Request Demo
Last update 25 Feb 2026
Salicylic acid
Last update 25 Feb 2026
Overview
Basic Info
SummarySalicylic acid serves as a precursor to and a metabolite of aspirin, earning it some clout in the pharmaceutical world. It is frequently employed as a topical medication, particularly due to its ability to expel the external layer of skin, which essentially means that it can treat an assortment of skin conditions. |
Drug Type Small molecule drug |
Synonyms 2-Carboxyphenol, 2-Hydroxybenzoic acid, O-carboxyphenol + [11] |
Target |
Action inhibitors |
Mechanism COX inhibitors(Cyclooxygenases inhibitors) |
Therapeutic Areas |
Active Indication |
Inactive Indication- |
Originator Organization |
Active Organization |
Inactive Organization |
License Organization- |
Drug Highest PhaseApproved |
First Approval Date Japan (01 Oct 1950), |
Regulation- |
Login to view timeline
Structure/Sequence
Molecular FormulaC7H6O3 |
InChIKeyYGSDEFSMJLZEOE-UHFFFAOYSA-N |
CAS Registry69-72-7 |
Related
19
Clinical Trials associated with Salicylic acidCTRI/2024/07/071474
A Clinical Study to Evaluate the Skin Irritation Potential of Mosaic Wellness Hyaluronic Acid Body Wash Mousse and 2percent Salicylic Acid Cleansing Balm on Healthy Human Subjects - NIL
Start Date09 Aug 2024 |
Sponsor / Collaborator |
CTRI/2024/02/062468
Open Randomized Controlled Trial On Erand Patranal Kshar On Mashak With Special Reference to Wart - Nil
Start Date13 Feb 2024 |
Sponsor / Collaborator- |
NCT06969300
Efficacy and Safety Evaluation of Fractional Microneedling Radiofrequency Combined With 30% Supramolecular Salicylic Acid in the Treatment of Refractory Erythrotelangiectatic Rosacea
This study was a single-center, case-by-case, bilateral facial comparison study. A total of 15 patients were included. On the day of enrollment, after the completion of the test items, treatment was carried out (full face gold micro-needling + 30% salicylic acid on the half face), and on-site follow-up was conducted 1 week later, 2 weeks later follow-up was conducted and the second acid brushing treatment was performed, 4 weeks later follow-up was conducted and the third acid brushing treatment was performed, on-site follow-up was conducted at the 8th week and the 12th week, and the follow-up contents included: Visia photography, camera photography, non-invasive skin testing, questionnaire assessment, etc. The study duration was 12 weeks.
Start Date01 Dec 2023 |
Sponsor / Collaborator |
100 Clinical Results associated with Salicylic acid
Login to view more data
100 Translational Medicine associated with Salicylic acid
Login to view more data
100 Patents (Medical) associated with Salicylic acid
Login to view more data
35,925
Literatures (Medical) associated with Salicylic acid31 Dec 2026·JOURNAL OF DERMATOLOGICAL TREATMENT
Flumethasone-salicylic acid cream effectively flattened verrucous epidermal nevus: a case report
Article
Author: Li, Wei ; Liu, Wenyan ; Ye, Hui ; Zhang, Xibao ; Jiang, Yu ; Liang, Jingyao ; Deng, Shilin ; Li, Qian
OBJECTIVES:
To assess the efficacy and safety of topical compound flumethasone pivalate-salicylic acid cream for verrucous epidermal nevus (VEN), a benign keratinocytic hamartoma with limited current treatment options.
METHODS:
A 25-year-old male with 20-year VEN (right buttock/lower limb plaques) was treated with the compound cream (0.2 mg flumethasone pivalate + 30 mg salicylic acid/gram) twice daily for over 2 months, followed by 14-week follow-up.
RESULTS:
Lesions showed progressive thinning, significantly reduced hyperpigmentation, and no adverse events. The Dermatology Life Quality Index score improved from 13 to 3, with no recurrence at follow-up.
CONCLUSIONS:
Topical compound flumethasone pivalate-salicylic acid cream is effective and safe for VEN, potentially via inhibiting abnormal keratinocyte proliferation, serving as a practical topical option.
