Request Demo
Last update 08 Mar 2026

Fujian Medical University
Last update 08 Mar 2026
Overview
Tags
Neoplasms
Nervous System Diseases
Digestive System Disorders
Small molecule drug
Proteolysis-targeting chimeras (PROTAC)
Exosomes
Disease domain score
A glimpse into the focused therapeutic areas
No Data
Technology Platform
Most used technologies in drug development
No Data
Targets
Most frequently developed targets
No Data
| Top 5 Drug Type | Count |
|---|---|
| Small molecule drug | 25 |
| Proteolysis-targeting chimeras (PROTAC) | 5 |
| Exosomes | 4 |
| Chemical drugs | 2 |
| Synthetic peptide | 2 |
Related
52
Drugs associated with Fujian Medical UniversityMechanism EDG6 modulators [+3] |
Active Org. |
Originator Org. |
Active Indication |
Drug Highest PhaseApproved |
First Approval Ctry. / Loc. United States |
First Approval Date21 Sep 2010 |
Target- |
Mechanism- |
Active Org. |
Originator Org. |
Inactive Indication |
Drug Highest PhaseApproved |
First Approval Ctry. / Loc. China |
First Approval Date01 Jan 1997 |
337
Clinical Trials associated with Fujian Medical UniversityNCT07314203
Clinical Efficacy of Adebrelimab With or Without Apatinib Mesilate and SOX Neoadjuvant Therapy in Locally Advanced Gastric Cancer
Exploring the pathological complete response rate (pCR) of locally advanced gastric cancer treated with adebelimab combined or not combined with apatinib mesylate and SOX neoadjuvant therapy
Start Date25 Dec 2026 |
Sponsor / Collaborator |
NCT07275034
A Virtual Reality-Based Horticultural Planting Program for Elderly With Subthreshold Depression:A Randomized Controlled Pilot Study
Subthreshold depression represents a state of psychological sub-health between normal individuals and clinical depression. It constitutes a high-risk stage for developing clinical depression and a critical phase for alleviating depressive symptoms. However, current research predominantly focuses on treating depression in the elderly, with insufficient attention to interventions for high-risk populations or limited to cross-sectional investigations of subthreshold depression risk factors. Therefore, prioritizing subthreshold depression in the elderly and implementing early interventions is essential. A 2024 article in The Lancet suggests that subthreshold depression treatment should prioritize psychotherapy and lifestyle adjustments over medication. Horticultural therapy, an interdisciplinary approach integrating horticulture, medicine, and psychology, demonstrates unique advantages over traditional medical treatments. In practical application, however, limited resources may hinder adequate support for horticultural activities, compromising activity quality, reducing therapeutic efficacy, and restricting the widespread adoption of horticultural therapy. Integrating VR technology with horticultural therapy can provide patients with more comprehensive, personalized, and effective mental health treatment plans, empowering them toward healthier, more positive lives. Therefore, the research team designed a VR horticultural intervention system for elderly individuals with subthreshold depression based on the Social Participation Competence Framework.
Start Date01 Feb 2026 |
Sponsor / Collaborator |
NCT07383909
Evaluation of the Organ-Protective Effects of Remote Ischemic Preconditioning in Patients Undergoing Surgery for Acute Type A Aortic Dissection: A Multicenter, Prospective, Double-Blind, Randomized Controlled Trial
The goal of this clinical trial is to learn if a technique called remote ischemic preconditioning (RIPC) helps protect organs during emergency surgery for acute type A aortic dissection (ATAAD). The main questions it aims to answer are:
Does RIPC reduce the risk of major complications after surgery, such as heart, brain, or kidney problems?
Is RIPC safe to use during emergency ATAAD surgery?
Researchers will compare the RIPC group to a control group (who will receive a placebo) to see if RIPC can reduce complications after surgery.
Participants will:
Receive either RIPC or a sham intervention during their surgery.
Be monitored for up to 30 days after surgery for complications.
Have follow-up visits at 3 months, 1 year, and then yearly for up to 5 years to track their recovery.
Does RIPC reduce the risk of major complications after surgery, such as heart, brain, or kidney problems?
Is RIPC safe to use during emergency ATAAD surgery?
