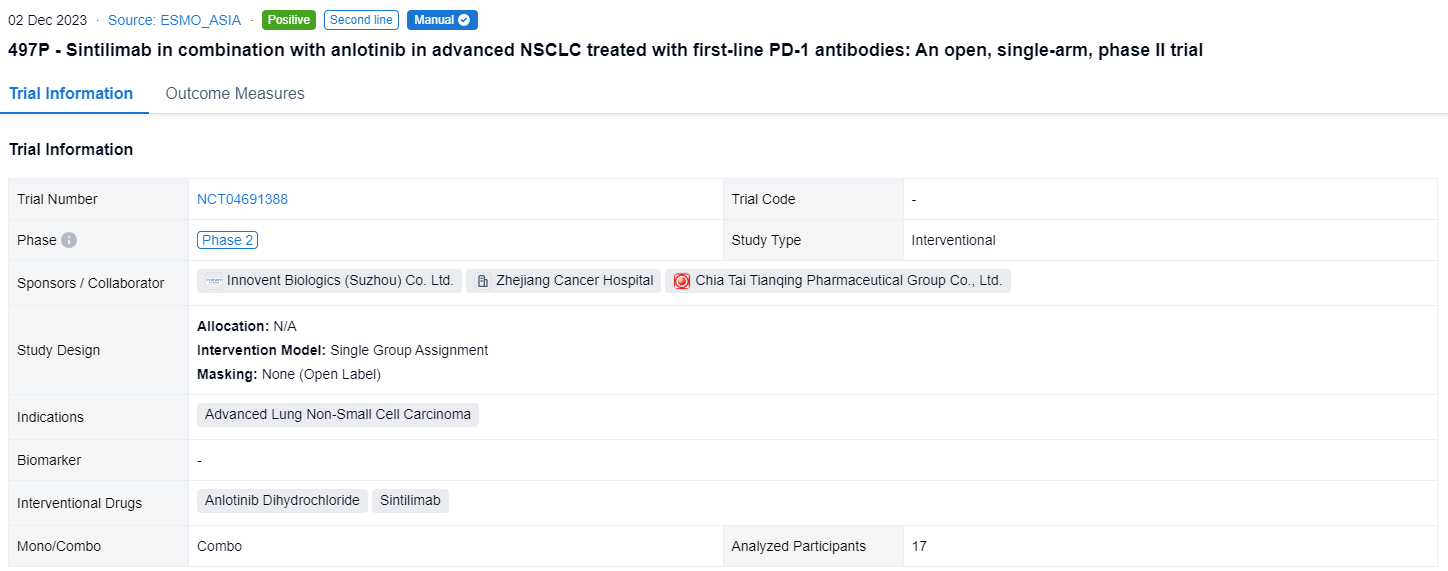
This single group assignment, open-labeled clinical trial (NCT04691388) was designed to evaluate the efficacy and safety of sintilimab in combination with anlotinib in NSCLC disease progressed after first-line treatment with anti-PD-1 antibody.