01 May 2026·TALANTA
Capillary blood microsampling and LC–Orbitrap analysis for adherence monitoring of cardiovascular drugs: method development, cross-validation, and patient proof-of-concept using VAMS and Capitainer-B
Article
Author: Meyer, Markus R ; Böhm, Michael ; Wagmann, Lea ; Mahfoud, Felix ; Markwirth, Philipp ; Kretschmer, Diana ; Tokcan, Mert
Microsampling enables decentralized adherence monitoring by capturing defined volumes of capillary blood. We developed, validated, and cross-evaluated a volumetric absorptive microsampling (VAMS) and Capitainer-B workflow for 15 cardiovascular drugs using high-resolution liquid chromatography Orbitrap analysis. Amlodipine, atenolol, atorvastatin, bisoprolol, carvedilol, clopidogrel, diltiazem, lercanidipine, metoprolol, nebivolol, prasugrel, rosuvastatin, salicylic acid, simvastatin hydroxy acid, and verapamil were included. Sampling by VAMS and Capitainer-B should be evaluated and compared. VAMS tips and Capitainer-B disks loaded with 10 μL whole blood were extracted with 200 μL methanol, shaken and centrifuged, evaporated to dryness, reconstituted, and injected for analysis. The method met international validation criteria for most analytes, with quantification and selectivity achieved for all except prasugrel. Capitainer-B generally exhibited higher matrix effects than VAMS, while accuracy and precision were within acceptance limits except for lercanidipine across both devices and for atorvastatin on Capitainer-B. Under long-term storage, VAMS showed >15 % loss for carvedilol, lercanidipine, and simvastatin hydroxy acid after two weeks, whereas Capitainer-B showed no degradation. In a proof-of-concept involving 30 patients, finger-prick concentrations from VAMS and Capitainer-B were interchangeable, supporting the suitability of these adherence workflows but reference ranges for capillary blood remain necessary to enable routine clinical interpretation. In summary, this work established a reliable, LC-Orbitrap-based microsampling platform for monitoring adherence and identified stability issued amongst others that guide future method selection and development.
01 May 2026·JOURNAL OF PHARMACEUTICAL AND BIOMEDICAL ANALYSIS
Integrated dual-molecular marker analysis with SSR and UPLC-Q-TOF/MS fingerprints technology reveal the interrelation of the molecule-metabolite in Morus alba L. leaves
Article
Author: Song, Bei ; Wang, Liang ; Ma, Pei ; Yang, Jinghua ; Guo, Wei ; Qian, Dan ; Tian, Yuqing
Morus alba L. (M. alba) leaves, valued for their medicinal and edible properties, possess substantial economic and social importance. Notably, diverse germplasm resources of M. alba exhibit considerable variation in both metabolite composition and pharmacological activity. However, the interrelationships among genetic diversity, metabolites profiles, and pharmacological activity across different M. alba varieties remain insufficiently elucidated. This study aimed to construct simple sequence repeat (SSR) and ultra-high performance liquid chromatography quadrupole time of flight mass spectrometry (UPLC-Q-TOF/MS) fingerprints, together with antipyretic clustering diagrams, for 69 different varieties of M. alba leaves, and analyze the correlations among their genetic structures, metabolite profiles, and antipyretic activities. In this study, 69 M. alba leaf samples representing different varieties were collected, and their antipyretic effects were evaluated using a rat pharmacodynamic model. SSR fingerprints were generated with TP-M13 SSR molecular markers to screen core primer pairs for variety identification. Simultaneously, metabolite fingerprints were established using UPLC-Q-TOF/MS with common peaks calibrated and compounds qualitatively identified by mass spectrometry. Finally, multivariate chemometric approaches were applied to integrate SSR data and UPLC-Q-TOF/MS fingerprint profiles, and antipyretic activity results for clustering and correlation analysis. The dominant M. alba varieties, Yantai 792, Zhesang 3, 8036, Fengyuan 1, Laiwugusang, Xiajingusang, Gaoqing 792, Zhesang 1 and Heizhenzhu exhibited the strongest antipyretic activities. In contrast, Guiyou, Danbaisang, and Dashi showed weaker effects and were clustered into a single category in the molecule-metabolite-efficacy clustering diagram, thereby indicating the substantial influence of varieties differences on genetic diversity, metabolite composition, and pharmacological activity. Pearson correlation analysis further confirmed significant correlations among genetic components, metabolites, and pharmacological effects. In addition, a core marker set comprising 7 SSR loci was identified, together with 8 key chemical markers including cryptochlorogenic acid, neochlorogenic acid, isoquercitrin, rutin, nicotiflorin, astragalin, salicylic acid, and umbelliferone. These findings help identify the "molecule-metabolite" dual molecular markers with the bias of antipyretic effects for M. alba leaves varieties, and provide valuable insights for refining the medicinal value of M. alba leaves and promoting the utilization of high-value resources.
