First Patient Dosed in Tubulis Phase I/IIa ADC Trial for TUB-040 in Ovarian and Lung Cancer
Tubulis announced that the first patient has been treated in its first Phase I/IIa trial. The study is evaluating Tubulis’ next-generation antibody-drug conjugate TUB-040 in patients with platinum-resistant high-grade ovarian cancer or relapsed/refractory adenocarcinoma non-small cell lung cancer, who have exhausted other available treatment options.
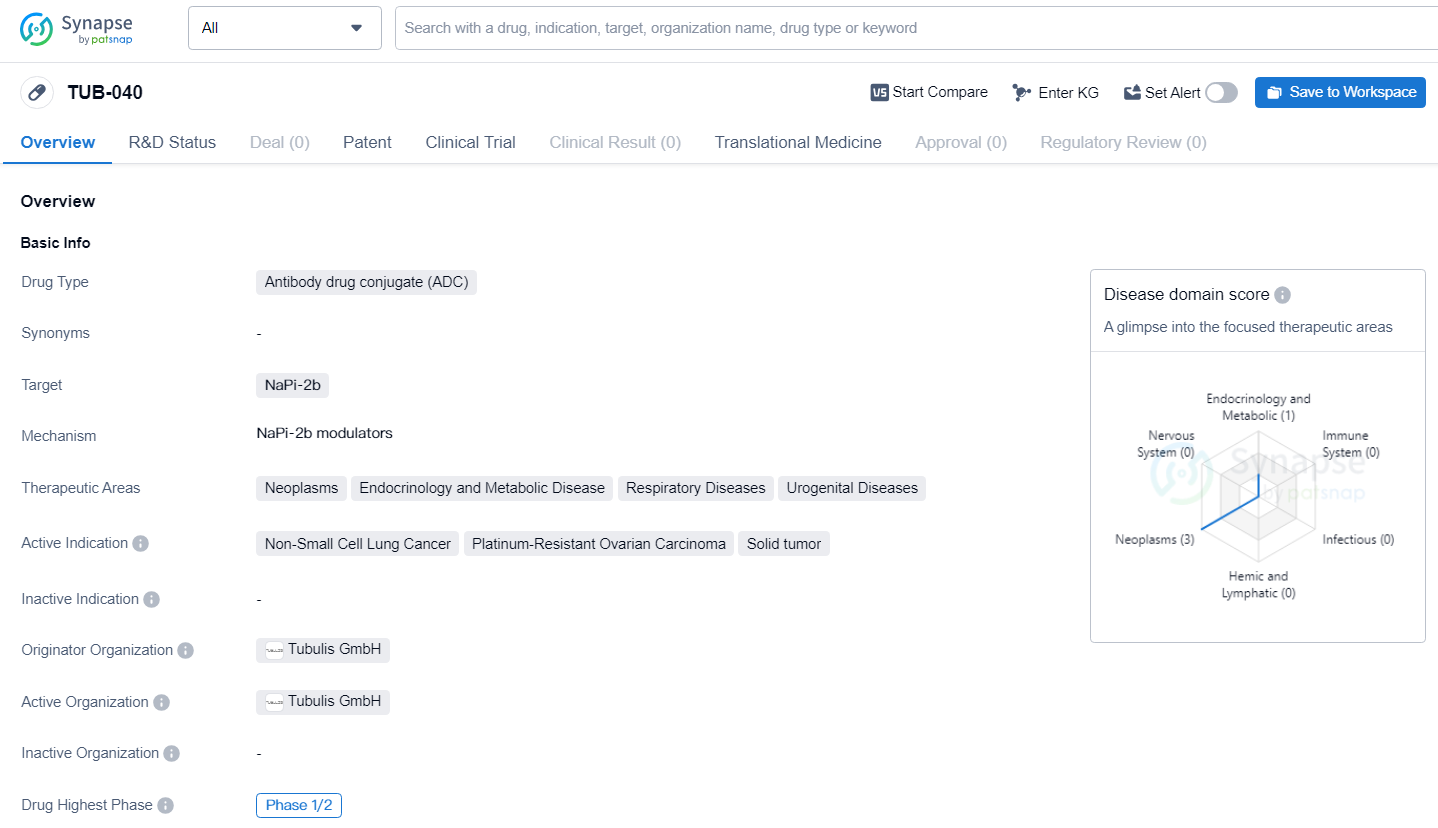
👇Unlock in-depth information about this drug - its R&D Status, Core Patent, Clinical Trials, and Global Approval Status. Click on the image below and explore the latest data immediately.
TUB-040 targets NaPi2b, a highly overexpressed antigen in ovarian cancer and lung adenocarcinoma. The candidate is the first to enter the clinic from the company’s growing pipeline and represents one of Tubulis’ two lead candidates developed using its proprietary suite of platform technologies, which enable the creation of uniquely matched ADCs with superior biophysical properties.
The multicenter, first-in-human, dose escalation and optimization Phase I/IIa study aims to investigate the safety, tolerability, pharmacokinetics, and efficacy of TUB-040 as a monotherapy. The trial will be conducted in the US as well as the UK, Spain, Belgium, and Germany. Phase Ia comprises the dose escalation and will determine safety and the maximum tolerated dose or the identified dose for optimization, whereas Phase IIa will focus on dose optimization, safety, and preliminary efficacy of TUB-040.
“ADCs are beginning to show their potential as a core treatment modality replacing conventional chemotherapy for several solid tumor indications. Based on our preclinical data we are convinced that TUB-040 can represent a new option for the effective treatment of NSCLC and ovarian cancer patients,” said Günter Fingerle-Rowson, MD, PhD, Chief Medical Officer at Tubulis.
