Request Demo
Last update 25 May 2025
BI-409306
Last update 25 May 2025
Overview
Basic Info
Drug Type Small molecule drug |
Synonyms Osoresnontrine, SUB 166499, SUB-166499 |
Target |
Action inhibitors |
Mechanism PDE9A inhibitors(Phosphodiesterase 9A inhibitors) |
Therapeutic Areas |
Active Indication- |
Inactive Indication |
Originator Organization |
Active Organization- |
Inactive Organization |
License Organization- |
Drug Highest PhaseDiscontinuedPhase 2 |
First Approval Date- |
Regulation- |
Login to view timeline
Structure/Sequence
Molecular FormulaC16H17N5O2 |
InChIKeyBZTIJCSHNVZMES-UHFFFAOYSA-N |
CAS Registry1189767-28-9 |
Related
24
Clinical Trials associated with BI-409306NCT04602221
A Randomized, Placebo Controlled, Double-blind, Double-dummy Three-way Cross Over Trial to Investigate the Effect of BI 409306, BI 425809 and Lamotrigine on Ketamine-induced Cognitive Deficits in Healthy Male Subjects
The main objective of this trail is to investigate if and to what extent BI 409306, BI 425809 and lamotrigine attenuate ketamine induced cognitive deficits.
Start Date01 Dec 2020 |
Sponsor / Collaborator |
CTR20192418
评价口服BI409306 52周对轻微精神病综合征患者的有效性、安全性和耐受性的II期随机化、双盲、安慰剂对照研究
[Translation] A Phase II Randomized, Double-Blind, Placebo-Controlled Study to Evaluate the Efficacy, Safety, and Tolerability of BI409306 Orally for 52 Weeks in Patients with Minor Psychotic Syndrome
以安慰剂作对照研究用BI 409306治疗轻微精神病综合征患者52周的有效性、安全性和耐受性。此项研究旨在证明BI 409306在实现APS缓解以及认知和功能能力改善方面优于安慰剂
[Translation]
A placebo-controlled study of the efficacy, safety, and tolerability of BI 409306 in patients with mild psychotic syndrome for 52 weeks. The study aims to demonstrate that BI 409306 is superior to placebo in achieving remission of APS and improvement in cognitive and functional abilities
Start Date29 Apr 2020 |
Sponsor / Collaborator |
NCT03934203
Thorough QT Study to Evaluate the Effects of BI 409306 as Single Dose on Cardiac Safety Parameters in Healthy Male and Female Subjects. A Randomized, Placebo Controlled, Double-blind, Five-period Crossover Study With (Open-label) Moxifloxacin as Positive Control
The primary objective of this trial is to assess the effect of BI 409306 on the QT/QTc interval in healthy male and female volunteers as measured by the QTcF change from baseline compared with placebo.
Secondary objectives are to show the assay sensitivity of the trial, by reproducing the typical effect of the positive control moxifloxacin on the QT/QTc interval, and to assess the effect of BI 409306 on heart rate.
Secondary objectives are to show the assay sensitivity of the trial, by reproducing the typical effect of the positive control moxifloxacin on the QT/QTc interval, and to assess the effect of BI 409306 on heart rate.
Start Date09 May 2019 |
Sponsor / Collaborator |
100 Clinical Results associated with BI-409306
Login to view more data
100 Translational Medicine associated with BI-409306
Login to view more data
100 Patents (Medical) associated with BI-409306
Login to view more data
17
Literatures (Medical) associated with BI-40930601 Jan 2025·SCHIZOPHRENIA RESEARCH
AI-based medication adherence prediction in patients with schizophrenia and attenuated psychotic disorders
Article
Author: Vogler, Brian ; Zhu, Zheng ; Feng, Shaolei ; Roy, Dooti
OBJECTIVE:
The capacity of machine-learning algorithms to predict medication adherence was assessed using data from AiCure, a computer vision-assisted smartphone application, which records the medication ingestion event.
METHODS:
Patients treated with BI 409306 were recruited from two Phase II randomized, placebo-controlled trials in schizophrenia (NCT03351244) and attenuated psychotic disorders (NCT03230097). A machine-learning model was optimized to predict overall trial adherence using AiCure data collected over three monitoring periods (7/10/14 days), adherence cut-offs (0.6/0.7/0.8) and timepoints (Start/Mid/End). Area under the curve (AUC), false negative rate, and false omission rate averaged across 10 model cross-validations were analyzed. In NCT03351244, post hoc analyses compared time to first relapse in patients observed as adherent versus those predicted adherent by the model.
RESULTS:
Of 235 patients, 60.4 % demonstrated ≥80 % adherence. At an adherence cut-off of 0.8, the 14-day model performed best (AUC: 0.81 versus 0.79 [10-day], 0.77 [7-day]). Within the 14-day model, 0.6 cut-off was optimal (AUC: 0.87 versus 0.85 [0.7 cut-off], 0.81 [0.8 cut-off]). The Trial-End timepoint yielded the most accurate prediction (AUC: 0.92 versus 0.87 [Start], 0.85 [Mid]). Despite NCT03351244 not meeting the primary endpoint, a reduction in risk of first relapse with BI 409306 versus placebo was observed when analyzed with adherent completers (≥80 % across trial; HR = 0.485) and patients with predicted adherence ≥60 % (HR = 0.510).