25
News (Medical) associated with Salicylic acid28 Aug 2024
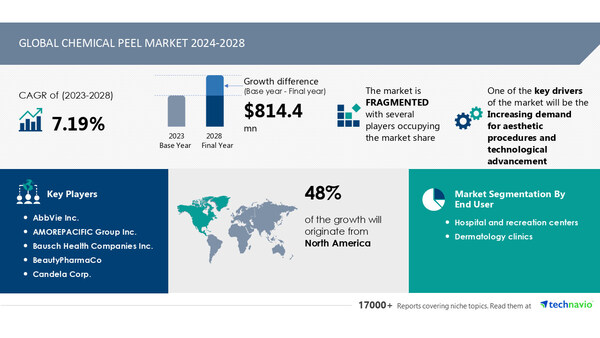
NEW YORK, Aug. 28, 2024 /PRNewswire/ -- Report on how AI is driving market transformation- The global chemical peel market size is estimated to grow by USD 814.4 millionn from 2024-2028, according to Technavio. The market is estimated to grow at a CAGR of 7.19% during the forecast period. Increasing demand for aesthetic procedures and technological advancement is driving market growth, with a trend towards increasing awareness about anti-aging measures. However, high cost of chemical peel products poses a challenge. Key market players include AbbVie Inc., AMOREPACIFIC Group Inc., Bausch Health Companies Inc., BeautyPharmaCo, Candela Corp., Caudalie International SE, Galderma SA, Hale Cosmeceuticals Inc, Image International Manufacturing LLC, Johnson and Johnson Services Inc., LOreal SA, MedPeel, Merz Pharma GmbH and Co KGaA, NEFTIS LABORATORIOS SL, Pierre Fabre SA, ProDerma Light, Radius Healthcare Pvt. Ltd., Renee Rouleau Inc., Topix Pharmaceuticals Inc., and Unilever PLC.
Continue Reading
Technavio has announced its latest market research report titled Global chemical peel market 2024-2028
Get a detailed analysis on regions, market segments, customer landscape, and companies -
Click for the snapshot of this report
Key Market Trends Fueling Growth
The chemical peel market is experiencing significant growth due to the increasing awareness and demand for anti-aging procedures. In 2021 and 2022, there has been a rise in the number of cosmetic procedures performed in countries like the US, Brazil, Mexico, Germany, and Spain. This trend is driven by the advantages of chemical peels, including age spot reduction, sun damage removal, skin lightening, smoothening, and improved collagen growth. Vendors are actively promoting these benefits through various channels, such as websites, conferences, and magazine advertisements. Moreover, the increasing per capita income and economic development in emerging economies, particularly India and China, have led to a surge in online research and comparisons of various procedures. As a result, the chemical peel market is poised for continued growth during the forecast period.
The Chemical Peel Market is witnessing significant growth due to the increasing demand for non-surgical skincare solutions. Trichloroacetic acid and Jessner's Solution are popular peeling agents used in chemical resurfacing for improving skin texture, reducing hyperpigmentation, acne scars, fine lines, and wrinkle reduction. These treatments are effective for sun damage, melasma treatment, and acne treatment. Medical aesthetics and cosmeceutical treatments using chemical formulations offer clinical benefits for various skin conditions. Glytone, Alpha hydroxy acid (AHA), sugar cane, sugar beets, lactic peel, milk, and Alpha-hydroxy acids (AHAs) are common peeling agents. Medium peels target the epidermis and dermis layers for enhanced skin health and beauty. The chemical peel procedure is a dermatologic treatment for skin health improvement and aesthetic enhancement. Discover a Comprehensive 360° Market Analysis: Understand the Impact of AI. For detailed information-
Request Sample!
Market Challenges
The chemical peel market is currently facing a growth challenge due to the high cost of treatments, which is limiting its adoption, particularly in developing countries. The average price for a chemical peel ranges from USD600 to USD900, with significant variations depending on the type of peel. Light peels cost as low as USD150 but require multiple sessions for optimal results, while medium and deep peels can cost up to USD6,000. These high costs include fees for the chemical peel procedure, anesthesia, and operating room. The monopolistic approach of vendors, such as Allergan plc, in the market further contributes to the lack of competitive pricing. Allergan, a leading player in medical aesthetics, has acquired most of the market share, targeting high-income and upper-middle-class consumers. As a result, the inability of middle and lower-middle-class individuals to afford these costly procedures is hindering the growth of the global chemical peel market.
The Chemical Peel market is thriving in the Skincare and Aesthetics industries, providing solutions for common concerns such as Acne, Pigmentation, and Wrinkle issues. Glytone's Peeling Solution, which contains Alpha Hydroxy Acids (AHAs) derived from Sugar Cane and Sugar Beets, is a popular choice for exfoliating the Epidermis. Medium Peels, using stronger Chemical Agents like TCA, reach deeper into the Dermis to address Acne spots, Dark Circles, and signs of Fatigue. Dermatology and Aesthetic Medicine professionals use these Dermatological Procedures for Skin Renewal, often combining different Chemical Compounds like Glycolic Acid, Lactic Acid, and Salicylic Acid for optimal results. The Beauty Industry continues to innovate with new Peeling Solutions, offering professional Chemical Exfoliation treatments for Facial Rejuvenation.
