Is Durlobactam/Sulbactam approved by the FDA?
Yes, durlobactam/sulbactam, marketed under the brand name Xacduro, is FDA approved. The approval was granted on May 23, 2023.
What is Durlobactam/Sulbactam?
Durlobactam/sulbactam is a combination antibiotic used to treat pneumonia, specifically hospital-acquired bacterial pneumonia (HABP) and ventilator-associated bacterial pneumonia (VABP) caused by susceptible isolates of Acinetobacter baumannii-calcoaceticus complex. It is intended for use in adults and is administered intravenously.
Usage and Dosage
- Dosage Form: Intravenous powder for reconstitution (0.5 g-1 g (1 g-1 g dose))
- Drug Class: Beta-lactamase inhibitors
- Administration: Given as an infusion into a vein by a healthcare provider. The usual adult dose for nosocomial pneumonia is 1 g of durlobactam and 1 g of sulbactam every 6 hours, for a duration of 7 to 14 days. The exact duration is determined based on the patient's clinical status and kidney function.
Side Effects
Common side effects of durlobactam/sulbactam may include:
- Low blood levels of potassium
- Diarrhea
- Abnormal liver function tests
- Anemia (low red blood cells)
Serious side effects that require immediate medical attention include:
- Severe stomach pain
- Diarrhea that is watery or bloody (even if it occurs months after the last dose)
- Signs of an allergic reaction: hives, difficult breathing, swelling of the face, lips, tongue, or throat
Warnings and Precautions
- Medical History: Inform your doctor if you have any allergies, especially to antibiotics such as meropenem, cephalexin, cefdinir, amoxicillin, ampicillin, penicillin, or others.