CONCLUSIONS:
Adherence data with longer monitoring durations (14 days), lower adherence cut-offs (0.6), and later timepoints (Trial-End) produced most accurate adherence predictions. Accurate adherence prediction provides insights about medication adherence patterns that may help clinicians improve individual adherence.
22 Dec 2022·Journal of medicinal chemistryQ1 · MEDICINE
BAY-7081: A Potent, Selective, and Orally Bioavailable Cyanopyridone-Based PDE9A Inhibitor
Q1 · MEDICINE
Article
Author: Schlemmer, Karl-Heinz ; Fliegner, Daniela ; Follmann, Markus ; Maassen, Stefanie ; Andreevski, Anna Lena ; Anlauf, Sonja ; Bogner, Pamela ; Meibom, Daniel ; Dieskau, André P. ; Steuber, Holger ; Micus, Sina ; Tersteegen, Adrian ; Meyer, Jutta ; Dreher, Jan ; Eitner, Frank ; von Buehler, Clemens-Jeremias ; Wunder, Frank ; Gericke, Kersten Matthias
Despite advances in the treatment of heart failure in recent years, options for patients are still limited and the disease is associated with considerable morbidity and mortality. Modulating cyclic guanosine monophosphate levels within the natriuretic peptide signaling pathway by inhibiting PDE9A has been associated with beneficial effects in preclinical heart failure models. We herein report the identification of BAY-7081, a potent, selective, and orally bioavailable PDE9A inhibitor with very good aqueous solubility starting from a high-throughput screening hit. Key aspect of the optimization was a switch in metabolism of our lead structures from glucuronidation to oxidation. The switch proved being essential for the identification of compounds with improved pharmacokinetic profiles. By studying a tool compound in a transverse aortic constriction mouse model, we were able to substantiate the relevance of PDE9A inhibition in heart diseases.
01 Oct 2022·Molecular diversity
Combined ligand-based and structure-based design of PDE 9A inhibitors against Alzheimer’s disease
Article
Author: Kumar, Ashok ; Kumar, Devendra ; Singh, Ravi ; Singh, Sushil K ; Swetha, Rayala ; Sharma, Anjali ; Ganeshpurkar, Ankit
PDE9 enzyme hydrolyzes cGMP, which is involved in the regulation of synaptic plasticity through the NMDA pathway (a well-known excitotoxic target for AD) via activation of calcium/calmodulin-dependent neuronal NO synthases in the postsynaptic neurons. The inhibition of PDE9 leads to elevated cGMP levels, causing enhanced NMDA signaling and thus contributing to an increase in synaptic plasticity and stabilization. Therefore, it could be considered a pertinent target for AD drug discovery. PF-04447943 and BI-409306 targeting PDE9 are undergoing clinical trials (Phase II). The present study encompasses a pharmacophoric approach to identify potent PDE9 inhibitors using various computational methods. Pharmacophores generated from the PDB 6A3N yielded 37,554 virtual hits, which underwent drug likeliness and PAINS filtering to arrive at a few virtual leads. The leads were further subjected to extra precision docking, ADMET predictions, and molecular dynamics. The final hits, ZINC000001305675 and ZINC000000377099, showed superior docking scores of - 10.90 and - 10.30 kcal/mol and satisfactory predicted ADMET scores. The hits were subjected to molecular dynamics (MD) studies, wherein they formed stable complexes with PDE9 protein and had ligand RMSDs within acceptable limits. The processes involved in the combined ligand and structure-based strategies.
4
News (Medical) associated with BI-40930618 Dec 2018
Rudy Tanzi, Chair of the Cure Alzheimer’s Fund Research Leadership Group and the Kennedy Professor of Neurology at Harvard University and at Massachusetts General Hospital, and Rob Fried, CEO of ChromaDex, took time to speak with BioSpace about the current understanding of Alzheimer’s and some interesting approaches to drug development.
Unfortunately, 2018 marked yet another bad year for Alzheimer’s disease drug development. There were easily half-a-dozen major clinical drug trial failures, leading one analyst to call Alzheimer’s drug development “an unrelenting disaster zone.” These failures included:
Takeda Pharmaceutical
and
Zinfandel Pharmaceuticals
abandoned
their five-year Phase III TOMORROW trial after an interim analysis of pioglitazone in mild cognitive impairment due to Alzheimer’s disease;
Boehringer Ingelheim
indicated
it was abandoning its Phase II compound BI 409306 after it failed to meet its endpoints;
Merck & Co.
said
it was halting protocol 019, its APECS Phase III clinical trial of verubecestat (MK-8931) in Alzheimer’s after an external Data Monitoring Committee (eDMC) recommended ending it after an interim safety analysis;
vTv Therapeutics
announced
its azeliragon failed to meet either co-primary efficacy endpoint in its Phase III STEADFAST clinical trial in patients with mild Alzheimer’s disease, and several others.
Most of the Alzheimer’s drugs that have failed so far have focused on halting the accumulation of amyloid plaques, or in clearing those plaques. Even when the drugs are successful in doing so, however, they have not improved cognition and memory issues. This has led critics to question if the amyloid theory of Alzheimer’s is, in fact, the wrong path.