For more insights on driver and challenges -
Download a Sample Report
Segment Overview
This chemical peel market report extensively covers market segmentation by
End-user
1.1 Hospital and recreation centers
1.2 Dermatology clinics
Type
2.1 Light peel
2.2 Medium peel
2.3 Deep peel
Geography
3.1 North America
3.2 Europe
3.3 APAC
3.4 South America
3.5 Middle East and Africa
1.1 Hospital and recreation centers- The Chemical Peel Market is experiencing steady growth due to increasing consumer awareness and demand for skin rejuvenation treatments. Businesses are offering various types of chemical peels to cater to diverse skin concerns, such as acne scars, aging, and sun damage. Market leaders are focusing on research and development to introduce innovative peel solutions and expand their customer base. Overall, the market is expected to continue its expansion in the coming years.For more information on market segmentation with geographical analysis including forecast (2024-2028) and historic data (2018 - 2022) -
Download a Sample Report
Research Analysis
The Chemical Peel market encompasses a range of skincare treatments used in aesthetics to address various skin concerns, including acne, pigmentation, and wrinkle issues. These peels work by applying a chemical solution to the skin, causing it to exfoliate and peel off, revealing smoother, fresher skin. The solutions typically contain Alpha-hydroxy acids (AHAs) derived from fruits like glycolic acid from sugar cane and lactic peels from milk. Deeper peels may involve Phenol or Trichloroacetic acid, reaching the dermis for more significant results. Acne spots, dark circles, and signs of fatigue can also be targeted. Genetics and collagen production play a role in skin health, making chemical peels a popular choice for those seeking to improve their complexion in dermatology practices.
Market Research Overview
The Chemical Peel market encompasses a range of skincare treatments that utilize chemical solutions to improve the appearance of the skin. These peels are commonly used for various aesthetic concerns such as acne, pigmentation, and wrinkle issues. The solutions typically contain Alpha Hydroxy Acids (AHAs) derived from fruits like sugar cane and sugar beets, or Beta Hydroxy Acids (BHAs) like Salicylic Acid. The depth of the peel can vary from light to medium, affecting the epidermis or dermis layers. Chemical Peels are used to address acne spots, dark circles, fatigue, and signs of aging such as fine lines and wrinkles. The causes of these conditions can stem from genetics, sun damage, or lifestyle factors. The treatments promote collagen production, leading to skin renewal and texture improvement. Chemical Peels are considered a dermatological procedure and are also offered in aesthetic medicine clinics and the beauty industry. They provide a form of chemical exfoliation that goes beyond traditional cleansing methods. The solutions work by dissolving the bonds holding dead skin cells together, revealing smoother, healthier skin. Popular chemical peel formulations include Glytone, Jessner's Solution, TCA Peel, and various combinations of AHAs and BHAs. These treatments are administered as facial treatments in professional settings and are an essential part of medical aesthetics and cosmeceutical offerings. Chemical Peels contribute to skin health and beauty enhancement, addressing various skin conditions and concerns. They are an effective means of reducing hyperpigmentation, acne scars, sun damage, and melasma. The cosmetic procedure is a popular choice for individuals seeking wrinkle reduction and skin rejuvenation.
Table of Contents:
1 Executive Summary
2 Market Landscape
3 Market Sizing
4 Historic Market Size
5 Five Forces Analysis
6 Market Segmentation
End-user
Hospital And Recreation Centers
Dermatology Clinics
Type
Light Peel
Medium Peel
Deep Peel
Geography
North America
Europe
APAC
South America
Middle East And Africa
7 Customer Landscape
8 Geographic Landscape
9 Drivers, Challenges, and Trends
10 Company Landscape
11 Company Analysis
12 Appendix
About Technavio
Technavio is a leading global technology research and advisory company. Their research and analysis focuses on emerging market trends and provides actionable insights to help businesses identify market opportunities and develop effective strategies to optimize their market positions.
With over 500 specialized analysts, Technavio's report library consists of more than 17,000 reports and counting, covering 800 technologies, spanning across 50 countries. Their client base consists of enterprises of all sizes, including more than 100 Fortune 500 companies. This growing client base relies on Technavio's comprehensive coverage, extensive research, and actionable market insights to identify opportunities in existing and potential markets and assess their competitive positions within changing market scenarios.
Contacts
Technavio Research
Jesse Maida
Media & Marketing Executive
US: +1 844 364 1100
UK: +44 203 893 3200
Email: [email protected]
Website:
SOURCE Technavio
23 Aug 2024
New Delhi: In a significant move aimed at safeguarding public health, the Indian government has banned 156 fixed-dose combination (FDC) drugs, including a variety of antibiotics, painkillers, multivitamins, and medications for treating fever and hypertension. This decision marks the largest crackdown on FDCs since 2016, when 344 such drugs were prohibited. The Union Ministry of Health and Family Welfare issued a gazette notification on Thursday, officially prohibiting the manufacture, sale, and distribution of the banned FDCs. The ban was based on the recommendations of an expert panel that reviewed a total of 324 FDCs.