- Infections and Diarrhea: Antibiotic medicines can cause diarrhea. Notify your healthcare provider if you experience watery or bloody diarrhea.
- Pregnancy and Breastfeeding: Discuss with your doctor if you are pregnant or breastfeeding.
- Age Restriction: Not approved for use in individuals younger than 18 years old.
Treatment Considerations
- Missed Dose: In a medical setting, missing a dose is unlikely. However, if it happens, the medical team will manage it appropriately.
- Overdose: In a medical setting, an overdose would be treated quickly.
- Activity Restrictions: Follow your doctor's instructions about any restrictions on food, beverages, or activities.
Drug Interactions
- Interactions: Inform your doctor about all the medications you are taking, including prescription and over-the-counter medicines, vitamins, and herbal products. Notable interactions include probenecid, which can affect the efficacy and side effects of durlobactam/sulbactam.
Conclusion
Durlobactam/sulbactam (Xacduro) is an FDA-approved treatment for hospital-acquired bacterial pneumonia and ventilator-associated bacterial pneumonia caused by specific bacteria. The approval on May 23, 2023, marks its availability for managing these serious infections, particularly in healthcare settings. As with any antibiotic treatment, it is crucial to follow medical advice and report any adverse effects promptly to ensure safe and effective use.
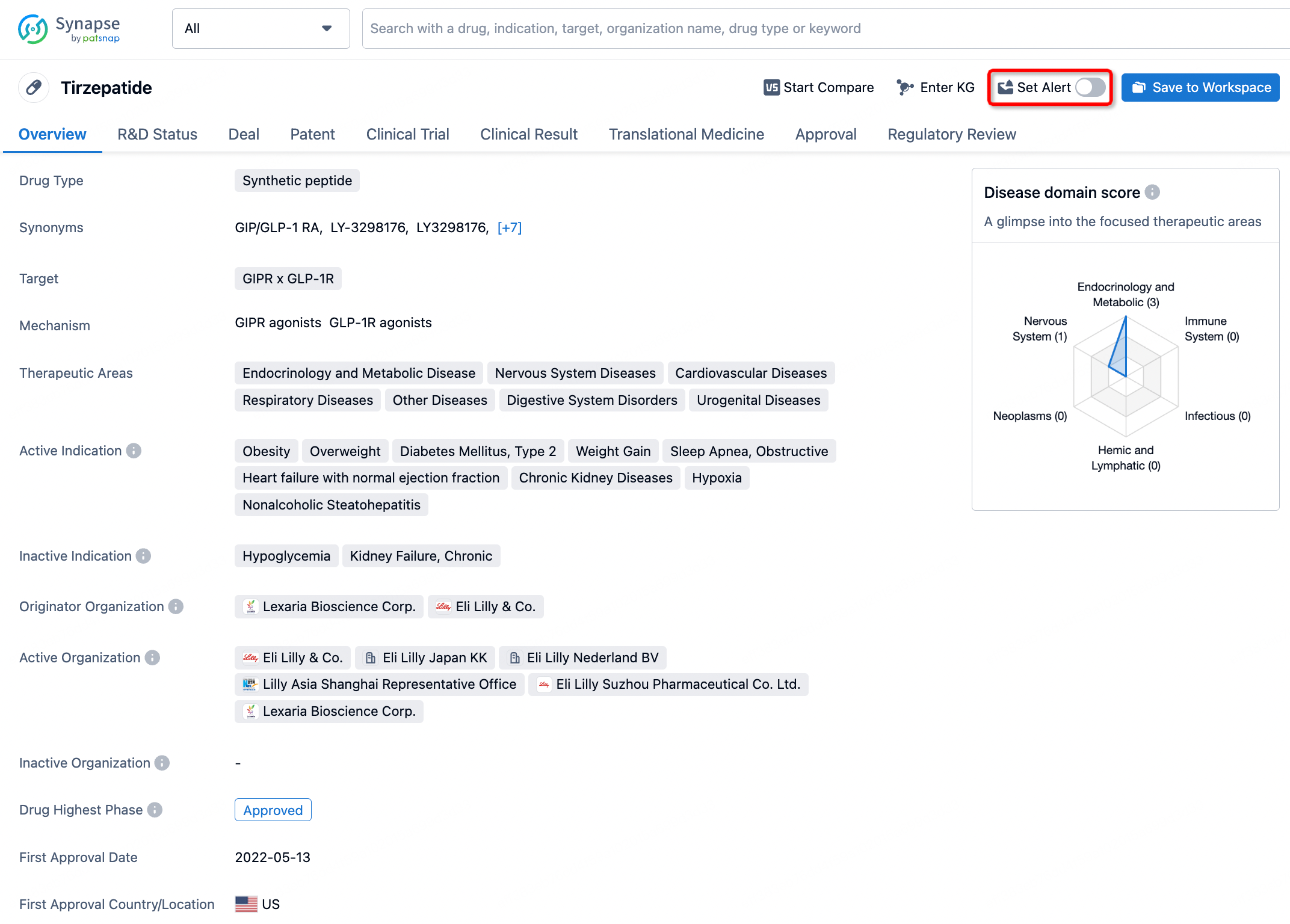
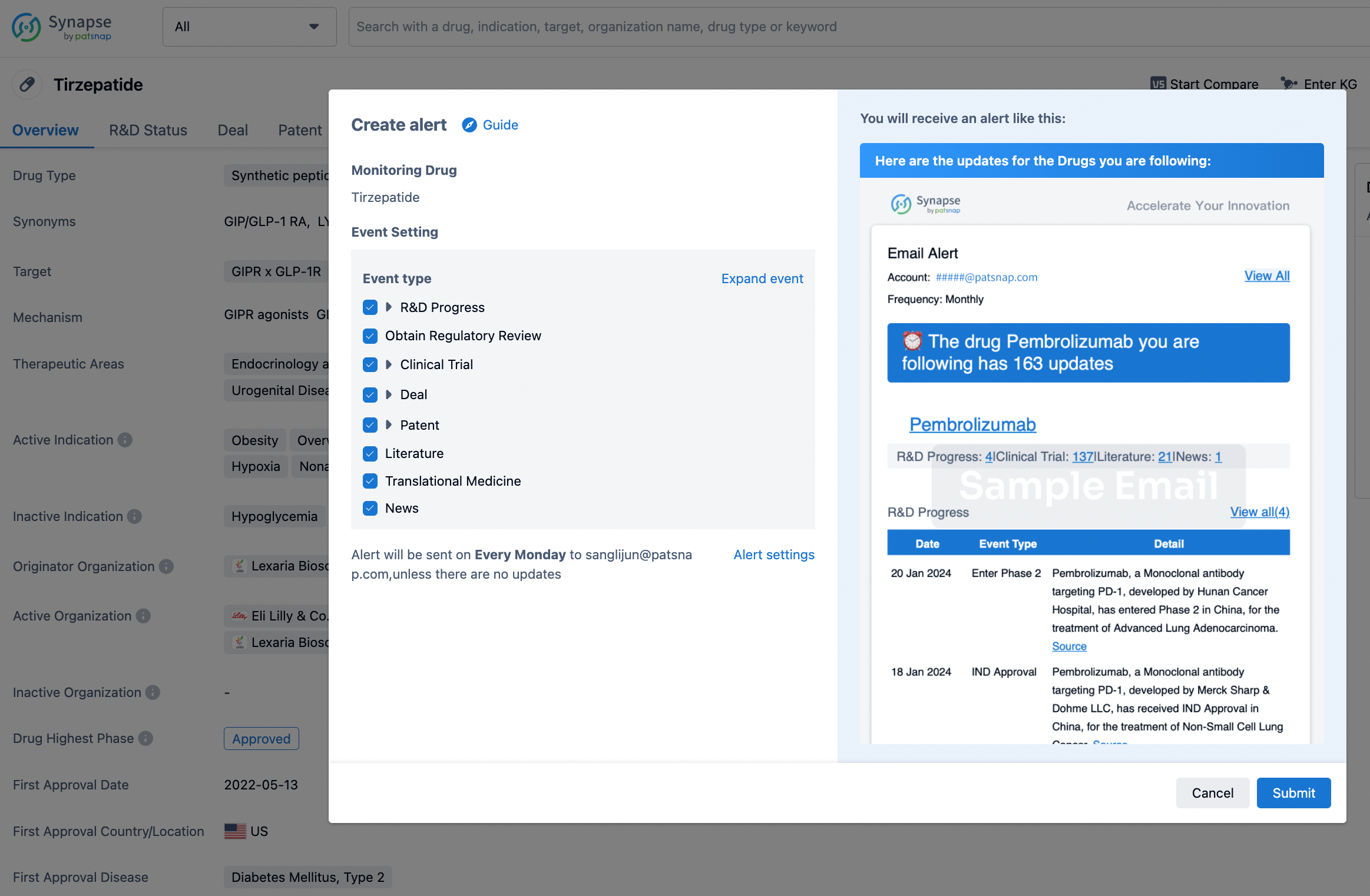
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!