Is Loncastuximab tesirine approved by the FDA?
Loncastuximab tesirine, marketed under the brand name Zynlonta, is a medication used for the treatment of large B-cell lymphoma, particularly in cases where the disease has relapsed or is refractory after at least two prior lines of therapy. Loncastuximab tesirine (Zynlonta) was approved by the U.S. Food and Drug Administration (FDA) on April 23, 2021. This approval provided a new treatment option for patients with large B-cell lymphoma who had not responded to other treatments.
Mechanism of Action
Loncastuximab tesirine is an antibody-drug conjugate (ADC). It combines a monoclonal antibody, which specifically targets the CD19 antigen on the surface of B-cells, with a potent cytotoxic agent. This dual-action approach allows the drug to selectively deliver the cytotoxic agent to cancerous B-cells, reducing the impact on healthy cells and enhancing the effectiveness of the treatment.
Usage and Administration
Dosage Form: Loncastuximab tesirine is available as an intravenous injection (lpyl 10 mg).
Dosage Instructions:
- The typical adult dose for lymphoma is 0.15 mg/kg administered intravenously over 30 minutes on Day 1 of each cycle (every three weeks) for two cycles. For subsequent cycles, the dose is reduced to 0.075 mg/kg administered over 30 minutes every three weeks.
- Premedication with dexamethasone is recommended to prevent serious side effects.
Administration:
- Loncastuximab tesirine is administered by a healthcare provider.
- The treatment schedule is typically once every three weeks.
- Patients may receive additional medications to manage side effects.
