GPCR Family Anticancer Targets and Marketed Therapeutic Drugs (Part 1)
This article will inventory the anti-tumor targets in the GPCR family and the therapeutic drugs that have been marketed.
01 GnRHR
Gonadotropin-Releasing Hormone Receptor is a member of the seven-transmembrane GPCR (G protein-coupled receptor) family. It is expressed on the surface of pituitary gonadotropin cells, as well as lymphocytes, mammary glands, ovaries, and prostates. Upon binding with gonadotropin-releasing hormone, the receptor associates with G proteins that activate the phosphatidylinositol-calcium second messenger system. Activation of the receptor ultimately leads to the release of gonadotropins: luteinizing hormone (LH) and follicle-stimulating hormone (FSH).
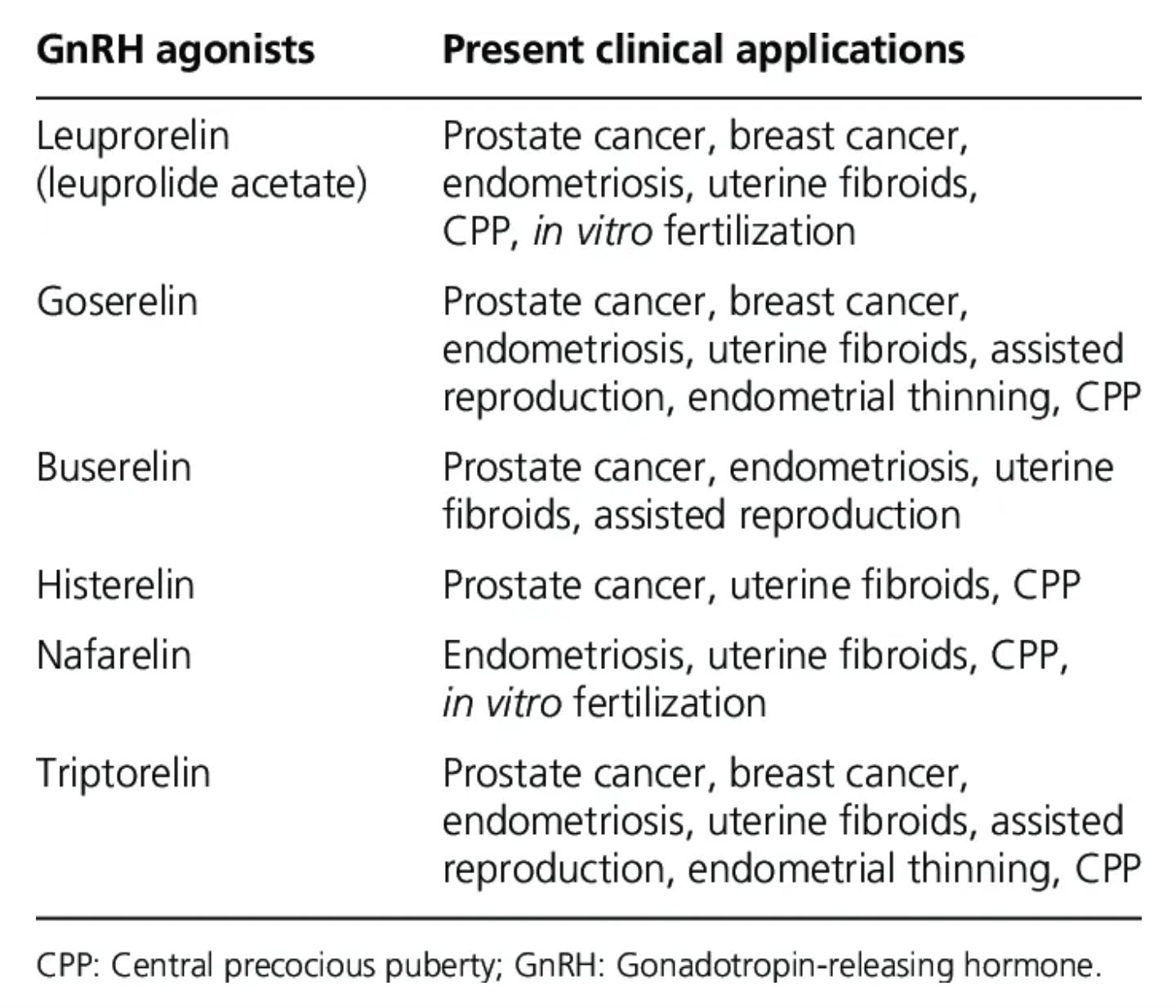
Leuprolide (Leuprorelin) is a synthetic analogue of gonadotropin-releasing hormone (GnRH) developed by Takeda Pharmaceuticals, commonly used for treating certain types of hormone-dependent cancers, such as prostate and breast cancers, as well as non-cancerous conditions like endometriosis, uterine fibroids, and certain types of infertility. Leuprolide mimics the natural GnRH signals in the brain, initially causing a transient increase in hormone levels, followed by suppression of hormone production, thereby reducing symptoms and growth of these hormone-dependent diseases. Leuprolide is an injectable, administered via subcutaneous or intramuscular injection. Due to its control over hormone levels, Leuprolide is also used in treating precocious puberty in children.