Researchers will compare the RIPC group to a control group (who will receive a placebo) to see if RIPC can reduce complications after surgery.
Participants will:
Receive either RIPC or a sham intervention during their surgery.
Be monitored for up to 30 days after surgery for complications.
Have follow-up visits at 3 months, 1 year, and then yearly for up to 5 years to track their recovery.
Start Date05 Jan 2026 |
Sponsor / Collaborator |
100 Clinical Results associated with Fujian Medical University
Login to view more data
0 Patents (Medical) associated with Fujian Medical University
Login to view more data
11,100
Literatures (Medical) associated with Fujian Medical University01 May 2026·JOURNAL OF COLLOID AND INTERFACE SCIENCE
Gallium-enriched bimetallic nanomedicine for cervical tumour suppression via bioimaging-guided surgery and highly synergistic reactive oxygen species burst
Article
Author: Zhang, Shufei ; Sheng, Ying ; Wang, Ao ; Yang, Lian ; Wang, Ganxin ; Liu, Jianfeng ; Hong, Li ; Huang, Jinling ; Jiang, Suhua ; Li, Bingshu
Cervical cancer continues to pose a substantial global health threat and is the fourth most prevalent cancer among women. Persistent infection with high-risk human papillomavirus is the main cause of the development and progression of these tumours. Unfortunately, the effectiveness of clinical treatments is frequently limited. In this research, we developed a novel one-step method to engineer mesoporous gallium-enriched cerium oxide nanoparticles (Ga&Ce). Indocyanine green (ICG) dye was then encapsulated in the mesopores, creating the Ga&Ce-ICG nanomedicine. This nanomedicine has the remarkable ability to precisely accumulate in tumours and respond to the acidic tumour microenvironment. As a result, ICG, Ce3+/Ce4+, and Ga3+ are released inside the cells from the bimetallic nanomedicine. Meaningfully, when exposed to laser irradiation, a photothermal-reactive oxygen species (ROS) burst is generated through bimetallic catalysis. Specifically, the engineered Ga&Ce-ICG can accurately define the boundaries of tumours. Under the guidance of fluorescence imaging, tumours can be fully excised. These two features effectively suppress tumour cells both in vitro and in vivo. In particular, Ga&Ce-ICG exhibits prolonged blood circulation and outstanding biocompatibility. In conclusion, this study introduces a promising nanomedicine for cancer treatment, broadening the application of mesoporous Ga&Ce in combined cancer therapies.
01 Apr 2026·BIOMATERIALS
“In situ endothelial modulation and transduction” strategy driven by biomimetic H2S delivery system for targeted repair of vascular injury
Article
Author: Yao, Peisen ; Zhang, Yibin ; Li, Shengnan ; Huang, Xiaofen ; Niu, Xuegang ; Kang, Dezhi ; Gao, Bin ; Lin, Yuanxiang
Vascular recanalization mediated by the interventional therapy can reduce cardio-cerebrovascular disease burden. However, its long-term outcomes are often undesired owing to (1) inevitable mechanical damage to the vasculature that triggers pathological remodeling, presented as local inflammation/oxidative stress, intimal hyperplasia, and delayed endothelial healing; and (2) long-term and frequent use of antiplatelet drugs, which increase bleeding risks. These challenges highlight the necessity for rapid vascular repair and minimizing dosing frequency, which are currently unmet due to the lack of a highly efficient delivery/therapy strategy. Herein, we formulated a damaged vascular endothelial-targeted hydrogen sulfide (H2S) nanomedicine, utilizing the endothelial cells (ECs) as a delivery destination rather than the traditional smooth muscle cells (SMCs) for overcoming drug delivery barriers. This drug targets the ECs, where it releases H2S in a sustained manner to promote endothelial regeneration and in situ transduce signaling from ECs for suppressing SMC-mediated intimal hyperplasia and reprogramming Mφ to inhibit local inflammation. A single dose of therapy achieved satisfactory vascular repair and safety within 28 days in the carotid artery injury model. This study provides a novel solution for vascular repair and advances the development of a drug delivery approach.