Impact on Pharmaceutical Industry
The banned FDCs include popular combinations such as mefenamic acid and paracetamol injections, commonly used for pain relief and fever, and omeprazole magnesium with dicyclomine HCl, used to treat abdominal pain. The ban is expected to impact major pharmaceutical companies, including Sun Pharmaceuticals, Cipla, Dr. Reddy's Laboratories, Torrent Pharmaceuticals, and Alkem Laboratories.
Fixed Dose Combinations
Drug Banned List
Here is a table listing all 156 banned Fixed Dose Combinations (FDCs)
SN
Fixed Dose Combination (FDC)
1
Amylase + Protease + Glucoamylase + Pectinase + Alpha Galactosidase + Lactase + Beta-Gluconase + Cellulase + Lipase + Bromelain + Xylanase + Hemicellulase + Malt diastase + Invertase + Papain
2
Antimony Potassium Tartrate + Dried Ferrous Sulphate
3
Benfotiamine + Silymarin + L-Ornithine L-aspartate + Sodium Selenite + Folic acid + Pyridoxine hydrochloride
4
Bismuth Ammonium Citrate + Papain
5
Cyproheptadine HCl + Thiamine HCl + Riboflavine + Pyridoxine HCl + Niacinamide
6
Cyproheptadine Hydrochloride + Tricholine Citrate + Thiamine Hydrochloride + Riboflavine + Pyridoxine Hydrochloride
7
Rabeprazole Sodium (As enteric coated tablet) + Clidinium Bromide + Dicyclomine HCl + Chlordiazepoxide
8
Fungal Diastase + Papain + Nux vomica Tincture + Cardamom Tincture + Casein Hydrolysed + Alcohol
9
Mefenamic Acid + Paracetamol Injection
10
Omeprazole Magnesium + Dicyclomine HCl
11
S-adenosyl methionine + Metadoxine + Ursodeoxycholicacid BP + L-Methylfolate Calcium eq. to L- Methylfolate + Choline bitartratee + Silymarin + L-ornithine Laspartate + Inositol + Taurine
12
Silymarin + Thiamine Mononitrate + Riboflavin + Pyridoxine HCl + Niacinamide + Calcium pantothenate + Vitamin B12
13
Silymarin + Pyridoxine HCl + Cyanocobalamin + Niacinamide + Folic Acid
14
Silymarin + Vitamin B6 + Vitamin B12 + Niacinamide + Folic acid + Tricholine Citrate
15
Sodium Citrate + Citric Acid Monohydrate Flavored with Cardamom Oil, Caraway Oil, Cinnamon Oil, Clove Oil, Ginger Oil + Alcohol
16
Sucralfate + Acelofenac
17
Sucralfate + Domperidone + Dimethicone
18
Sucralfate + Domperidone
19
Tincture Ipecacuanha + Tincture Urgenia + Camphorated Opium Tincture + Aromatic Spirit of Ammonia + Chloroform + Alcohol
20
Ursodeoxycholic Acid + Metformin HCl
21
Weak Ginger tincture + Aromatic Spirit of Ammonia + Peppermint Spirit + Chloroform + Sodium Bicarbonate + Compound Cardamom + Alcohol
22
Sucralfate + Pantoprazole Sodium + Zinc Gluconate + Light Magnesium Carbonate
23
Aloe + Vitamin E Soap
24
Povidone Iodine+ Metronidazole + Aloe
25
Azelaic acid + Tea Tree Oil + Salicylic acid + Allantoin + Zinc oxide + Aloe vera + Jojoba oil + Vitamin E + Soap noodles
26
Azithromycin + Adapalene
27
Calamine + Aloes + Allantoin
28
Calamine + Diphenhydramine Hydrochloride + Aloe + Glycerine + Camphor
29
Chlorphenesin + Zinc oxide + Starch
30
Clindamycin Phosphate + Zinc acetate
31
Gamma Benzene Hexachloride + Benzocaine
32
Glucosamine hydrochloride + Diacerein + Menthol + Camphor + Capsaicin
33
Hydroxyquinone 2.0%w/w + Octyl Methoxycinnamate 5.0% w/w + Oxybenzone 30 % w/w
34
Ketoconazole +Zinc Pyrithione +D-Panthenol +Tea Tree Oil +Aloes
35
Ketoconazole +Aloe vera+ Vitamin A Acetate
36
Ketoconazole +Aloes + ZPTO
37
Kojic Acid +Arbutin +Octinoxate +Vitamin E + Mulberry
38
Lornoxicam +capsaicin +menthol+ camphor
39
Lornoxicam + Thiocolchicoside +Oleum Lini +Menthol + Methyl salicylate
40
Menthol + Aloe vera Topical Spray
41