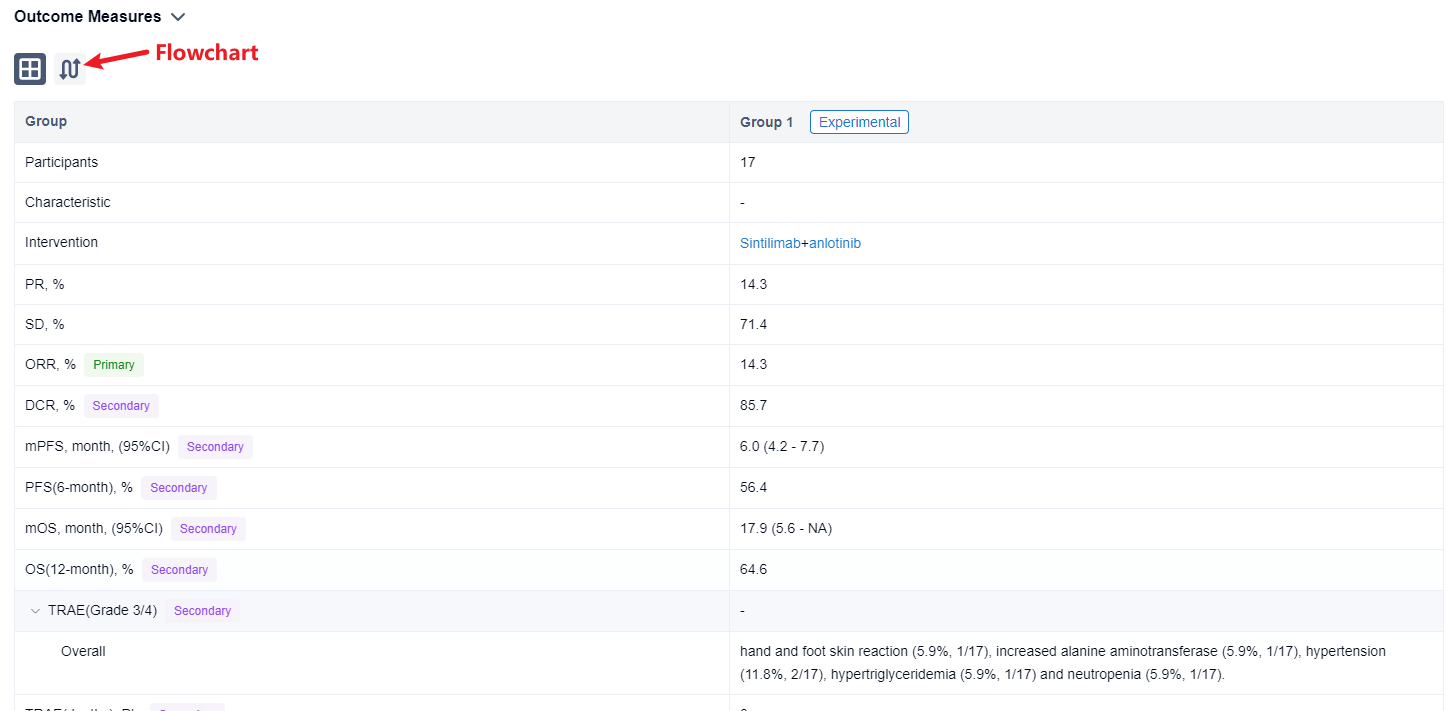
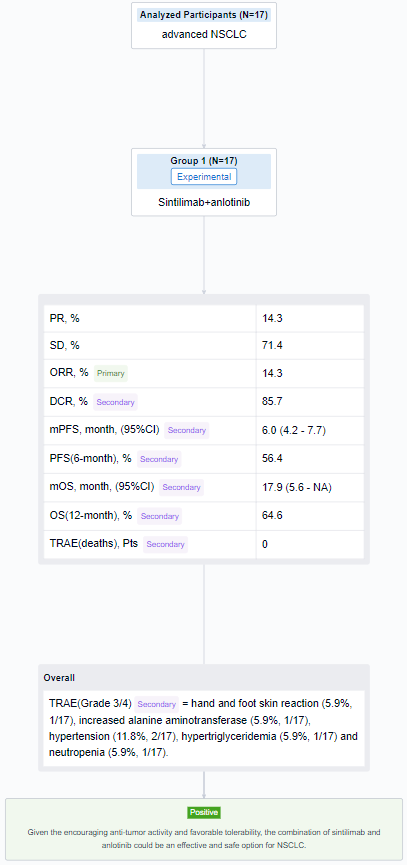
In this study, NSCLC patients with disease progression after first-line treatment with anti-PD-1 antibody were enrolled. Sintilimab (200 mg, day 1) in combination with anlotinib (12mg, day 1 to 14) were given every 3 weeks. Primary end point is objective response rate (ORR) evaluated via RECIST 1.1. Key secondary endpoints include progression-free survival (PFS), overall survival (OS), disease control rate (DCR) and safety (CTCAE 5.0).
