Is Trijardy XR approved by the FDA?
Yes, Trijardy XR is FDA approved. The U.S. Food and Drug Administration (FDA) approved Trijardy XR on January 27, 2020, for the treatment of type 2 diabetes in adults.
What is Trijardy XR?
Trijardy XR is a combination medication that includes empagliflozin, linagliptin, and metformin. It is used alongside diet and exercise to improve blood sugar control in adults with type 2 diabetes mellitus. Additionally, it helps to reduce the risk of death from heart attack, stroke, or heart failure in adults with type 2 diabetes who also have heart disease.
Key Components:
- Empagliflozin: An SGLT2 inhibitor that helps the kidneys remove glucose from the bloodstream.
- Linagliptin: A DPP-4 inhibitor that increases hormones that help the body produce more insulin.
- Metformin: A biguanide that decreases glucose production in the liver and improves the body's sensitivity to insulin.
Usage Instructions
Trijardy XR is taken orally, typically once daily in the morning with a meal. The extended-release tablets should be swallowed whole and not crushed, chewed, or broken.
Dosage Forms:
- 10 mg empagliflozin / 5 mg linagliptin / 1000 mg metformin
- 12.5 mg empagliflozin / 2.5 mg linagliptin / 1000 mg metformin
- 25 mg empagliflozin / 5 mg linagliptin / 1000 mg metformin
- 5 mg empagliflozin / 2.5 mg linagliptin / 1000 mg metformin
Side Effects
Common side effects of Trijardy XR include:
- Stomach pain
- Diarrhea
- Constipation
- Bladder infection
- Headache
- Cold symptoms (runny or stuffy nose, sneezing, sore throat)
Serious side effects may include:
- Allergic reactions (hives, itching, swelling)