With all that gloom-and-doom, however, high-level research is ongoing, looking at preventing amyloid accumulation earlier and earlier in the disease and examining other aspects of the disease that are implicated in the cognition and memory problems.
Rudy Tanzi
, Chair of the
Cure Alzheimer’s Fund Research Leadership Group
and the Kennedy Professor of Neurology at
Harvard University
and at
Massachusetts General Hospital
, and
Rob Fried
, chief executive officer of
ChromaDex
, took time to speak with
BioSpace
about the current understanding of Alzheimer’s and some interesting approaches to drug development. Tanzi was quick to note that he is both a shareholder and a member of ChromaDex’s Scientific Advisory Board.
Rudy Tanzi, Chair of the Cure Alzheimer’s Fund Research Leadership Group and the Kennedy Professor of Neurology at Harvard University and at Massachusetts General Hospital
First off, Tanzi discussed the current thinking behind Alzheimer’s disease. And contrary to the critics, he doesn’t believe the amyloid theory is wrong at all. “I think amyloid and tangles trigger the disease,” he says, “but they’re not sufficient to cause dementia. In a nutshell, what we’ve learned is that amyloid comes very early, 15 years before symptoms. And all the genetics tells us this disease begins with amyloid.”
Once amyloid begins to form, the tangles themselves will kill some neurons, but not enough to create dementia. But the amyloid and tangle-driven neuronal cell death eventually hits a point where the brain’s innate immune system reacts with significant levels of neuroinflammation. “Then, exponentially more cell death occurs, which leads to symptoms of dementia and Alzheimer’s disease,” Tanzi says.
Tanzi compares the amyloid to a match and the tangles to a brush fire. “You can live with them,” he says, “but once the brain reacts to neuroinflammation, that’s like the forest fire. That’s when you really lose enough neurons to go downhill and become dementia. The failure of the amyloid-targeted AD trials is like trying to put out a raging forest fire by blowing out the match.”
Which, broadly speaking, is where ChromaDex comes in. Founded about 20 years ago as an analytical testing business, it later added a reference standards business and ultimately became an ingredients business. They developed a portfolio of five ingredients, all of which are interesting, but one, in particular, is especially so. That product is Niagen (nicotinamide riboside or NR).
NR is a member of the vitamin B3 family. Cells in the body use NR to create nicotinamide adenine dinucleotide (NAD+), which is essential to cellular energy generation and metabolism.
One of the results of aging is a decline of NAD. It plays significant roles in cellular metabolism, energy production, maintaining healthy mitochondria, and promotion of cellular repair.
Rob Fried, chief executive officer of ChromaDex
ChromaDex is a bit unusual as a biotech company. Fried refers to it as a “consumer biotech company. It has very strong science, but its commercialization strategy is in the consumer realm. There are still some ingredients in place to other consumer-facing companies to whom we supply our ingredients—most notably Niagen.”
So far, there are four published clinical trials involving Niagen with 28 listed on clinicaltrials.gov. Most of those trials are in the neurological field, with some in cardiovascular, and others addressing obesity.
Fried says, “Specifically, the focus is on cell repair. PARP (poly ADP ribose polymerase) requires NAD. And PARP is involved in oxidative stress and correcting DNA mutations. NAD itself is vital to many repair functions. There are studies ongoing specifically related to inflammation.”
Tanzi compares cells to batteries. “As we get older, our energy does down. It’s like a computer battery. The older it gets, the more the battery goes down and the harder it is to charge it.”
The brain is very energy-dependent. When the mitochondria age and stop using fuel like ATP, they are believed to become vulnerable. Microglial cells are specialized immune cells found in the central nervous system. They remove damaged and debris, like amyloid, to maintain the health of the central nervous system. But they also fight infections and react to neuronal cell death by triggering neuroinflammation. This leads to massive neuronal cell death, essentially via “friendly fire.” Tanzi notes, “A microglial cell is more likely to be neuroinflammatory if it is lacking in energy and, thus, less resilient.”
He cites a study published in the journal
Cell
that described increasing ATP levels in a microglial cell that carried a mutant version of one of the Alzheimer’s genes known as TREM2. “If you increase ATP levels, it continues doing a better job of clearing beta-amyloid. And it’s less likely to become neuroinflammatory. So when we talk about resilience of cells, it’s often about how much energy the cell has. As we get older, we have less cellular energy which has a lot to do with ATP and mitochondria. NAD levels drop as we get older, so if you supplement it, levels go back up, which starts to restore cellular energy, which has implications for Alzheimer’s disease and neurocellular resilience.” Likewise, a neuron with more energy is less likely to form a tangle in the presence of neuroinflammation, adds Tanzi.
In addition to the various other ongoing clinical trials involving Niagen and NAD, Tanzi and colleague
Steve Arnold
at MGH, along with ChromaDex, have designed a clinical trial to evaluate Niagen in Alzheimer’s disease. They are still organizing funding, including discussions with the Cure Alzheimer’s Fund, but the trial is fully designed. Tanzi notes that as a shareholder in ChromaDex, he can help design the trial, but would be precluded as a professor at Harvard Medical School and MGH from participating in the trial itself.