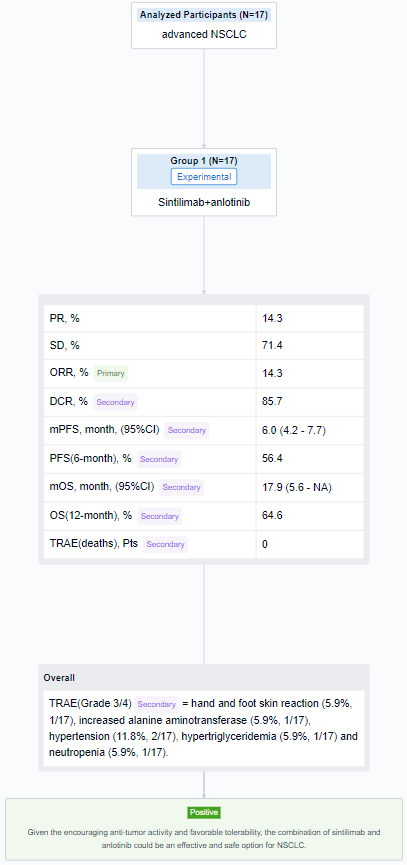
The result showed that from 3/2021 to 6/2023, 17 patients were enrolled. The median age was 63 years old (range 45-73) and 100% (17/17) was male. In 14 patients with efficacy data, 14.3% (2/14) achieved partial response (PR) and 71.4% (10/14) achieved stable disease (SD). ORR and DCR were 14.3% (and 85.7%, respectively. Until the last follow-up, the median PFS was 6.0 months(95% CI, range 4.2-7.7 months), 6-month PFS rate was 56.4%. The median OS was 17.9 months(95% CI, range 5.6-NA months), 12-month OS rate was 64.6%. Grade 3/4 treatment-related adverse events (TRAEs) included: hand and foot skin reaction (5.9%, 1/17), increased alanine aminotransferase (5.9%, 1/17), hypertension (11.8%, 2/17), hypertriglyceridemia (5.9%, 1/17) and neutropenia (5.9%, 1/17). No treatment-related deaths occurred.
It can be concluded that the combination of sintilimab and anlotinib could be an effective and safe option for NSCLC.
How to Easily View the Clinical Results Using Synapse Database?
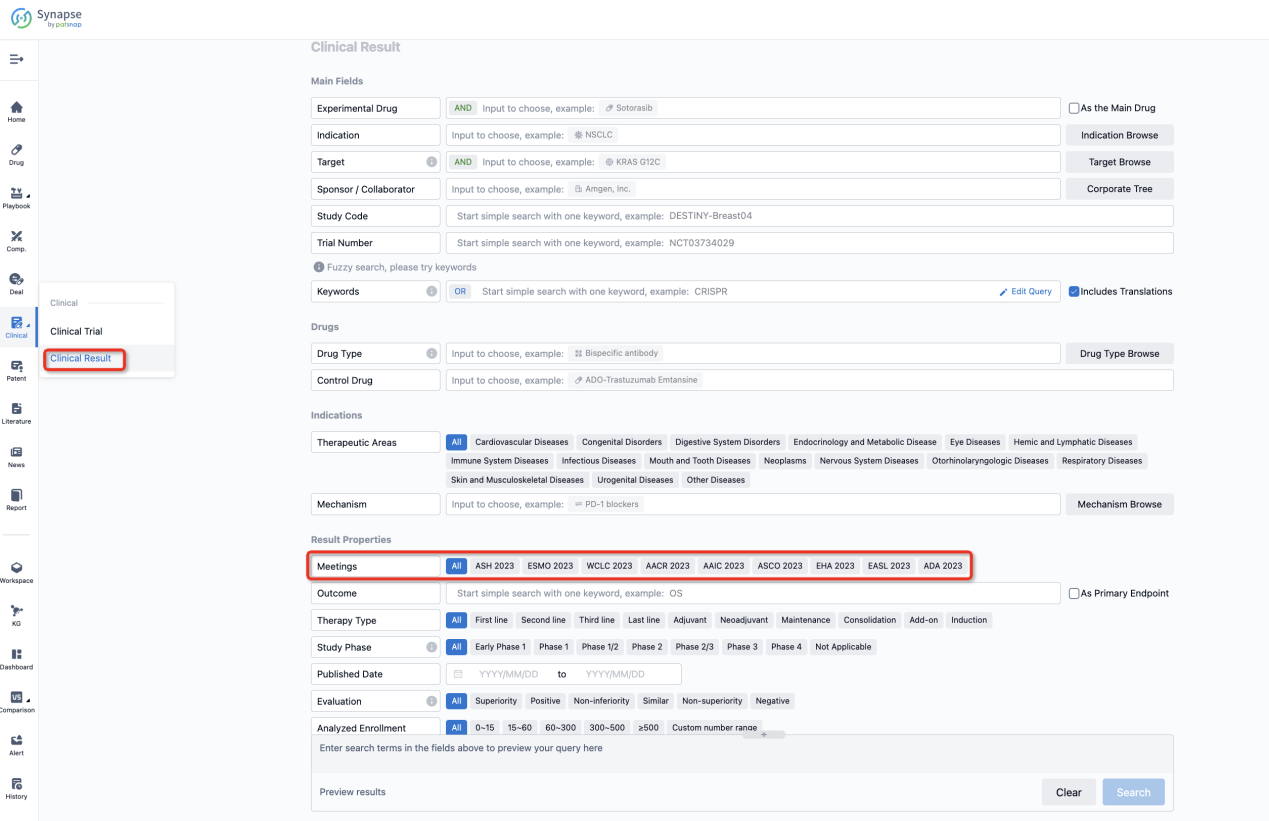
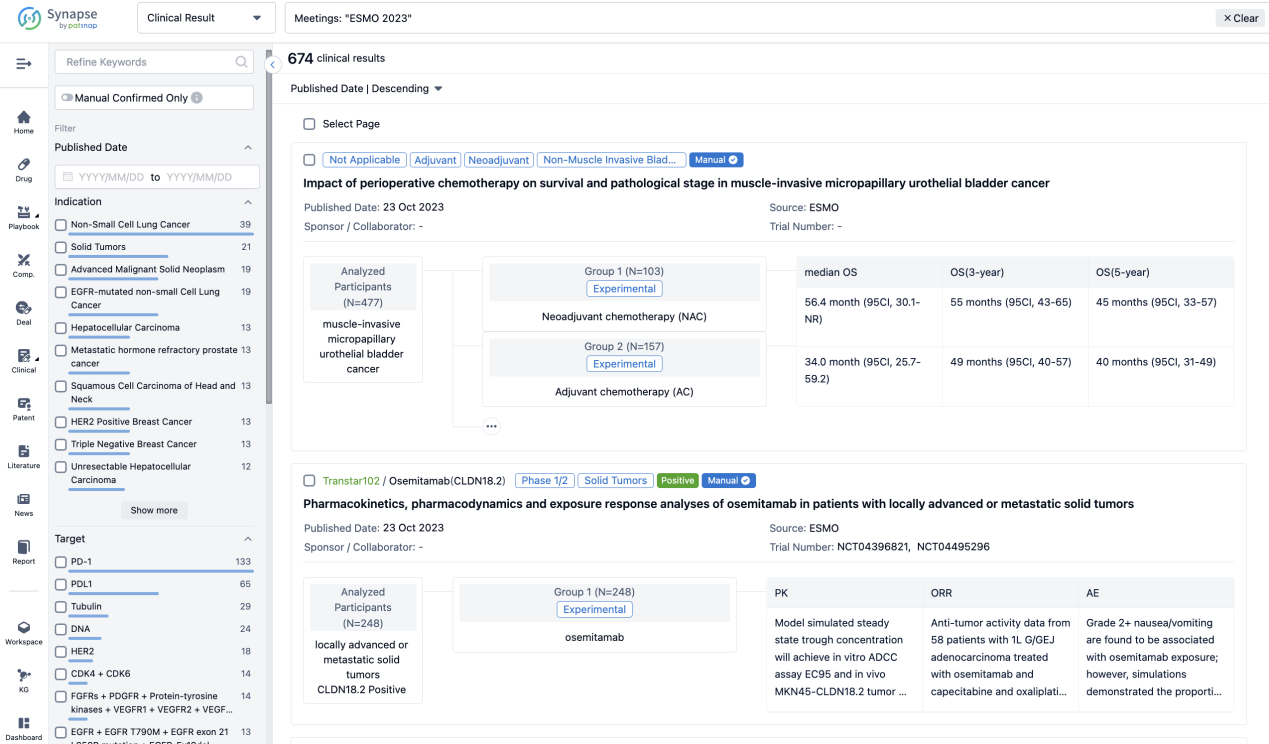
If you want to know the other clinical results of popular conferences, please lick on the “Clinical Results” on the