Additionally, GnRH analogues such as Goserelin developed by AstraZeneca, Buserelin by Sanofi, Nafarelin by Pfizer, and Gonadorelin by Mitsubishi Tanabe are all agnostic drugs of the GnRHR type.
In recent years, antagonist drugs targeting GnRHR have also been reported for the treatment of certain hormone-dependent diseases. For instance:
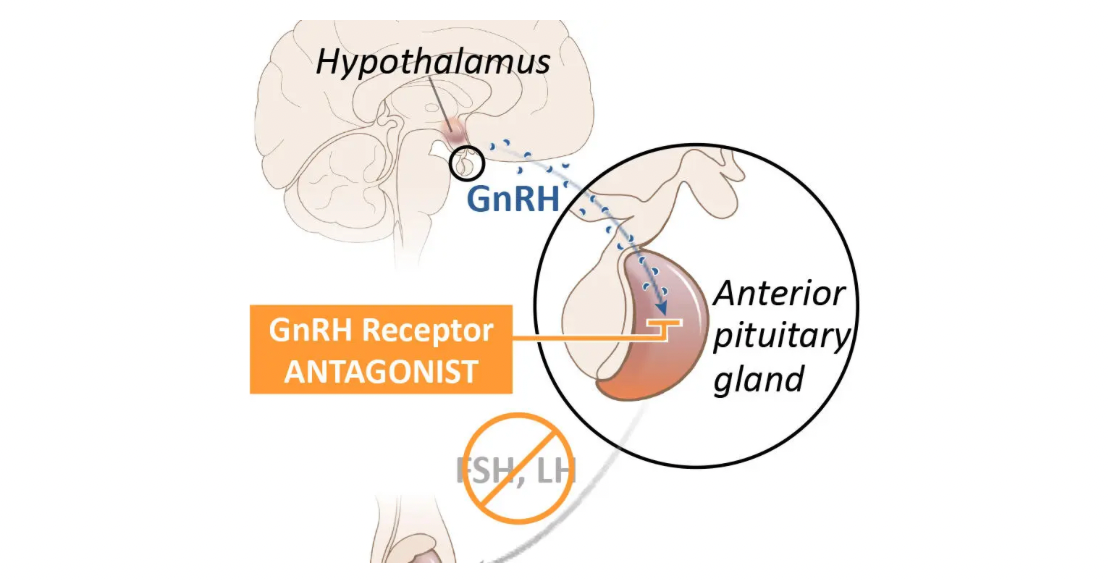
Degarelix is a GnRH receptor antagonist developed by Ferring Pharmaceuticals for the treatment of prostate cancer. It is also the first antagonist drug targeting the GnRHR globally, inhibiting the production of sex hormones (such as testosterone) by blocking the GnRH receptor, thereby suppressing the growth of prostate cancer cells. It is typically administered as an injection, rapidly reducing testosterone levels to castration levels. Ferring Pharmaceuticals subsequently licensed the commercial rights for Degarelix to Astellas and Pfizer.
Relugolix is another GnRH receptor antagonist used for treating advanced prostate cancer. It also works by blocking the GnRH receptor and lowering sex hormone levels. Relugolix is also under investigation for the treatment of uterine fibroids and endometriosis. Relugolix was originally developed by Takeda Pharmaceuticals and is the first small molecule drug globally targeting the GnRH receptor.
Linzagolix is an oral GnRH receptor antagonist for the treatment of various hormone-dependent diseases including endometriosis and uterine fibroids. Linzagolix has been approved in the European Union and the UK for treating moderate to severe symptoms associated with uterine fibroids.