01 Apr 2026·Intensive and Critical Care Nursing
Effect of diary intervention on sleep quality and risk of stress disorders among critically ill children in pediatric intensive care units and on their parents’ anxiety level: A randomized controlled trial
Article
Author: Liao, Jinhua ; Lin, Shuting ; Lin, Jinling ; Xin, Huining ; He, Yingshuang ; Zhang, Yuhong ; Cheng, Xiao ; Liu, Guihua ; Li, Sijia
OBJECTIVES:
To investigate benefits of diary intervention in a pediatric intensive care unit (PICU) on sleep quality and stress-related disorders in critically ill children and anxiety and depression in their parents.
METHODS:
This single-blind, two-arm, randomized controlled trial enrolled 94 dyads. The control group (n = 47) received standard care; the experimental group (n = 47) additionally received a diary intervention. We compared the sleep quality and stress disorders of children, and anxiety, depression, and stress disorders among parents at various time-points, between groups.
RESULTS:
Data of 83 dyads were ultimately analyzed (loss-to-visit rate: 11.7 %). During the PICU stay, the experimental group showed significantly shorter total awakening time of each sleep episode and mean duration of each awakening per sleep session than the control group (P = 0.006; P = 0.032). The Childhood Stress Disorder Checklist scores of children in the experimental group were significantly lower than those in the control group at 1 and 3 months post-discharge (P = 0.003; P = 0.006). Parents in the experimental group reported significantly lower anxiety scores at PICU discharge and 1 month thereafter (P < 0.001; P = 0.015). At discharge, depression scores were significantly lower in the experimental group; this difference remained significant at 1 and 3 months post-discharge (P < 0.001; P < 0.05). At PICU discharge, parental scores were significantly lower in the experimental group than in the control group (P < 0.001).
CONCLUSIONS:
The PICU diary intervention effectively improved the children's sleep quality and was associated with stress reduction in parents during their children's PICU stay and up to 3 months post-discharge. The intervention was also associated with a reduction in negative parental emotions up to 3 months after PICU discharge.
IMPLICATIONS FOR CLINICAL PRACTICE:
These findings offer valuable insights into supporting the psychological well-being of pediatric patients and their caregivers during and after PICU admission.
5
News (Medical) associated with Fujian Medical University30 Jan 2026
FRIDAY, Jan. 30, 2026 -- Elevated body mass index (BMI) and physical inactivity are metabolic drivers of hypertension -related chronic kidney disease (HT-CKD), with elevated BMI exerting a greater impact, according to a study published online Jan. 11 in Frontiers in Nutrition .
Xiaohua Lin, from The First Affiliated Hospital of Fujian Medical University in Fuzhou, China, and colleagues quantified the global burden of HT-CKD attributable to elevated BMI or low physical activity from 1990 to 2021 and projected trends to 2050 based on the Global Burden of Disease 2021 data.
The researchers found that HT-CKD attributable to elevated BMI caused 179,788 deaths and 4.26 million disability-adjusted life-years (DALYs) globally in 2021, representing increases of 392.9 and 322.2 percent, respectively, since 1990. A total of 4,479 deaths and 77,879 DALYs were caused by HT-CKD attributable to low physical activity, with similar substantial growth. For both risk factors, age-standardized rates (ASRs) for deaths and DALYs showed significant global upward trends from 1990 to 2021. With age, the overall disease burden increased, peaking in those aged 80 years and older and reaching maximal ASRs in those aged 95 years and older for both metrics and risk factors. Continued increases in deaths and DALYs attributable to both risk factors were seen from 2022 to 2050 in the autoregressive integrated moving average and the exponential smoothing models, especially for elevated BMI.
"These findings underscore elevated BMI and low physical activity as major, modifiable metabolic drivers of the growing HT-CKD pandemic, necessitating urgent and targeted public health interventions worldwide," the authors write.
Abstract/Full Text

AHA
13 Jan 2026
NANTONG, China, Jan. 13, 2026 /PRNewswire/ -- Ractigen Therapeutics is pleased to announce that the first patient has been dosed in the Phase II clinical trial of RAG-17, an innovative siRNA therapy targeting SOD1-mutated amyotrophic lateral sclerosis (ALS). The initial dosing occurred at Second Affiliated Hospital, Zhejiang University School of Medicine, under the leadership of Dr. Zhi-Ying Wu, head of the Department of Medical Genetics/Center for Rare Diseases.