homepage of Patsnap Synapse, which provides multi-dimensional screening and filtering of drugs, indications, targets, companies, result evaluation, release date, popular conferences, etc. to help you quickly locate the data you need.
Select the clinical meeting you are interested in, such as ESMO. In the results, you can quickly locate the data you want to view by indication, phase and drug name.
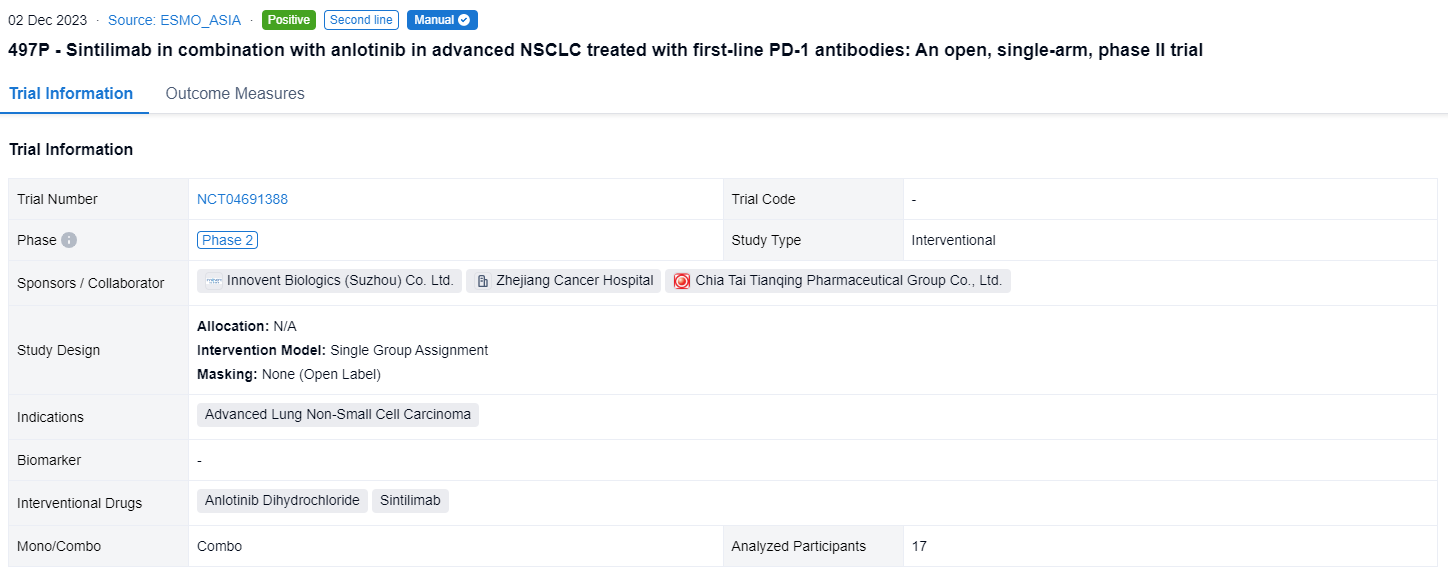
A single result clearly shows important information such as registration number, phase, indication, Sponsor/Collaborator, biomarker, Trial number, dosing regimen and more.