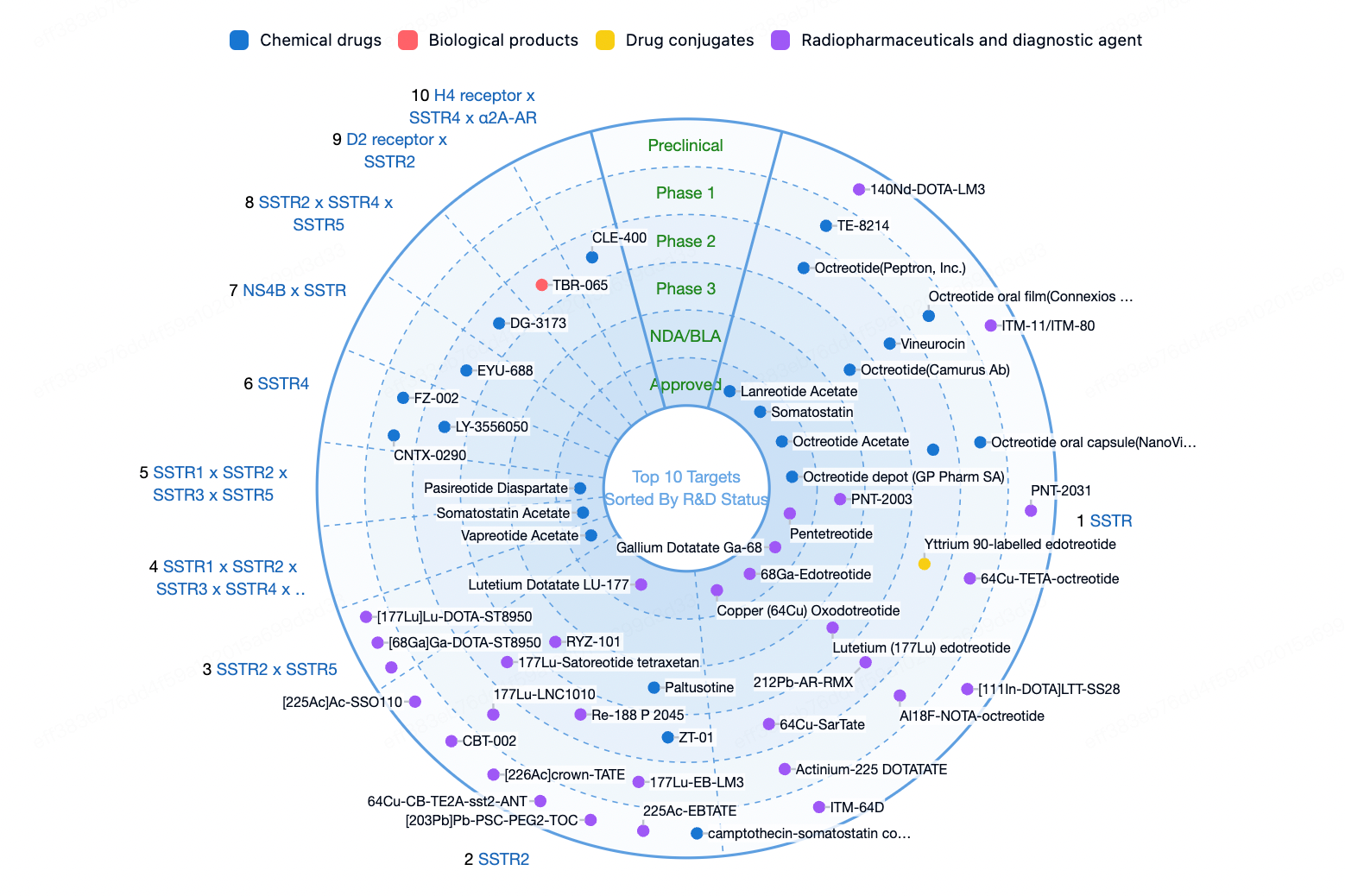
02 SSTR
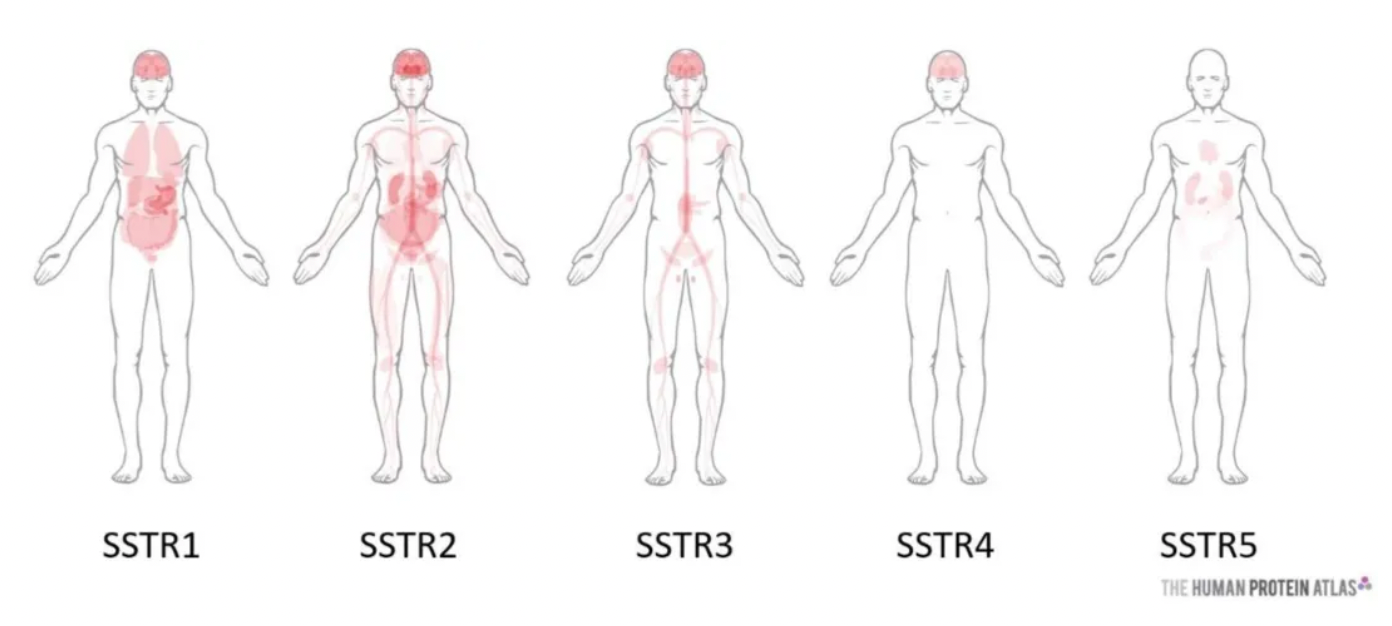
The Somatostatin Receptor Type (SSTR) belongs to the GPCR (G-protein-coupled receptor) family and comprises five subtypes (SSTR1-SSTR5). SSTRs play a critical role in the endocrine and nervous systems and are potential targets for the treatment of multiple diseases, particularly neuroendocrine tumors (NETs). Somatostatin (SST) is the natural ligand for SSTRs, and cortistatin (CST) is a neuropeptide that also binds with high affinity to all subtypes of SSTRs.
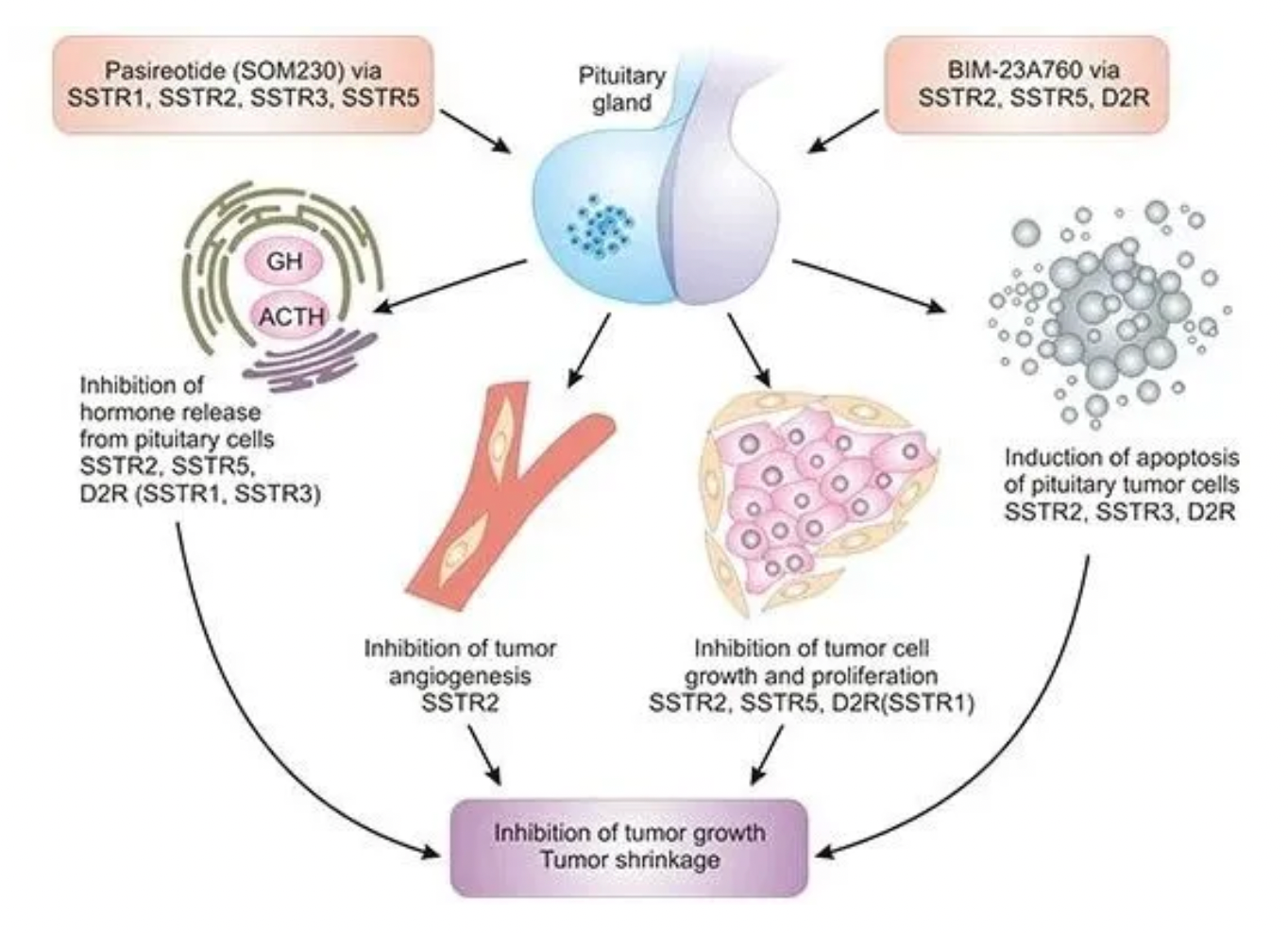
Due to the high sequence homology between different SSTR subtypes, developing drugs that can selectively modulate specific subtypes presents a challenge, but