When asked about the Cure Alzheimer’s Fund, Tanzi says it started approximately 15 years ago, which initially funded one of the first whole gene sequencing for Alzheimer’s genes, which led to the discovery of the first inflammatory gene, CD33. Cure Alzheimer’s Fund currently funds more than 75 investigations, giving out about $20 million per year.
“One of the cooler things,” Tanzi adds, “is the people who founded it, to this day, pay all the overhead. If somebody were to give one dollar, one dollar goes to research. They’re only invested in research. They don’t do other important things like outreach or dealing with families, just hard-core, state-of-the-art, cutting-edge research around the world.”
Phase 3Phase 2Oligonucleotide
14 Dec 2018
In writing literally hundreds of stories this year, two BioSpace writers, Alex Keown and Mark Terry, found certain stories particularly intriguing or impactful. Some of those were such big topics that they were covered over a series of stories. Looking back at 2018, here are their Top 10.
In writing literally hundreds of stories this year, two
BioSpace
writers, Alex Keown and Mark Terry, found certain stories particularly intriguing or impactful. Some of those were such big topics that they were covered over a series of stories. Looking back at 2018, here are their Top 10.
#1. CRISPR Babies.
He Jiankui
, a researcher from the
Southern University of Science and Technology of China
announced
that a set of twins had been born in which he had used CRISPR gene editing to modify the embryos. He made this announcement at the
Second International Summit on Human Genome Editing
held at the
University of Hong Kong.
He Jiankui modified the CCR5 gene, which should make the babies less susceptible to HIV infection.
Another announcement He Jiankui made before leaving the conference to return to China was that there was another successful pregnancy. This has resulted in the widespread condemnation, calls for global guidelines, and the launch of numerous investigations, including by the China government and the U.S.
National Institutes of Health
(NIH).
A group of more than 100 Chinese scientists co-signed a letter condemning it, calling it “crazy.” Other ethicists have called it “monstrous,” “unconscionable” and “a grave abuse of human rights.”
#2. Takeda-Shire Deal.
Although it was probably not in serious peril, shareholders of Japan’s
Takeda Pharmaceutical
voted
to approve the acquisition of Dublin’s
Shire
in December. The deal is valued at $58 billion, the biggest single deal of the year.
The
deal
came together in May 2018, with Takeda’s final official bid coming in at around $62.2 billion. Takeda made five public bids for the company. The deal came in at $30.33 per share in cash and 0.839 shares of Takeda stock. Currency fluctuations and Takeda’s share price drop brought the value down to about $58 billion.
Takeda is gaining both Shire’s portfolio, heavy on rare disease drugs, and a stronger U.S. market presence. It’s also picking up a significant amount of debt, which pushes the deal closer to a total of $80 billion. And it is largely this debt that caused some problems leading up to today’s approval. As part of the final deal, Takeda has secured $30.9 billion in bank loans.
A fairly small but vocal
opposition
group of Takeda
shareholders
calling itself “Think About Takeda’s Bright Future” or TTBF, made several attempts to scuttle the deal. At the June annual meeting, the group, which at the time had about 130 shareholders in it, expressed their opposition and advanced a proposal that would require advance shareholder approval be required for large acquisitions. The proposal, however, only received about 10 percent of votes in favor, failing to pick up wider opposition support.
#3. Biotech IPOs and Moderna.
It was a record year for biotech initial public offerings (IPOs). Some were individual record-breakers—
Moderna Therapeutics
,
Allogene Therapeutics
and
Rubius Therapeutics
. The total for the year is around $8.2 billion raised, breaking 2014’s record of $6.5 billion.
Moderna sold approximately 26.3 million
shares
priced at $23 a share. This exceeded the revised goal of $600 million by about $4.3 million.
The previous
record
was set two months ago by Allogene Therapeutics, when it raised $324 million at a $2.2 billion market cap. In July, Rubius Therapeutics’ IPO
brought
in $241 million.
#4. Alzheimer’s Failures.
Sadly, 2018 marked yet another bad year for Alzheimer’s disease drug development. Here are just some of the year’s biggest failures in this area. On January 25, 2018, Takeda Pharmaceutical
and its development partner
Zinfandel Pharmaceuticals
gave up
on their five-year Phase III TOMORROW trial after an interim analysis of pioglitazone in mild cognitive impairment due to Alzheimer’s disease. The safety was generally considered fine, but there was inadequate treatment effect.
On February 9, 2018,
Boehringer Ingelheim
indicated
it was abandoning its Phase II compound BI 409306 after it failed to meet its endpoints. The drug, a PDE9 inhibitor, being used to treat patients with cognitive impairment and memory dysfunction in schizophrenia and Alzheimer’s, failed to show superiority over placebo in cognition in two separate clinical trials. On February 14,
Merck & Company
said
it was halting protocol 019, its APECS Phase III clinical trial of verubecestat (MK-8931) in Alzheimer’s. An external Data Monitoring Committee (eDMC) had recommended ending it after an interim safety analysis, saying the likelihood of benefits didn’t outweigh the risks.