If you would like to view more information about this result, you can go to the result detail page by clicking on the title.
Above the headings, we provide the original source of the outcome data. The basic information is supplemented with more information beyond the list, such as company, study. design, etc.
In the important Outcome Measures section, we provide both list and flowchart forms, which are convenient for you to overview the comparison group information and core indicator data.
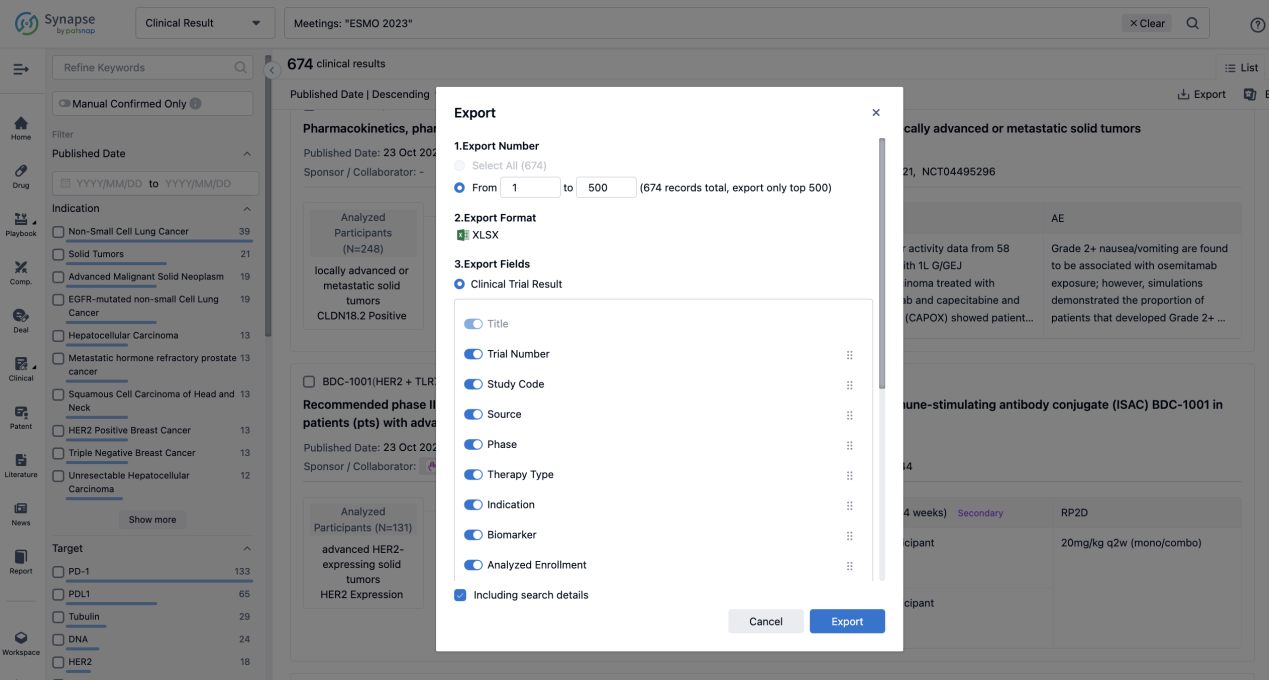
Finally, if you need to download these results, you can conveniently check the check boxes on the left side of the list, or directly click the "Export" button to download the data for personalized analysis and file sharing.
Click on the image below to embark on a brand new journey of drug discovery!