This Phase II trial is a randomized, double-blind, placebo-controlled, multiple ascending dose (MAD) study designed to evaluate the safety, tolerability, pharmacokinetics (PK), pharmacodynamics (PD), and preliminary efficacy of repeated intrathecal injections of RAG-17 in patients with SOD1 mutations. The participating sites include Beijing Tiantan Hospital, Capital Medical University (led by Dr. Yi-Long Wang), The Second Affiliated Hospital, Zhejiang University School of Medicine (led by Dr. Zhi-Ying Wu), West China Hospital of Sichuan University (led by Dr. Hui-Fang Shang), Fujian Medical University Union Hospital (led by Dr. Zhan-Yu Zou) and the First Affiliated Hospital, Sun Yat-sen University (led by Dr. Jing-Sheng Zeng) .
This milestone marks the progression into the Phase II stage of the study, building on the successful completion of the Phase I single ascending dose (SAD) portion. The transition to the Phase II MAD phase was supported by highly encouraging data from the SAD study, which demonstrated an exceptional safety profile and sustained biomarker modulation following a single injection, including significant reductions in CSF SOD1 protein and improvements in plasma neurofilament light chain (NfL) levels. These results validate the capability of our proprietary SCAD™ delivery platform for central nervous system (CNS) in humans.
Dr. Long-Cheng Li, Founder and CEO of Ractigen Therapeutics, emphasized Ractigen's dedication to advancing treatments for ALS: "The successful dosing of the first patient represents a significant milestone in our mission to develop transformative therapies for ALS. The positive results from the Phase I trial affirm the promise of RAG-17 and provide us with strong confidence in its potential to markedly improve the lives of patients suffering from SOD1-mutated ALS."
Dr. Zhi-Ying Wu shared optimistic expectations for the Phase II trial: "We are excited to be part of this important study. Based on the improvements we have observed clinically, we look forward to further investigating RAG-17's potential to enhance the quality of life for ALS patients as we progress."
About RAG-17
RAG-17 is an investigational siRNA therapeutic candidate designed using Ractigen's proprietary SCAD™ delivery platform technology to specifically target and silence the superoxide dismutase 1 (SOD1) gene mRNA. Mutations in the SOD1 gene cause a toxic gain-of-function and are a known cause of familial ALS. By reducing the production of the toxic mutant SOD1 protein, RAG-17 aims to slow or halt the progression of SOD1-ALS.
RAG-17 has obtained Orphan Drug Designation (ODD) from the U.S. Food and Drug Administration (FDA) and been selected for the CARE Program of the Center for Drug Evaluation (CDE), National Medical Products Administration (NMPA), which facilitates the accelerated development of rare disease therapies.
About ALS
Amyotrophic Lateral Sclerosis (ALS) is a progressive neurodegenerative disease affecting nerve cells in the brain and spinal cord, leading to muscle weakness, paralysis, and ultimately death, typically within three to five years of diagnosis. SOD1 gene mutations account for approximately 10-20% of familial ALS cases and about 1-2% of sporadic ALS cases. There remains a critical unmet medical need for effective treatments that can slow or stop disease progression.
About Ractigen Therapeutics
Ractigen Therapeutics is a clinical-stage biopharmaceutical company innovating next-generation RNA therapeutics, with a primary focus on small activating RNAs (saRNAs) developed through its clinically validated RNA activation (RNAa) technology. Leveraging proprietary delivery platforms such as SCAD™, LiCO™, and GLORY™, Ractigen is advancing a robust pipeline addressing unmet medical needs in oncology, neurological diseases, and genetic disorders. Its versatile technologies also enable the rapid development of RNA-based solutions, including siRNAs, where applicable, to target life-threatening, fast-progressing conditions such as those in the CNS. Committed to scientific excellence and patient-centered innovation, Ractigen strives to transform healthcare through the power of RNA therapeutics. For more information, visit .