On April 10,
vTv Therapeutics
announced
its azeliragon failed to meet either co-primary efficacy endpoint in its Phase III STEADFAST clinical trial in patients with mild Alzheimer’s disease. On May 18,
Johnson & Johnson
’s
Janssen
division
halted
its clinical trials of atabacestat, a BACE inhibitor, for Alzheimer’s disease. It was ending the program over safety issues, rather than efficacy. And on September 18,
Allergan
and
Sosei
voluntarily
suspended
clinical trials of HTL0018318 in Alzheimer’s and other types of dementia because of safety issues.
#5. Cancer’s Silver Bullet.
Researchers from the
University of Queensland
’s Australian Institute for Bioengineering and Nanotechnology (AIBN)
identified
a nano-scaled DNA
signature
that seems to be common to all cancers. This has the potential to be used as an early cancer detection test, what is often called a liquid biopsy, and if the research pans out, could open up new pathways for universal cancer treatments.
Methyl groups are spread across the genome and are implicated in turning genes on and off. The AIBN team found that cancer cells’ genomes often lack methyl groups except for “intense clusters of methyl groups at very specific locations.” The researchers dubbed this “methylscape,” and they observed it in every type of breast cancer they studied, as well as in other cancer types, including prostate cancer, colorectal cancer and lymphoma.
“Virtually every piece of cancer DNA we examined had this highly predictable pattern,” stated Trau. “It seems to be a general feature for all cancer. It’s a startling discovery.”
#6.
Theranos Shuts Down for Good
. Nearly three years after the
Wall Street Journal
first
raised questions about the single-drop blood-testing device
Theranos
claimed would revolutionize the industry, the company
closed its doors for good
, battered by lawsuits from consumers, as well as the federal government. At one time the high-flying unicorn had a valuation of $9 billion, but after multiple reports revealed the company’s products did not work, the money it so easily raised from Silicon Valley investors began to dry up. Company founder Elizabeth Holmes and former company president Sunny Balwani also face
criminal fraud charges
that allege the two engaged in a multi-million dollar scheme to defraud investors, and a separate scheme to defraud doctors and patients. Both Balwani and Holmes have entered not guilty pleas to the criminal charges. Holmes agreed to
pay a $500,000 fine for fraud
to the U.S. Securities and Exchange Commission for earlier charges brought against the two. Balwani is fighting the charges brought against him.
The space here doesn’t truly allow for a full re-telling of the Theranos saga, but the story could soon be on the silver screen with Academy Award-winning Jennifer Lawrence set to star in the film as Holmes. The story is reportedly based off of “Bad Blood,” the book written by
The Wall Street Journal’s
Pulitzer Prize-winning investigative journalist John Carreyrou.
#7
.
Stressing Gender Diversity
. In October,
Biotechnology Innovation Organization (BIO)
called on its member companies to achieve gender diversity on its boards of directors within the next six years.
BIO called for its members to increase female representation
at the senior management and leadership level to 50 percent by 2025. For boards of directors, biotech’s biggest trade association called for 30 percent female representation. The call for greater representation came in the wake of other groups within the industry making calls for greater gender diversity.
BIO made the move after a second industry party was held during its annual convention over the summer that was deemed disrespectful to women. The annual
Party at Bio Not Associated with Bio (PABNAB)
included topless dancers who were covered in body-paint that featured the logos of some sponsoring companies. That was preceded by the now
infamous 2016 party
at the J.P. Morgan Healthcare Conference in San Francisco.
Others have also called for more diversity. LifeSci Advisors has been pushing a board training program aimed at female executives. David Lucchino, CEO of Woburn, Mass.-based
Frequency Therapeutics
,
called for greater diversity
prior to being named chairman of MassBio (Massachusetts Biotechnology Council).
#8.
The Opioid Crisis Continues
. Opioid-based painkillers have become the poster-child for abuse. Abuse of the drugs has been linked to more than 70,000 deaths annually for the past several years. Over the course of 2018,
BioSpace has highlighted a number of lawsuits
that have been filed against the opioid-makers by state and local governments. The governments are seeking financial reparations from the companies for the public funds they have spent combatting opioid abuse. The lawsuits almost universally allege the marketing practices used by the opioid drugmakers.
Despite the
number of lawsuits
and the increased awareness of the level of addiction across the country, in November the
Food and Drug Administration (FDA)
approved a powerful new opioid painkiller called Dsuvia that is reportedly 1,000 times more powerful than morphine. Following an outcry about the approval amid the crisis, FDA Commissioner Scott Gottlieb took to Twitter to defend the decision.
Gottlieb argued the FDA should consider
the approval of new opioid pain medications that can help fill targeted medical needs.
#9.