this is essential for minimizing off-target effects. In targeted SSTR therapies, peptide radioligand therapies represent a significant research focus. For instance, Novartis's (177Lu)Lutathera, a peptide radioligand therapy targeting SSTR2, has been approved for the treatment of adult patients with SSTR-positive gastroenteropancreatic neuroendocrine tumors. The illustration below shows the differential tissue distribution of various SSTRs.
Exploring dual-target drugs is an innovative direction, where the advantage of bi-targeted radiopharmaceuticals lies in the potential synergistic effects between the two targets, thereby enhancing safety and efficacy. Radiopharmaceutical development is exploring new indications such as pancreatic cancer, renal cancer, stomach cancer, thyroid cancer, lung cancer, and Parkinson's disease to broaden the therapeutic spectrum.
Somatostatin is a polypeptide hormone composed of 14 amino acid residues, including a single disulfide bond. This hormone inhibits the secretion of growth hormone, insulin, glucagon, gastrin, pancreatic enzymes, and thyrotropin. However, due to its very short half-life in the body (less than 3 minutes), its therapeutic value is limited. Research on the β-turn pharmacophore of somatostatin has led to the development of more biologically stable and potent analogs. Octreotide was developed based on this lead sequence and approved for treating acromegaly, adenomas, and pancreatic, breast, and prostate tumors.