SOURCE Ractigen Therapeutics
21%
more press release views with
Request a Demo
Phase 2Orphan Drug
14 Aug 2025
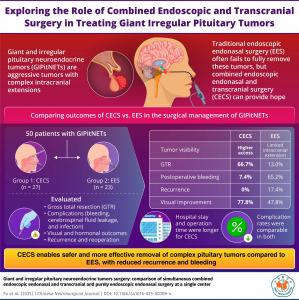
Two-team surgical approach removes more complex pituitary tumors, reduces bleeding risk, and lowers recurrence compared to traditional endonasal surgery
BEIJING, BEIJING, CHINA, August 14, 2025 /
EINPresswire.com
/ -- The pituitary gland, located at the base of the brain, secretes hormones that regulate vital body functions and control the activity of other hormone-secreting glands. Pituitary neuroendocrine tumors (PitNETs) are abnormal growths in this gland. In recent years, endoscopic endonasal surgery (EES), a minimally invasive technique, has become a widely used method for treating these tumors. In this approach, an endoscope is inserted through the nasal passages and sinuses—a route referred to as endonasal.
However, giant and irregular pituitary neuroendocrine tumors (GIPitNETs) pose a significantly greater challenge. These tumors are typically larger than 4 cm and often extend beyond the sella, the bony structure that houses the pituitary gland. They may grow upwards into the cranial cavity, the space within the skull that houses the brain. This extension can render the tumor inaccessible or invisible through the standard EES approach.
An alternative and innovative method used in such cases is the combined endoscopic endonasal and transcranial surgery (CECS). In this technique, one surgical team performs the EES approach while another team simultaneously carries out transcranial surgery, which involves accessing the tumor by creating small openings in the skull.
To analyze and compare the efficacy and complications of CECS and EES for GIPitNETs treatment, a team of researchers from China conducted a retrospective observational single-center cohort study. The study led by Dr. Changzhen Jiang and Dr. Xiaorong Yan from the Neurosurgery Research Institute, Fujian Medical University, China, was published in the
Chinese Neurosurgical Journal
and made available online on February 03, 2025. “We wanted to define the limitations and benefits of the two surgical procedures in the management of GIPitNETs,” says Dr. Jiang while explaining the aim of the study.
The research included 50 patients who underwent either EES or CECS between March 2018 and May 2023 at The First Affiliated Hospital of Fujian Medical University. All patients had tumors larger than 4 cm with significant intracranial extension. Endocrine tests were conducted before and after surgery to assess hormone levels. Magnetic resonance imaging (MRI) was used to evaluate tumor size and post-surgical outcomes. Researchers also analyzed hospital records for symptoms and complications, and ophthalmologists assessed visual function before and after surgery.
27 out of the 50 patients enrolled for this study were treated by CECS, and EES was performed on the remaining 23 patients. The researchers compared the obtained data using statistical analysis.
The results revealed a higher rate of gross total tumor removal (GTR) in the CECS group. GTR was achieved in 66% of patients in the CECS group, compared to just 13% in the EES group. Postoperative bleeding, a common and serious complication, was more prevalent in the EES group—65.2% compared to 7.4% in the CECS group. Additionally, all four cases of tumor recurrence due to residual tumor were reported in the EES group, suggesting that incomplete removal in EES may increase the risk of recurrence.
Interestingly, even though CECS achieved better tumor removal, visual outcomes were similar in both groups. “Partial tumor removal can also alleviate the pressure on optic nerves and lead to symptom relief. Thus, visual symptom improvement is possible after undergoing EES,” explains Dr. Yan.
However, CECS is not without its limitations. “The CECS technique requires a longer operation time and has greater surgical trauma with similar postoperative infection rates, compared to EES,” notes Dr. Jiang. Patients in the CECS group also had longer hospital stays. Despite the increased invasiveness of CECS, postoperative infection rates were comparable to those of the less invasive EES, indicating that CECS remains a safe option when performed carefully by experienced surgical teams.
Overall, the findings suggest that CECS may offer significant advantages in treating GIPitNETs, particularly when the tumor’s size or shape makes it inaccessible by EES alone. The improved GTR rate and lower complication rate point to CECS as a more effective approach for complex cases, despite the longer operation and recovery times.