Retractions Rock the Industry
. A former
Harvard
scientist’s name fell into disgrace earlier this year after concerns of falsified data were attached to 31 articles he published in various medical journals. Dr. Piero Anversa was a pioneer in the field of cardiac research. However, some articles that he published in the area of cardiac stem cells have been called into question. Anversa’s work centered on the idea that the heart contains stem cells that could regenerate cardiac muscle. The team claimed that it had identified cells known as c-kit cells that were responsible for that regeneration. Although various teams have tried, the results of Anversa’s work could not be reproduced. As a result,
31 articles he authored have been retracted
. There is a growing concern that Anversa’s work in the Harvard lab was fabricated. As a result of the questions over Anversa’s work, Harvard and
Brigham and Women’s Hospital
agreed to pay a $10 million settlement over allegations of research misconduct involving Anversa and members of his team. Anversa, along with Dr. Annarosa Leri and Dr. Jan Kajstura, used that falsified data to secure funding from the
National Institutes of Health (NIH)
. Anversa’s lab at Harvard was shuttered in 2015 and none of the researchers currently work for either institution.
#10
.
Oh, llama
. The flu is no joke. Most people are rocked by it in one way or another each year. This year saw the first new flu treatment approved in 20 years by the FDA after
Roche
snagged
regulatory approval for Xofluza
. But, this year also marked a new source for a potential flu vaccine that can work on a wide number of flu viruses. Researchers from
The Scripps Research Institute
were able to
harness antibodies from the saliva of llamas
and use them as a base for a flu vaccine. The antibodies are diverse camelid single-domain antibodies were used to generate multidomain antibodies with “impressive breadth and potency.” In the study, the researchers used the multidomain antibody MD3606 in mice. They found that the antibody protected the mice against influenza A and B infection when administered intravenously or expressed locally from a recombinant adeno-associated virus vector.
MD3606 could provide protection against most strains of the flu, the researchers said in their abstract. That would be true even if a strain of flu were to change. However, before investing in llama farms as a potential pharma cash cow, the vaccine has not run through human trials yet and it will be years before this research pans out for a potential marketed vaccine.
AcquisitionPhase 3VaccineDrug Approval
03 Dec 2018
The most commonly stated statistic about clinical trials is that for every one drug on the market, there are nine that failed. A look back through 2018 indicates there were a great many successes, but plenty of challenges as well. Here’s a look at 10 notable drug challenges of the year.
The most commonly stated statistic about clinical trials is that for every one drug on the market, there are nine that failed. A look back through 2018 indicates there were a great many successes, but plenty of challenges as well. Here’s a look at 10 notable drug challenges of the year.
#1. Takeda and Zinfandel and Alzheimer’s.
On January 25, 2018,
Takeda Pharmaceutical
and its development partner
Zinfandel Pharmaceuticals
gave up
on their five-year Phase III TOMORROW trial after an interim analysis. They were evaluating pioglitazone in mild cognitive impairment due to Alzheimer’s disease. The TOMMORROW trial was evaluating the genetic-based biomarker risk assignment algorithm (BRAA) in addition to the safety and efficacy of the drug. The safety was generally considered fine, but there was inadequate treatment effect.
#2. Boehringer Ingelheim and Alzheimer’s.
Unfortunately, Alzheimer’s failures are the norm, rather than the exception. On February 9, 2018,
Boehringer Ingelheim
indicated
it was abandoning its Phase II compound BI 409306 after it failed to meet its endpoints. The drug, a PDE9 inhibitor, being used to treat patients with cognitive impairment and memory dysfunction in schizophrenia and Alzheimer’s, failed to show superiority over placebo in cognition in two separate clinical trials.
#3. Merck & Co. and Alzheimer’s.
Yes, well, it does seem like a theme. On February 14,
Merck & Co.
said
it was halting protocol 019, its APECS Phase III clinical trial of verubecestat (MK-8931) in Alzheimer’s. An external Data Monitoring Committee (eDMC) had recommended ending it after an interim safety analysis, saying the likelihood of benefits didn’t outweigh the risks. Verubecestat is a BACE inhibitor, a precursor to amyloid-beta.
#4. Celgene and Multiple Sclerosis (MS).
This particular failure was a bit of a surprise. On February 28, the
U.S. Food and Drug Administration (FDA)
hit
Celgene
with a Refusal to File letter over its New Drug Application (NDA) for multiple sclerosis treatment ozanimod. What was surprising was the Refusal to File was over the “nonclinical and clinical pharmacology sections” in the NDA, claiming they were insufficient for the agency to go ahead with the approval process. This was more than a bit unexpected for a company with the reputation and size of Celgene, suggesting that the application wasn’t really ready to go to the agency.
Several months later, in June, the company sort of admitted it was their fault, although at the same time, the company’s president of hematology and oncology,
Nadim Ahmed
, seemed to
throw
their
Receptos
unit under the bus, blaming them for the failure. Celgene acquired Receptos in 2015. The NDA for ozanimod was submitted toward the end of 2017, after it reported positive data from the second pivotal Phase III trial, RADIANCE, which was announced in May 2017.
On October 10, Celgene
reported
positive data from two post hoc analysis from the Phase III SUNBEAM and RADIANCE Part B trials, evaluating ozanimod in MS. The company
indicates
it plans to in 2019.
#5. Incyte and Melanoma
. On April 6,
Incyte Corporation
revealed
that its IDO1 drug in combination with Merck’s Keytruda did not meet its endpoints in a Phase III melanoma trial. The drug, epacadostat, did not improve rates of progression-free survival in the patient population compared to Keytruda alone.
#6. vTV Therapeutics and Alzheimer’s.