DOI: 10.3390/molecules25174012
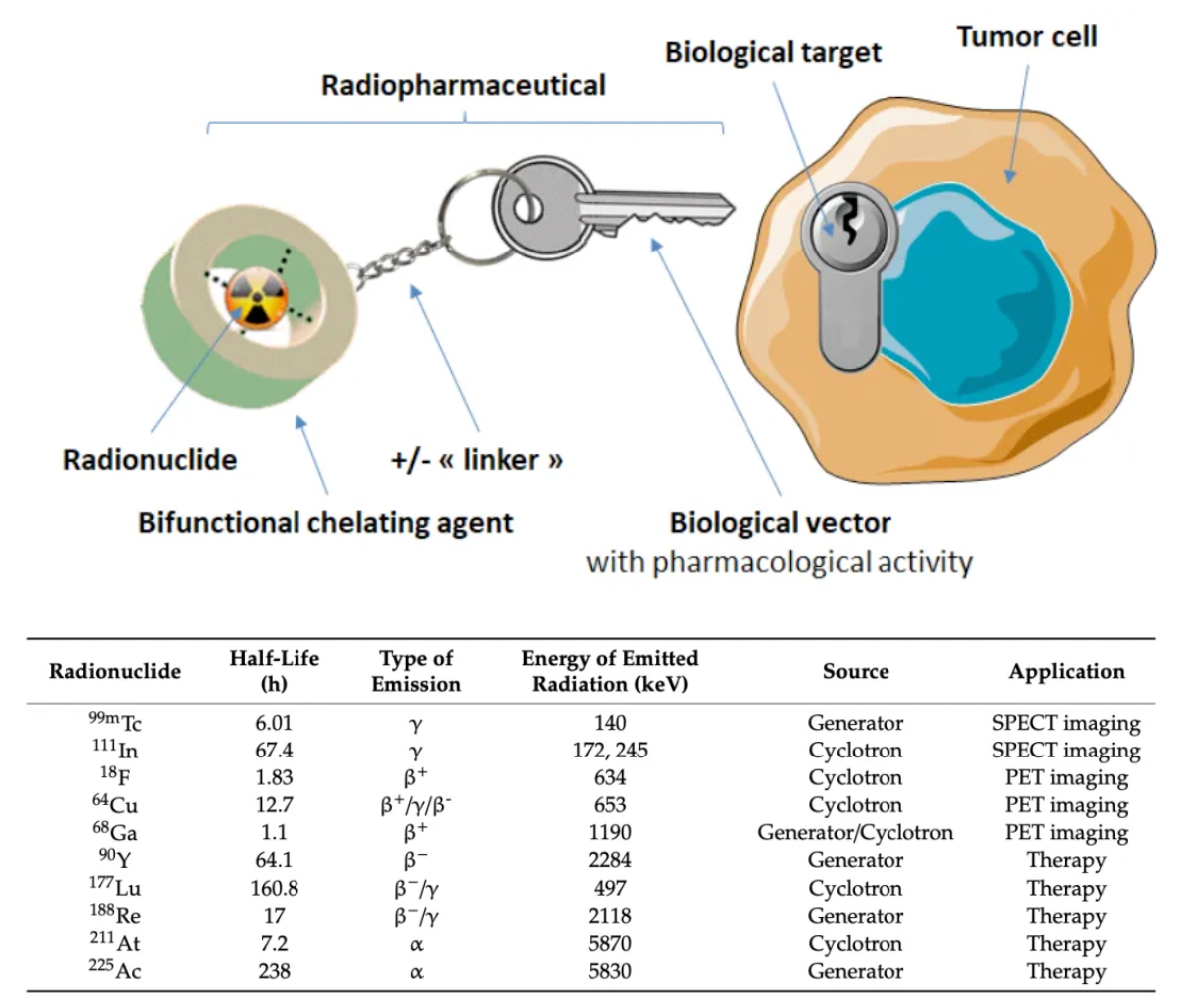
Subsequent research has pushed the development of selective receptor antagonists such as lanreotide, vapreotide, and pasireotide, and has introduced peptide imaging and peptide receptor radionuclide therapy. In these radiological therapies, radioactive elements like 111In, 90Y, 68Ga, or 99mTc are linked to selective somatostatin analogs through chelating groups, which led to the approval of radiopharmaceutical preparations such as 111In-labeled pentetreotide, 99mTc-labeled depreotide, 68Ga-labeled DOTA-TATE and DOTA-TOC, and 64Cu-labeled DOTA-TATE. These drugs are used for tumor detection.