- Severe genital infections
- Lactic acidosis (muscle pain, trouble breathing, irregular heart rate)
- Ketoacidosis (nausea, vomiting, stomach pain)
- Pancreatitis (severe upper stomach pain)
- Dehydration (dizziness, confusion, extreme thirst)
- Signs of a bladder infection (pain or burning during urination)
- Symptoms of heart failure (shortness of breath, swelling in legs, rapid weight gain)
Warnings and Precautions
Patients should not use Trijardy XR if they have:
- Severe kidney disease or are on dialysis
- A history of severe allergic reactions to any of its components
- Metabolic acidosis or diabetic ketoacidosis
Additionally, patients should inform their doctor if they have a history of liver or kidney disease, bladder infections, pancreas disorders, heart failure, or any other significant health issues.
Conclusion
Trijardy XR is an effective medication for managing type 2 diabetes and reducing cardiovascular risks in patients with heart disease. It combines three active ingredients to provide comprehensive blood sugar control. As with any medication, it is essential to follow the prescribed dosage and be aware of potential side effects and interactions.
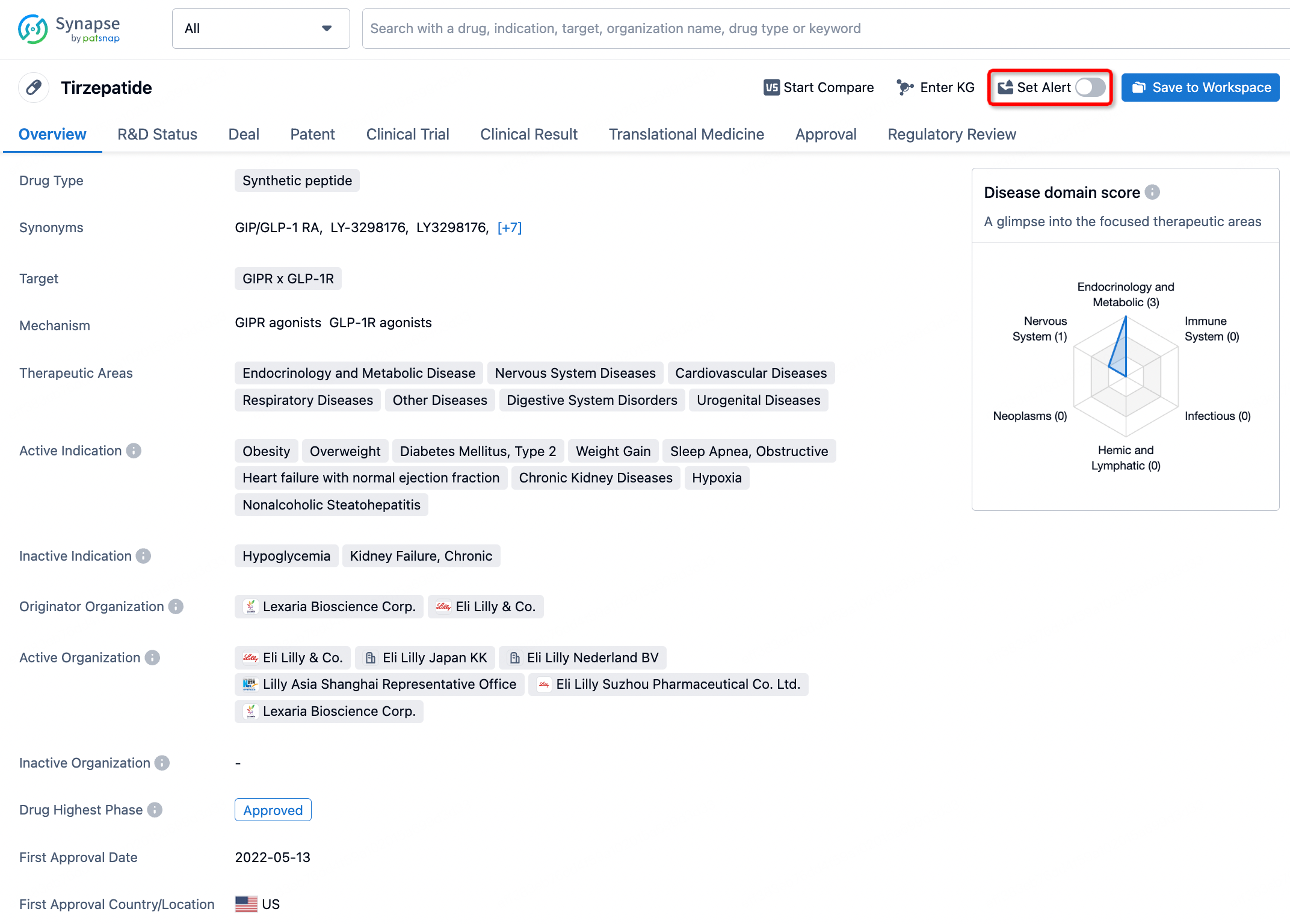
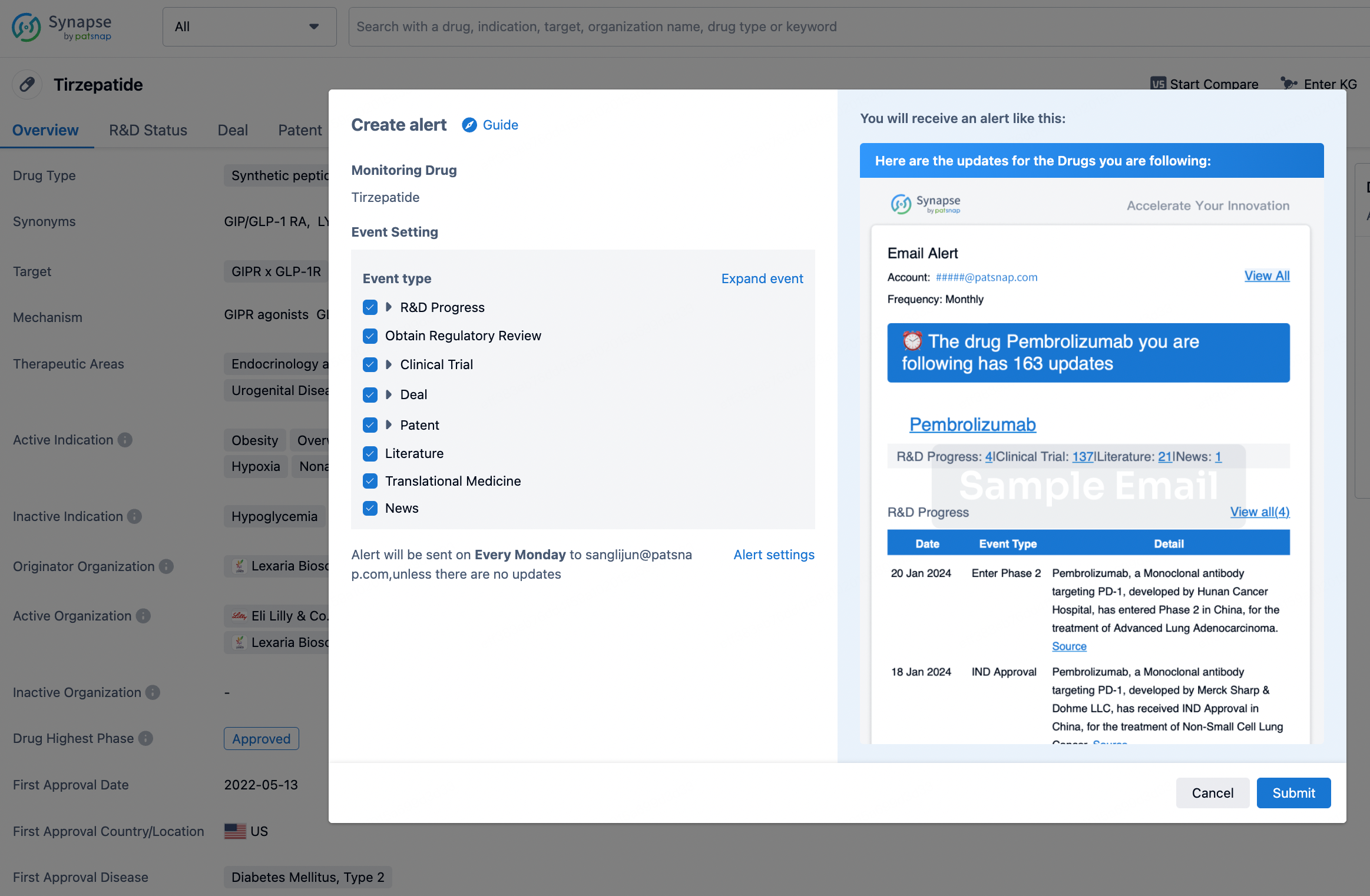
How to obtain the latest development progress of all drugs?
In the Synapse database, you can stay updated on the latest research and development advances of all drugs. This service is accessible anytime and anywhere, with updates available daily or weekly. Use the "Set Alert" function to stay informed. Click on the image below to embark on a brand new journey of drug discovery!