|
|
|
|
|
|
Drug Highest PhasePhase 1/2 |
First Approval Ctry. / Loc.- |
First Approval Date- |
GC012F注射液治疗复发/难治性多发性骨髓瘤的 I/II 期临床研究
[Translation] Phase I/II clinical study of GC012F injection in the treatment of relapsed/refractory multiple myeloma
I期研究目的:评估GC012F注射液治疗复发/难治性多发性骨髓瘤受试者的安全性与耐受性,确定II期临床研究推荐剂量(RP2D);评估GC012F注射液治疗复发/难治性多发性骨髓瘤受试者的有效性,药代动力学和药效动力学特征,免疫原性
II期研究目的:评估GC012F注射液治疗复发/难治性多发性骨髓瘤受试者的有效性;评估GC012F注射液治疗复发/难治性多发性骨髓瘤受试者的安全性,药代动力学和药效动力学特征,免疫原性
[Translation] Purpose of the Phase I study: To evaluate the safety and tolerability of GC012F injection in the treatment of relapsed/refractory multiple myeloma in subjects, and to determine the recommended dose (RP2D) of the Phase II clinical study; to evaluate the safety and tolerability of GC012F injection in the treatment of relapsed/refractory multiple myeloma Efficacy, pharmacokinetic and pharmacodynamic characteristics, and immunogenicity in patients with multiple myeloma
Phase II study purpose: To evaluate the effectiveness of GC012F injection in the treatment of subjects with relapsed/refractory multiple myeloma; to evaluate the safety and pharmacokinetics of GC012F injection in the treatment of subjects with relapsed/refractory multiple myeloma Chemical and pharmacodynamic characteristics, immunogenicity
GC007g注射液治疗异基因移植后复发难治性CD19+急性B淋巴细胞白血病的I/II期临床研究
[Translation] Phase I/II clinical study of GC007g injection in the treatment of relapsed and refractory CD19+ acute B lymphocytic leukemia after allogeneic transplantation
II期剂量扩展阶段:
主要研究目的:确证GC007g注射液在异基因移植后复发难治性CD19+ B-ALL患者中的有效性。
次要研究目的:
1) 评估GC007g注射液在异基因移植后复发难治性CD19+ B-ALL患者中的有效性与安全性;
2) 评估GC007g注射液在患者体内的增殖及存续;
3) 探索GC007g注射液输注后人体产生的针对CAR的免疫原性;
4) 检测GC007g输注后人体内的RCL情况;
5) 评估GC007g注射液输注后人体内慢病毒插入位点的风险。
[Translation] Phase II dose expansion phase:
Main research purpose: To confirm the efficacy of GC007g injection in patients with relapsed and refractory CD19+ B-ALL after allogeneic transplantation.
Secondary research purposes:
1) To evaluate the efficacy and safety of GC007g injection in patients with relapsed and refractory CD19+ B-ALL after allogeneic transplantation;
2) To evaluate the proliferation and survival of GC007g injection in patients;
3) To explore the immunogenicity of CAR produced by the human body after infusion of GC007g injection;
4) Detect RCL in human body after GC007g infusion;
5) To assess the risk of lentiviral insertion sites in humans after GC007g injection infusion.
GC019F注射液治疗CD19阳性复发或难治性急性B淋巴细胞白血病的Ⅰ/Ⅱ期临床试验
[Translation] Phase I/II clinical trial of GC019F injection in the treatment of CD19 positive relapsed or refractory acute B lymphocytic leukemia
主要目的:评估GC019F治疗CD19阳性复发或难治性急性B淋巴细胞白血病的安全性、耐受性。
次要目的:评估GC019F输注后在体内的PK情况;评估GC019F治疗CD19阳性复发或难治性急性B淋巴细胞白血病的有效性;评估GC019F输注后人体产生的免疫原性;评估外周血、脑脊液细胞因子(如适用)与细胞因子释放综合征、神经毒性的相关性;评估CRS、神经毒性与受试者初始瘤负荷和其他次要终点,包括PK、有效性的相关性(如适用);评估GC019F产品特性对临床安全性和有效性的影响(如适用)。
[Translation] Main objective: To evaluate the safety and tolerability of GC019F in the treatment of CD19-positive relapsed or refractory acute B lymphocytic leukemia.