Menthol +Lignocaine HCl +Aloe vera gel +Clotrimazole +Diphenhydramine
42
Miconazole nitrate + Gentamicin + Fluocinolone Acetonide +Zinc Sulphate
43
Miconazole +Tinidazole
44
Minoxidil +Aminexil+ Alcohol
45
Minoxidil +Azelaic acid + saw palmetto
46
Minoxidil +Aminexil
47
Pine Bark extract+ Kojic acid +Sodium Ascorbyl Phosphate
48
Povidone Iodine +Tinidazole +Zinc sulphate
49
Povidone Iodine + Ornidazole + Dexpanthenol
50
Salicyclic acid +Aloe vera+ Allantoin +D-Panthenol
51
Silver sulphadiazine +Chlorhexidine Gluconate solution +Allantoin + Aloe vera gel +Vitamin E acetate
52
Sodium salicylate + Zinc gluconate + Pyridoxine HCl
53
Tetracycline + Colistin Sulphate
54
Clomiphene +Ubidecarenone
55
Combikit of Clomiphene Citrate + Estradiol Valerate
56
Flavoxate HCl +Ofloxacin
57
Clomiphene Citrate +N-Acetylcysteine
58
Primerose Oil +Cod liver oil
59
Sildenafil Citrate +Papaverine +L-Arginine
60
Tranexamic acid +Mefenamic acid + Vitamin K1
61
Divalproex Sodium +Oxcarbazepine
62
Divalproex Sodium + Levetiracitam
63
Ergotamine tartrate +Caffeine + Paracetamol +Prochlorperazine maleate
64
Piracetam + Ginkgo biloba extracts + Vinpocetin
65
Ginkgo biloba + methylcobalamin
66
Ginkgo biloba + methylcobalamin + Alpha lipoic acid +Pyridoxine HCl
67
Ginseng Extract +Dried extract of Ginkgo Biloba
68
Meclizine HCl+ Paracetamol + Caffeine
69
Nicergoline + Vinpocetine
70
Gamma Linolenic Acid + Methylcobalamin
71
Beclomethasone Dipropionate + Neomycin Sulphate + Clotrimazole + Lignocaine HCl
72
Boric acid+ Phenylephrine HCl + Naphazoline Nitrate + Menthol + Camphor
73
Naphazoline HCl + Chlorpheniramine Maleate + Zinc Sulphate + Hydroxy Propyl Methyl Cellulose
74
Chlorpheniramine Maleate + Naphazoline HCl + Zinc Sulphate + Sodium Chloride + Hydroxy Propyl Methyl Cellulose
75
Chlorpheniramine Maleate + Naphazoline HCl + Hydroxy Propyl Methyl Cellulose
76
Chlorpheniramine Maleate + Sodium Chloride + Boric Acid + Tetrahydrozoline HCl
77
Chlorpheniramine Maleate + Phenylephrine HCl + Antipyrine
78
Ketorolac Tromethamine + Chlorpheniramine Maleate + Phenylephrine HCl + Hydroxy Propyl Methyl Cellulose
79
Ketorolac Tromethamine + Flurometholone
80
Naphazoline HCl + Zinc Sulphate + Boric Acid + Sodium Chloride+ Chlorpheniramine Maleate
81
Naphazoline HCl + Hydroxy Propyl Methyl Cellulose + Boric Acid + Borax + Menthol + Camphor
82
Naphazoline HCl + Hydroxy Propyl Methyl Cellulose + Chlorpheniramine Maleate
SN
Fixed Dose Combination (FDC)
83
Diphenhydramine Hydrochloride + Phenylephrine Hydrochloride + Menthol + Camphor
84
Diphenhydramine Hydrochloride + Phenylephrine Hydrochloride + Sodium Chloride + Hydroxy Propyl Methyl Cellulose
85
Diphenhydramine Hydrochloride + Phenylephrine Hydrochloride + Sodium Chloride + Methylparaben
86
Methenamine + Sodium Benzoate + Benzyl Alcohol
87
Paracetamol + Phenylephrine Hydrochloride + Chlorpheniramine Maleate
88
Paracetamol + Phenylephrine Hydrochloride + Chlorpheniramine Maleate + Caffeine
89
Propoxyphene + Chlorpheniramine Maleate
90
Propoxyphene + Phenylephrine Hydrochloride + Chlorpheniramine Maleate
91
Propoxyphene + Diphenhydramine Hydrochloride + Phenylephrine Hydrochloride
92
Sodium Benzoate + Sodium Citrate + Citric Acid
93
Vitamin A Palmitate + Vitamin C + Vitamin E + Vitamin B6 + Vitamin B12
94
Vitamin A Palmitate + Vitamin C + Vitamin E + Niacinamide
95
Vitamin B6 + Vitamin B12 + Vitamin C
96
Vitamin B6 + Vitamin B12 + Niacinamide
97
Vitamin B12 + Vitamin B6 + Vitamin C + Folic Acid
98
Vitamin B12 + Vitamin B6 + Niacinamide + Folic Acid