The research team plans to further investigate the long-term efficacy and safety of CECS. They aim to conduct follow-up studies over extended periods and hope to analyze more patient data through large-scale multicenter collaborations. With continuous improvements in surgical techniques, approaches like CECS may help make the treatment of complex tumors like GIPitNETs safer and more successful.
***
Reference
Title of original paper: Giant and irregular pituitary neuroendocrine tumors surgery: comparison of simultaneous combined endoscopic endonasal and transcranial and purely endoscopic endonasal surgery at a single center
Journal: Chinese Neurosurgical Journal
DOI: 10.1186/s41016-025-00389-4
About Fujian Medical University
Fujian Medical University (FJMU), established in 1937, is a leading medical research university in China. It has over 20,000 full-time students and a strong faculty, more than half of whom hold doctorates or other advanced degrees. FJMU drives innovation in molecular medicine, oncology, and public health. The university hosts several national and provincial research platforms, including key laboratories and clinical trial centers. FJMU’s emphasis on translational medicine and interdisciplinary collaboration fosters cutting-edge discoveries. FJMU also participates in global research networks and offers English-medium postgraduate programs, attracting international scholars and supporting China’s broader scientific and medical advancement.
Website:
https://oec.fjmu.edu.cn/en/
About Dr. Xiorong Yan, from Fujian Medical University
Dr. Xiaorong Yan is a neurosurgeon at the Neurosurgery Research Institute, First Affiliated Hospital of Fujian Medical University in Fuzhou, China. She holds an MD in Neurosurgery from Shandong Medical University. Her work mainly focuses on endoscopic surgery for pituitary adenomas and other brain lesions. She co-led the recent comparison study of combined endoscopic endonasal‑transcranial surgery versus purely endonasal surgery for giant pituitary tumors, contributing to improved surgical strategies and patient outcomes. Dr. Yan has also contributed to other minimally invasive neurosurgical techniques, publishing clinical studies on spine and brain‑tumor procedures. She has over 20 published papers.
Funding information
This work was sponsored by Joint Funds for Innovation of Science and Technology, Fujian Province (No. 2021Y9089), University-Industry Research Joint Innovation Project of Science and Technology, Fujian Province (No. 2023Y4018), Fujian Province Finance Project (No. BPB-2022YXR), scientific research, Fujian Medical University (grant number 2022QH1098).
Yi Lu
Chinese Neurosurgical Journal
+861059978478 ext.
- luyi617@sina.cn
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
Clinical Result
100 Deals associated with Fujian Medical University
Login to view more data
100 Translational Medicine associated with Fujian Medical University
Login to view more data
Corporation Tree
Boost your research with our corporation tree data.
login
or

Pipeline
Pipeline Snapshot as of 21 May 2026
The statistics for drugs in the Pipeline is the current organization and its subsidiaries are counted as organizations,Early Phase 1 is incorporated into Phase 1, Phase 1/2 is incorporated into phase 2, and phase 2/3 is incorporated into phase 3
Discovery
3
45
Preclinical
Phase 1
3
5
Other
Login to view more data
Current Projects
| Drug(Targets) | Indications | Global Highest Phase |
|---|---|---|
CD19/CD20 CAR-T (Fujian Medical University) ( CD19 x CD20 ) | Diffuse large B-cell lymphoma recurrent More | Phase 1 |
Chimeric Natural Killer Receptor-Universal T Cells(Fujian Medical University) | Steroid Refractory Graft Versus Host Disease More | Phase 1 |
Koumine Hydrochloride | Rheumatoid Arthritis More | Phase 1 |
Exosome of Mesenchymal Stem Cells(Fujian Medical University) | Multiple Organ Failure More | Clinical |
FP671120.4 ( HMOX1 x Nrf2 ) | Sepsis More | Preclinical |
Login to view more data
Deal
Boost your decision using our deal data.
login
or

Translational Medicine
Boost your research with our translational medicine data.
login
or

Profit
Explore the financial positions of over 360K organizations with Synapse.
login
or

Grant & Funding(NIH)
Access more than 2 million grant and funding information to elevate your research journey.
login
or

Investment
Gain insights on the latest company investments from start-ups to established corporations.
login
or

Financing
Unearth financing trends to validate and advance investment opportunities.
login
or

AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free