Secondary objectives: To evaluate the PK profile of GC019F in vivo after infusion; To evaluate the efficacy of GC019F in the treatment of CD19-positive relapsed or refractory acute B lymphocytic leukemia; To evaluate the immunogenicity of GC019F in humans after infusion; Correlation of cerebrospinal fluid cytokines (if applicable) with cytokine release syndrome, neurotoxicity; assess the correlation of CRS, neurotoxicity with subjects' initial tumor burden and other secondary endpoints, including PK, efficacy (if applicable) ; Assess the impact of GC019F product characteristics on clinical safety and efficacy (if applicable).
100 Clinical Results associated with Suzhou Gracell Biotechnologies Co., Ltd.
0 Patents (Medical) associated with Suzhou Gracell Biotechnologies Co., Ltd.
100 Deals associated with Suzhou Gracell Biotechnologies Co., Ltd.
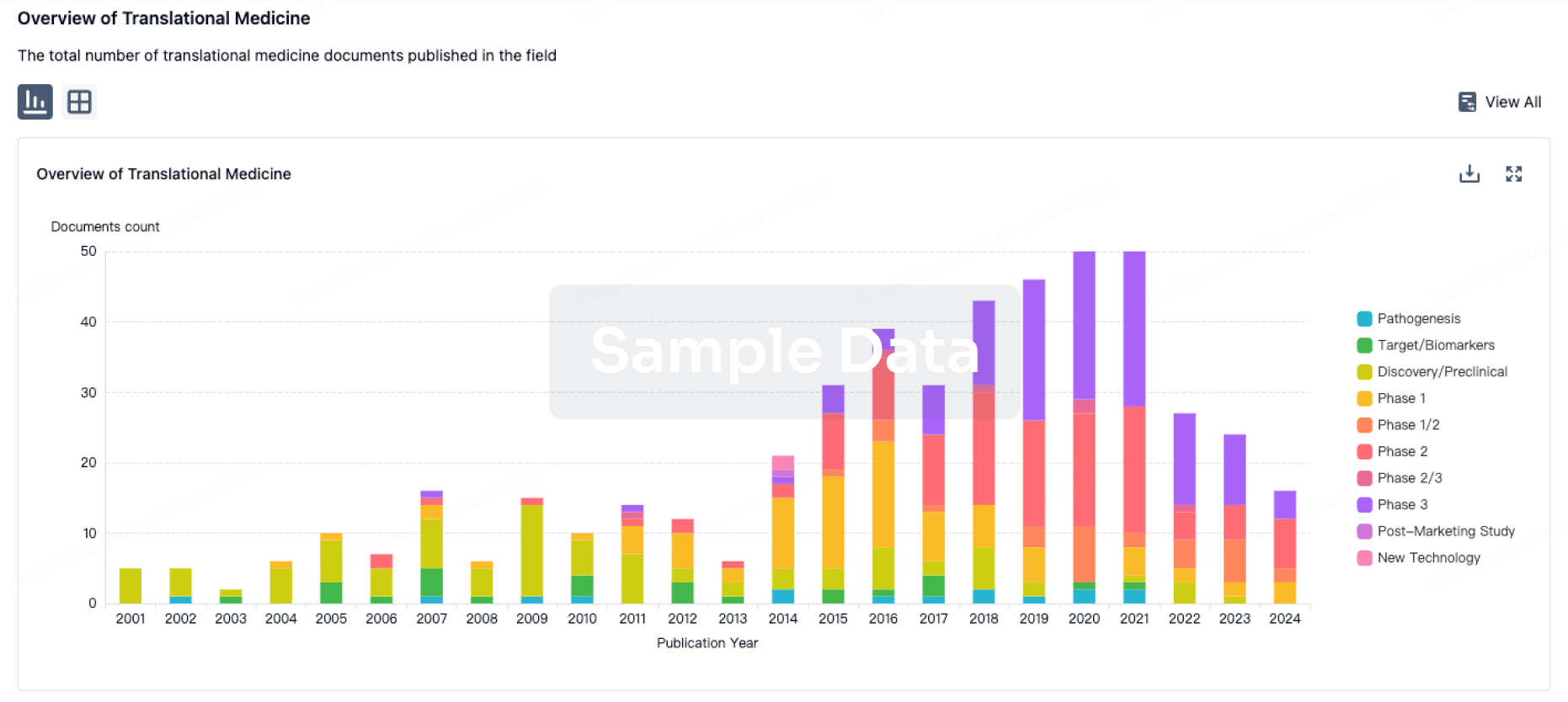
100 Translational Medicine associated with Suzhou Gracell Biotechnologies Co., Ltd.