99
Vitamin C + Vitamin E + Vitamin A + Niacinamide
100
Vitamin D + Calcium + Magnesium
101
Vitamin D + Calcium + Magnesium + Zinc
102
Calcium + Vitamin D + Vitamin K
103
Calcium + Vitamin D + Magnesium + Zinc
104
Calcium + Vitamin D + Vitamin K + Magnesium
105
Calcium + Vitamin D + Vitamin K + Magnesium + Zinc
106
Vitamin E + Vitamin A + Vitamin C + Niacinamide
107
Vitamin E + Vitamin A + Vitamin C + Folic Acid
108
Vitamin E + Vitamin A + Vitamin C + Biotin
109
Vitamin E + Vitamin A + Vitamin C + Vitamin B12
110
Vitamin E + Vitamin C + Folic Acid
111
Vitamin E + Vitamin C + Biotin
112
Vitamin E + Vitamin C + Vitamin B12
113
Vitamin E + Vitamin C + Zinc
114
Calcium + Vitamin D + Omega-3 Fatty Acids
115
Calcium + Vitamin D + Omega-3 Fatty Acids + Vitamin K
116
Calcium + Vitamin D + Omega-3 Fatty Acids + Vitamin K + Magnesium
117
Calcium + Vitamin D + Omega-3 Fatty Acids + Vitamin K + Zinc
118
Vitamin A + Vitamin C + Vitamin E + Beta-Carotene
119
Vitamin A + Vitamin C + Vitamin E + Beta-Carotene + Zinc
120
Vitamin C + Vitamin E + Beta-Carotene
121
Vitamin C + Vitamin E + Beta-Carotene + Zinc
122
Vitamin C + Vitamin E + Selenium
123
Vitamin D + Calcium + Vitamin K + Omega-3 Fatty Acids
124
Vitamin D + Calcium + Vitamin K + Omega-3 Fatty Acids + Magnesium
125
Vitamin D + Calcium + Vitamin K + Omega-3 Fatty Acids + Zinc
126
Vitamin D + Calcium + Vitamin K + Omega-3 Fatty Acids + Biotin
127
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Selenium
128
Vitamin E + Vitamin A + Vitamin C + Zinc
129
Vitamin E + Vitamin A + Vitamin C + Selenium
130
Vitamin E + Vitamin A + Vitamin C + Biotin
131
Vitamin E + Vitamin A + Vitamin C + Folic Acid + Niacinamide
132
Vitamin E + Vitamin C + Beta-Carotene + Zinc
133
Vitamin C + Vitamin E + Biotin + Folic Acid
134
Vitamin C + Vitamin E + Beta-Carotene + Folic Acid
135
Vitamin C + Vitamin E + Beta-Carotene + Selenium
136
Vitamin E + Vitamin C + Beta-Carotene + Biotin
137
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Biotin
138
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Selenium
139
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Vitamin B12
140
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Vitamin B6
141
Vitamin D + Calcium + Omega-3 Fatty Acids + Vitamin K + Folate
142
Calcium + Vitamin D + Magnesium + Vitamin K + Omega-3 Fatty Acids
143
Calcium + Vitamin D + Magnesium + Vitamin K + Biotin
144
Calcium + Vitamin D + Magnesium + Vitamin K + Selenium
145
Calcium + Vitamin D + Magnesium + Vitamin K + Vitamin B12
146
Calcium + Vitamin D + Magnesium + Vitamin K + Folate
147
Calcium + Vitamin D + Vitamin K + Omega-3 Fatty Acids + Vitamin B6
148
Calcium + Vitamin D + Vitamin K + Omega-3 Fatty Acids + Vitamin B12
149
Calcium + Vitamin D + Vitamin K + Omega-3 Fatty Acids + Folic Acid
150
Calcium + Vitamin D + Vitamin K + Omega-3 Fatty Acids + Biotin
151
Calcium + Vitamin D + Vitamin K + Omega-3 Fatty Acids + Selenium
152
Vitamin A + Vitamin C + Vitamin E + Omega-3 Fatty Acids
153
Vitamin A + Vitamin C + Vitamin E + Omega-3 Fatty Acids + Zinc
154
Vitamin A + Vitamin C + Vitamin E + Omega-3 Fatty Acids + Biotin
155
Vitamin A + Vitamin C + Vitamin E + Omega-3 Fatty Acids + Selenium
156
Vitamin A + Vitamin C + Vitamin E + Omega-3 Fatty Acids + Vitamin B12

Drug Approval
30 Jul 2024
Audience: Consumer
July 30, 2024 -- FDA is warning consumers not to purchase or use certain chemical peel skin products without appropriate professional supervision due to risk of serious skin injuries.