- Regular medical tests are required to monitor the patient's response and adjust treatment as necessary.
Side Effects and Warnings
Common Side Effects:
- Fatigue
- Rash
- Swelling
- Nausea
- Muscle or joint pain
- Increased blood sugar levels
- Changes in blood or laboratory test results
Serious Side Effects:
- Severe skin reactions, including rash, peeling, redness, or irritation
- Swelling, weight gain, chest pain, shortness of breath, or fluid build-up around the lungs
- Low blood cell counts leading to fever, chills, tiredness, mouth sores, skin sores, easy bruising, or unusual bleeding
- Signs of serious infections, such as flu-like symptoms, cough, body aches, headache, trouble breathing, or skin wounds with pain, redness, warmth, or swelling
Warnings:
- Patients are at an increased risk of serious or fatal infections. It is crucial to monitor for signs of infection and report any symptoms to a doctor immediately.
- Loncastuximab tesirine can cause harm to an unborn baby. Both men and women should use effective birth control during treatment and for several months after the last dose.
Conclusion
This antibody-drug conjugate provides a targeted approach to treatment, aiming to minimize the impact on healthy cells while effectively combating cancerous B-cells. As with any medication, it is essential to be aware of the potential side effects and to work closely with healthcare providers to ensure the best possible outcomes.
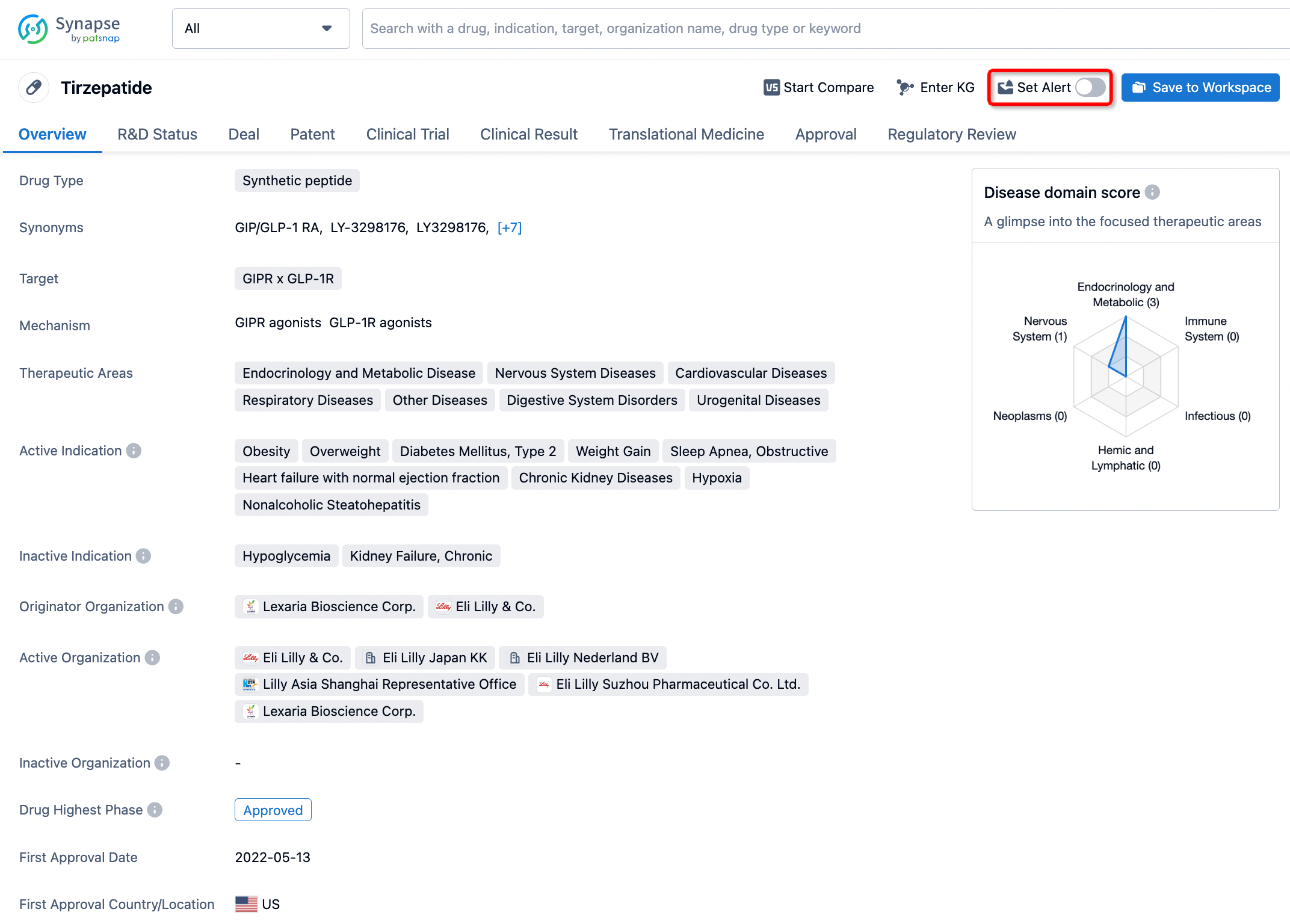
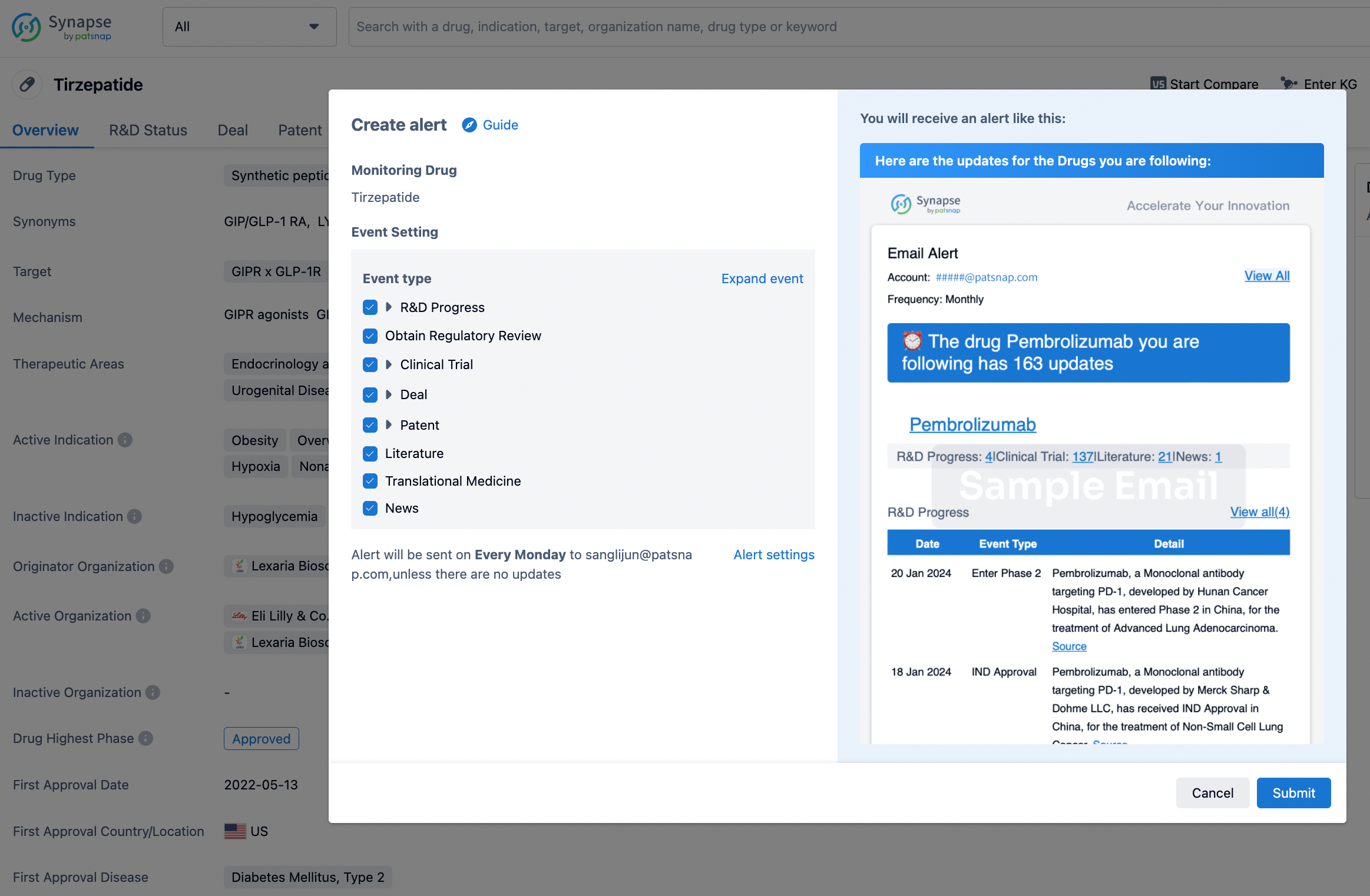
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!