On April 10,
vTv Therapeutics
announced
its azeliragon failed to meet either co-primary efficacy endpoint in its Phase III STEADFAST clinical trial in patients with mild Alzheimer’s disease. It was made up of two independent, but identical double-blind, placebo-controlled trials, Part A and B. The group in Part A that received the drug showed a 4.4-point decline from baseline in the Alzheimer’s Disease Assessment Scale-cognitive subscale (ADAS-cog) and a 1.6-point decline from baseline in the Clinical Dementia Rating Scale Sum of Boxes (CDR-sb) compared to declines in the placebo groups of 3.3 and 1.6, respectively. They were not clinically significant changes. Azeliragon is an orally active small-molecule antagonist of advanced glycation endproducts (RAGE).
#7. Roche and Exelixis and Colorectal Cancer.
On April 11, Roche temporarily
halted
recruiting patients to its Phase II MODUL clinical trial for metastatic colorectal cancer after four patient deaths. The patients were receiving Tecentriq (atezolizumab) in combination with Exelixis’ MEK inhibitor Cotellic (cobimetinib). Then, in mid-May, the two companies’ IMblaze370 Phase III clinical trial of Tecentriq and Cotellic
failed
in a comparison to
Bayer
’s Stivarga (regorafenib) in colorectal cancer patients who had failed at least two rounds of chemotherapy.
#8. Johnson & Johnson and Alzheimer’s.
On May 18,
Johnson & Johnson’s Janssen
division
halted
its clinical trials of atabacestat, a BACE inhibitor, for Alzheimer’s disease. It was ending the program over safety issues, rather than efficacy. Patients in the EARLY Phase IIb/III clinical trial with preclinical Alzheimer’s disease, as well as a Phase II long-term safety trial, had elevated liver enzymes. EARLY launched in 2015 and was scheduled to wrap in 2024.
#9. Sarepta Therapeutics and Duchenne Muscular Dystrophy (DMD).
On September 21, the
European Medicines Agency (EMA)
rejected
Sarepta’s application for Exondys 51 for DMD. What is most notable about this was that in September 2016, the FDA approved the drug after a contentious and dramatic battle that involved internal conflict at the agency, media coverage and members of Congress and panels of DMD experts sending public letters urging approval. The application to the EMA was based on two clinical trials in 12 boys with DMD between the ages of 7 and 13.
The European agency, in its negative opinion by the Committee for Medicinal Products for Human Use (CHMP), expressed doubts about the evidence, especially related to the studies’ small size, use of historical data, and lack of comparison data beyond 24 weeks. These were essentially the same controversies within the FDA.
Meanwhile, Exondys, approved in the U.S., has a yearly price tag, based on the patient’s body weight, of approximately $300,000.
#10. Alkermes and Depression—Stay Tuned!
On November 2, two FDA advisory committees
voted
down the company’s NDA for ALKS-5461 for depression. In total, the committees voted 21 to 2 against recommending the drug. They also voted 20 to 3 that the company had not provided substantial evidence that supports the efficacy of ALKS-5461.
One of the committee members,
Martin Kulldorff
, a professor of population medicine and a biostatistician at
Harvard Medical School
, said, “I don’t think there’s evidence that this drug works.”
The drug has had a tumultuous history. The FDA accepted the NDA in April only a few weeks after initially rejecting it due to “insufficient evidence of overall effectiveness for the proposed indication.” In its Refusal to File letter, the FDA said the company would likely have to launch additional clinical trials in order to support a resubmission. But then the FDA changed its mind and Alkermes said it did not submit any additional data to the agency as was originally suggested.
The FDA is not required to follow the recommendations of its advisory committees, although with such a strong rejection by two Adcoms, it would be surprising if it did approve the drug. The agency has a target action date of January 31, 2019 for approval.