“Initiating our first clinical trial represents an important milestone for the entire Tubulis team and underscores our vision to innovate on all fronts of the ADC design for patient benefit,” said Dominik Schumacher, PhD, Chief Executive Officer and Co-founder of Tubulis. “Our objective is to achieve clinical proof-of-concept for our lead candidate, TUB-040, and validate our differentiated platform approach to ADC development.”
TUB-040 consists of a humanized, target-specific, Fc-silenced IgG1 antibody equipped with Tubulis’ proprietary Tubutecan linker-payload technology, which is based on P5 conjugation chemistry and the topoisomerase-1 inhibitor Exatecan. Tubulis recently presented a comprehensive preclinical data set at AACR, demonstrating the superior stability and minimal loss of linker-payload conjugation for their lead candidate. In a range of preclinical models, Tubulis was also able to show high and long-lasting anti-tumor responses, even at lower expression levels of NaPi2b, with an excellent safety and tolerability profile.
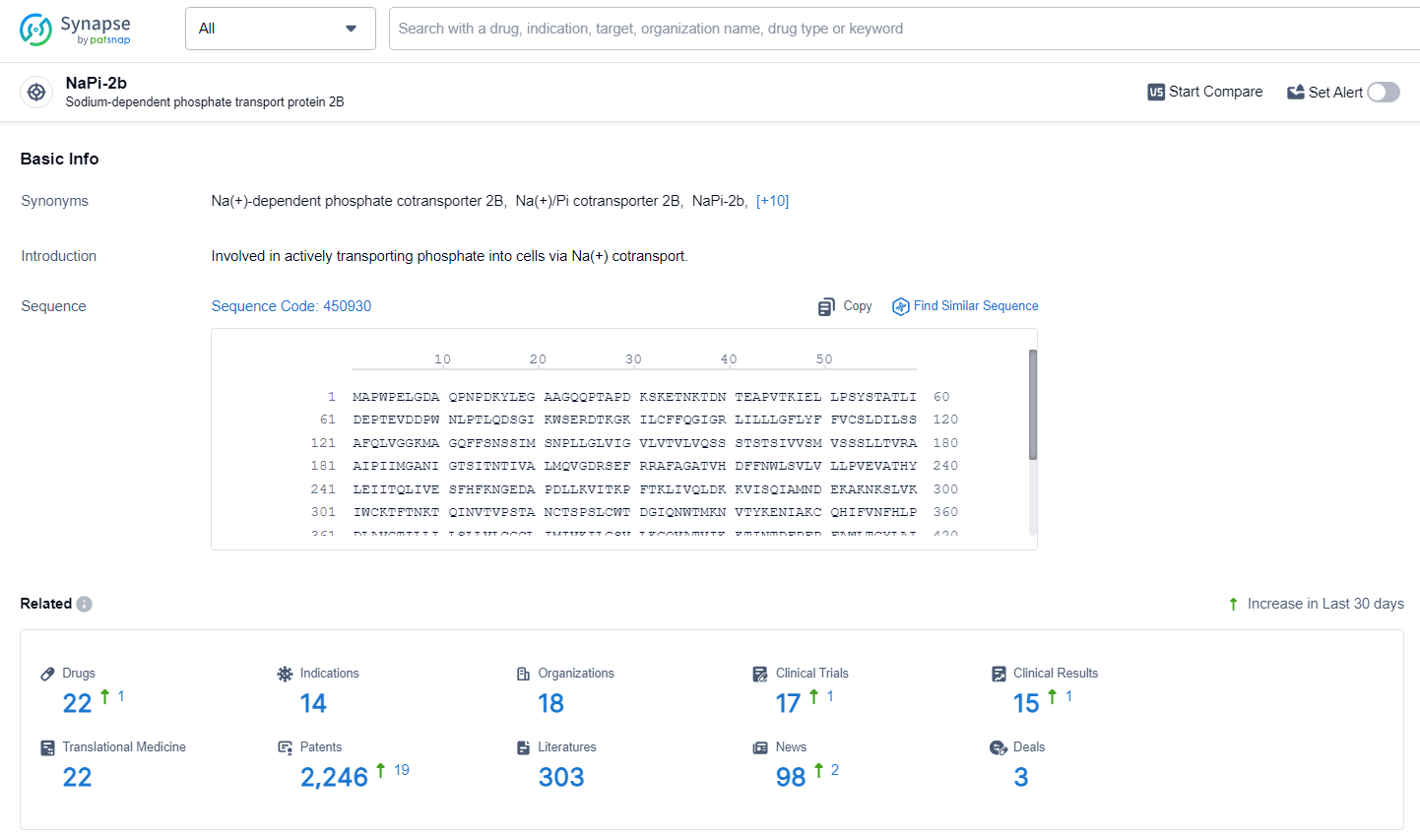
👇Explore the latest research progress on drug-related developments, indications, therapeutic organizations, clinical trials, results, and patents by clicking on the targeted picture link below. Unfold a world of comprehensive information on this target in just a click!
According to the data provided by the Synapse Database, As of June 24, 2024, there are 22 investigational drugs for the NaPi2b target, including 14 indications, 18 R&D institutions involved, with related clinical trials reaching 17, and as many as 2246 patents.
TUB-040 targets NaPi-2b for the treatment of various cancers and other diseases. The drug has advanced to Phase 1/2 of development and holds promise for addressing unmet medical needs in the identified therapeutic areas. Further clinical data and regulatory progress will be necessary to determine the ultimate potential of TUB-040 in the pharmaceutical areas.