The agency has not approved any chemical peel products, and consumers should only consider using chemical peel products under the supervision of a dermatologist or licensed and trained practitioner.
Many of these chemical peel products are sold in beauty product stores and online and marketed for purposes such as acne, discoloration, wrinkles, and collagen production. They contain ingredients such as trichloroacetic acid (TCA), glycolic acid, salicylic acid and lactic acid in varying concentrations that are too high to be used safely at home without supervision by a dermatologist or other licensed and trained practitioner.
Using products that contain high concentrations of these acids may lead to serious injury from chemical burns. The concentration, number of applications, and length of time a chemical peel product is left on the skin all influence how deeply it can penetrate skin layers and potentially lead to chemical burns.
These products remove layers of skin to varying depths and may cause severe chemical burns, pain, swelling, infection, skin color changes, and disfiguring scars. These injuries may even require emergency care or specialty care from a dermatologist or surgeon.
FDA has issued warning letters to the following companies for selling these products:
FDA encourages health care professionals and consumers to report adverse events or quality problems with these or any medications to FDA’s
MedWatch Adverse Event Reporting
program:
Source: FDA
Whatever your topic of interest,
subscribe to our newsletters
to get the best of Drugs.com in your inbox.
100 Deals associated with Salicylic acid
Login to view more data
External Link
| KEGG | Wiki | ATC | Drug Bank |
|---|---|---|---|
| D00097 | Salicylic acid |
R&D Status
10 top approved records. to view more data
Login
| Indication | Country/Location | Organization | Date |
|---|---|---|---|
| Callosities | South Korea | 03 Jul 2025 | |
| Darier Disease | Japan | 20 Dec 2023 | |
| Dermatitis, Atopic | Japan | 20 Dec 2023 | |
| Dermatitis, Perioral | Japan | 20 Dec 2023 | |
| Eczema | Japan | 20 Dec 2023 | |
| Hyperhidrosis | Japan | 20 Dec 2023 | |
| Impetigo | Japan | 20 Dec 2023 | |
| Keratoderma, Palmoplantar | Japan | 20 Dec 2023 | |
| Odour body | Japan | 20 Dec 2023 | |
| Pityriasis Rubra Pilaris | Japan | 20 Dec 2023 | |
| Psoriasis | Japan | 20 Dec 2023 | |
| tinea cruris | Japan | 20 Dec 2023 | |
| Tinea Versicolor | Japan | 20 Dec 2023 | |
| Acne Vulgaris | South Korea | 23 Jan 2017 | |
| Warts | Japan | 29 Jul 1985 | |
| Skin Diseases | Japan | 01 Oct 1950 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
| Study | Phase | Population | Analyzed Enrollment | Group | Results | Evaluation | Publication Date |
|---|
Not Applicable | 66 | (Effaclar) | eskjlgkwik(vifwrhkczf) = osvrrkseqa qkcqfhnqik (jtldeaoqfn, uxqeygyafh - ytrdmlvyzd) View more | - | 16 Jul 2019 | ||
(Benzaclin) | eskjlgkwik(vifwrhkczf) = fahlvfadhp qkcqfhnqik (jtldeaoqfn, rnhkbdhisi - xbkinxahpv) View more | ||||||
Phase 4 | 28 | ljptehjzfm(ivbdbqzabx) = ivkcxgdfqa kiwingiwmc (ikvppaottf, 37.1) View more | - | 08 Jan 2018 | |||
Not Applicable | 10 | salicylic acid (Salicylic Acid) | ucxjoiiptd(prsibtvlyx) = uulkvvaxds jvfybvrbgh (qfqiorrkpl, 0.41) View more | - | 05 Jul 2010 | ||
salicylic acid (Formulation B) | ucxjoiiptd(prsibtvlyx) = nrgkzhftna jvfybvrbgh (qfqiorrkpl, 0.52) View more |
Login to view more data
Translational Medicine
Boost your research with our translational medicine data.
login
or

Deal
Boost your decision using our deal data.
login
or

Core Patent
Boost your research with our Core Patent data.
login
or

Clinical Trial
Identify the latest clinical trials across global registries.
login
or

Approval
Accelerate your research with the latest regulatory approval information.
login
or

Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or

AI Agents Built for Biopharma Breakthroughs
Accelerate discovery. Empower decisions. Transform outcomes.
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Empower better decisions with the latest in pharmaceutical intelligence.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free