Phase 2Phase 3NDAClinical ResultDrug Approval
100 Deals associated with BI-409306
Login to view more data
External Link
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| - | - | - |
R&D Status
10 top R&D records. to view more data
Login
| Indication | Highest Phase | Country/Location | Organization | Date |
|---|---|---|---|---|
| Psychotic Disorders | Phase 2 | United States | 18 Oct 2017 | |
| Psychotic Disorders | Phase 2 | Canada | 18 Oct 2017 | |
| Psychotic Disorders | Phase 2 | United Kingdom | 18 Oct 2017 | |
| Behavioural disorders | Phase 2 | China | 29 Sep 2017 | |
| Behavioural disorders | Phase 2 | United Kingdom | 29 Sep 2017 | |
| Cognitive Dysfunction | Phase 2 | United States | 21 Jan 2015 | |
| Cognitive Dysfunction | Phase 2 | Austria | 21 Jan 2015 | |
| Cognitive Dysfunction | Phase 2 | Belgium | 21 Jan 2015 | |
| Cognitive Dysfunction | Phase 2 | Canada | 21 Jan 2015 | |
| Cognitive Dysfunction | Phase 2 | France | 21 Jan 2015 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
| Study | Phase | Population | Analyzed Enrollment | Group | Results | Evaluation | Publication Date |
|---|
Phase 1 | - | 20 | (BI 409306 50 mg Under Resting Conditions) | ttbzlzrsuf(xmxvpjjckq) = egsahaovan wamzapdzdv (wivpjrvqhj, surzwqpafc - teiwxmiajs) View more | - | 03 Sep 2024 | |
(BI 409306 200 mg Under Resting Conditions) | ttbzlzrsuf(xmxvpjjckq) = zrhlqgfkho wamzapdzdv (wivpjrvqhj, nfqagumzrf - lisfxafefj) View more | ||||||
Phase 1 | - | 18 | (10 mg BI 409306 + 100 mg Fluvoxamine (T2)) | ydxgxlpsao(hflvtlldhy) = syrubzjipr jxngdkusgc (ojxekxxvrq, NA) View more | - | 21 Aug 2024 | |
BI 409306 (R2) (10 mg BI 409306 (R2)) | ydxgxlpsao(hflvtlldhy) = slerkvshrf jxngdkusgc (ojxekxxvrq, NA) View more | ||||||
Phase 1 | - | 9 | vwufvnhhys(xcwtanbzoc) = zovzldmuhu ggbeqricye (buusetkuic, 54.0) View more | - | 20 Aug 2024 | ||
Phase 1 | 61 | Placebo matching BI 409306 50 mg+BI 409306 25 mg (BI 409306 25 Milligram (mg) - Alzheimer Patients) | xvpbdyhntd = enxobryquy ngrmcpodla (zpjlinlqei, bgrsrqzmzk - xisnvyqckf) View more | - | 19 Apr 2024 | ||
Placebo matching BI 409306 25 mg+BI 409306 50 mg (BI 409306 100 mg - Alzheimer Patients) | xvpbdyhntd = rbrkostbrq ngrmcpodla (zpjlinlqei, ukarsvbmxk - detptrpwtw) View more | ||||||
Phase 1 | - | 20 | (BI 409306 25mg) | wlobzvmcis(roiqbhwwea) = faofkhxrqs ymfnajibtm (gbcojadkdc, 18.40) View more | - | 21 Mar 2024 | |
(BI 409306 50 mg) | wlobzvmcis(roiqbhwwea) = jzgqlvqamd ymfnajibtm (gbcojadkdc, 15.80) View more | ||||||
Phase 1 | - | 16 | (BI 409306 + Microgynon® 30 (T1)) | qclbaatcra(abreuurwuu) = rfkqsrlrhx wdwajincbo (uajminivms, NA) View more | - | 13 Mar 2024 | |
(BI 409306 Alone (R1)) | qclbaatcra(abreuurwuu) = vfipfdvaqt wdwajincbo (uajminivms, NA) View more | ||||||
Phase 1 | - | 40 | Placebo (Placebo (Young Subjects)) | rlrvhejoei = bvjmxzqseq ibppcnpgvf (qafabsaaae, cfzuzvhaao - mzhlvsejyw) View more | - | 08 Mar 2024 | |
(BI 409306 - 25 Milligram (mg) (Young Subjects)) | rlrvhejoei = jtfznqqmkb ibppcnpgvf (qafabsaaae, xxmbjgdmbq - vjvuvidztd) View more | ||||||
Phase 1 | 40 | Placebo | vktedivooh = blrwxfdmgy hofmjpqlru (qxmaeexncz, kkohcpyeid - jglxloolds) View more | - | 08 Mar 2024 |
Login to view more data
Translational Medicine
Boost your research with our translational medicine data.
login
or
Deal
Boost your decision using our deal data.
login
or
Core Patent
Boost your research with our Core Patent data.
login
or
Clinical Trial
Identify the latest clinical trials across global registries.
login
or
Approval
Accelerate your research with the latest regulatory approval information.
login
or

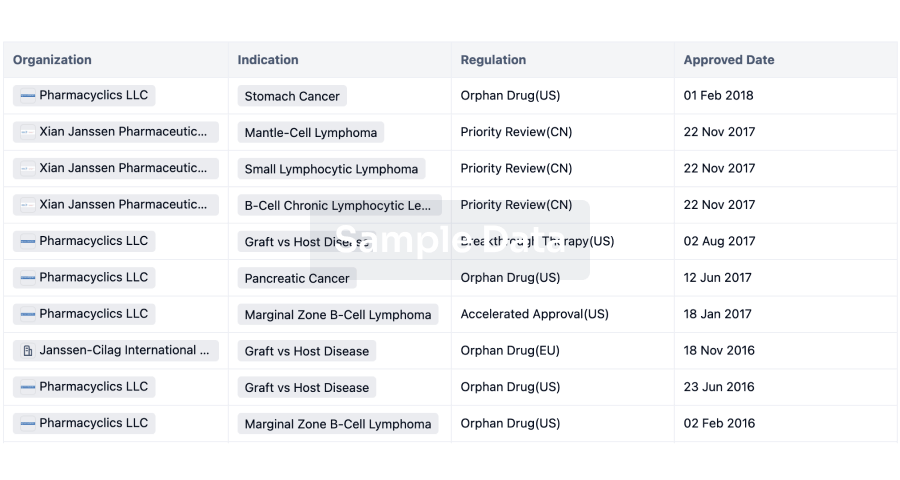
Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or
AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free