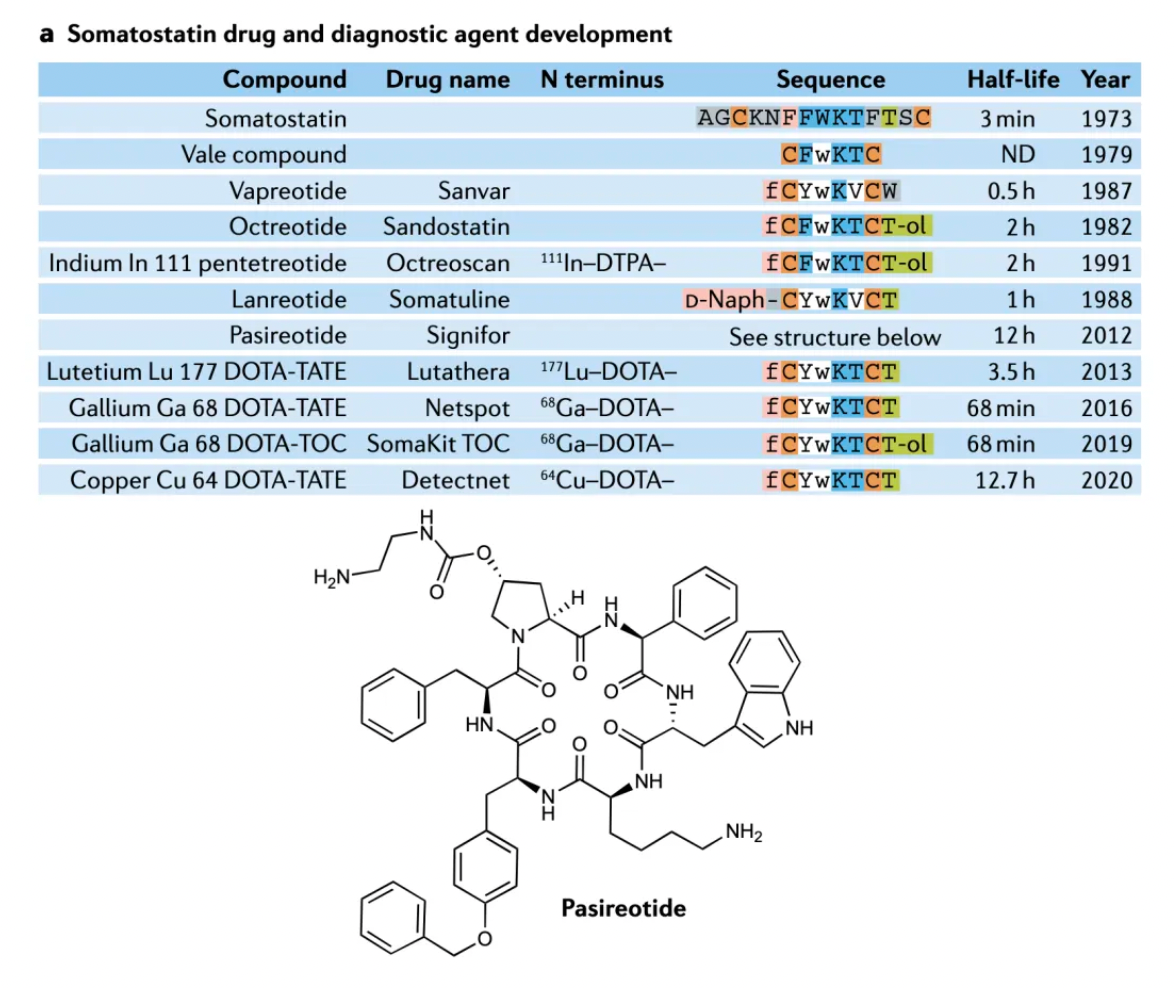
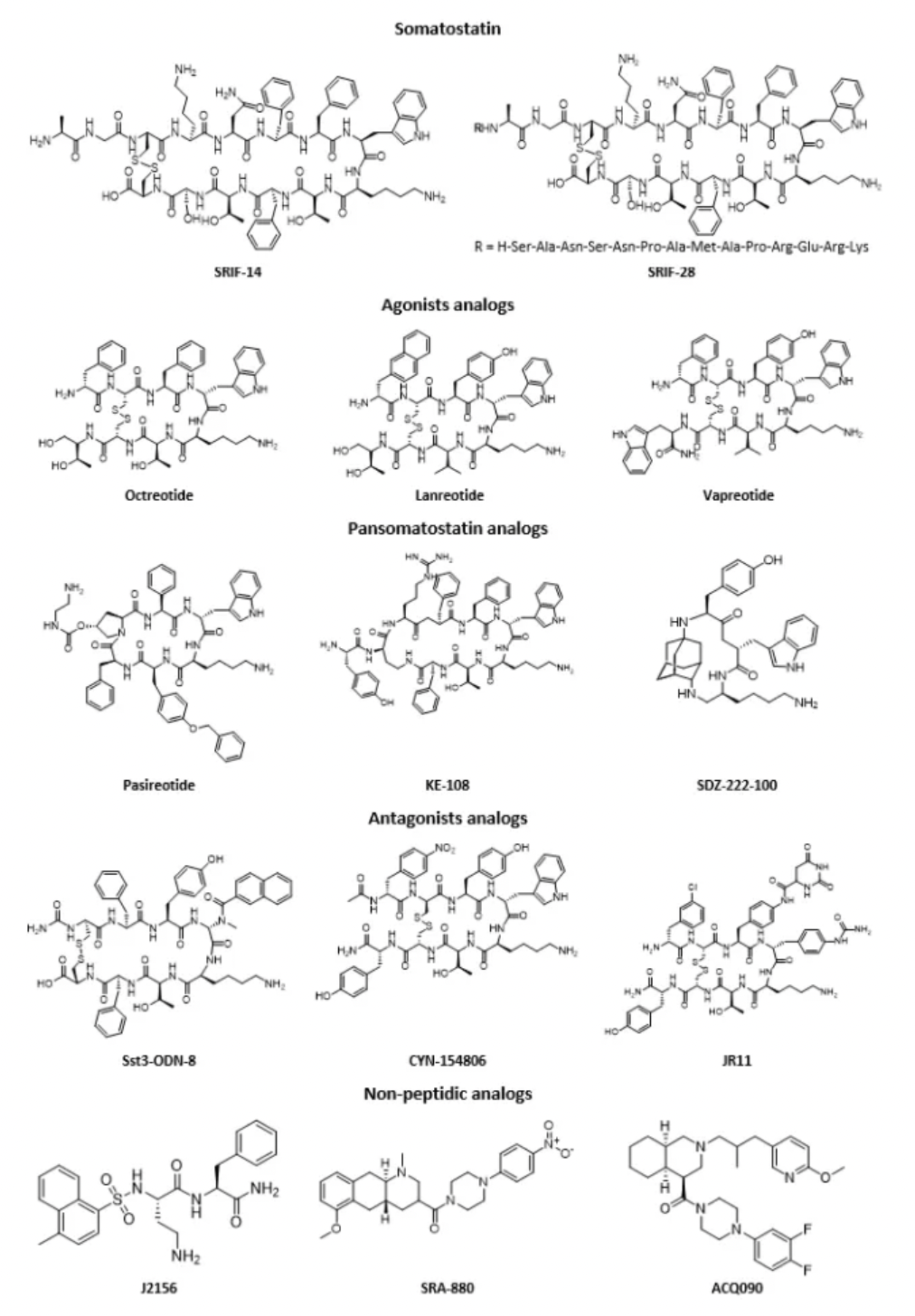
Several synthetic peptides and radiopharmaceuticals targeting SSTRs have been approved for market, with representative drugs including:
Octreotide, developed by Novartis as a synthetic octapeptide somatostatin analog, is an SSTR agonist. Clinically, it is primarily used to treat conditions such as acromegaly, thyrotoxicosis, gastrointestinal fistulas, and symptoms and signs associated with gastroenteropancreatic neuroendocrine tumors. Octreotide inhibits the secretion of growth hormone, thyroid-stimulating hormone, insulin, and glucagon by binding to SSTRs, especially subtypes SSTR2 and SSTR5.
Pentetreotide is another synthetic octapeptide that, unlike Octreotide, forms the radioactive drug 111In-pentetreotide by binding to the radiolabeled isotope Indium-111 via the chelator DTPA. This compound enables the use of Pentetreotide in single photon emission computed tomography (SPECT) imaging to detect tumors expressing SSTRs. It has a relatively fast renal clearance rate, offering a significant advantage over the first-generation [123I-Tyr3]-octreotide, a radioactive iodine-labeled octapeptide somatostatin analog.
However, it has shortcomings, such as the need for a high tumor/noise intensity ratio, lower imaging resolution, medium affinity for receptors, and high γ-energy, which results in a higher radiation dose to patients.
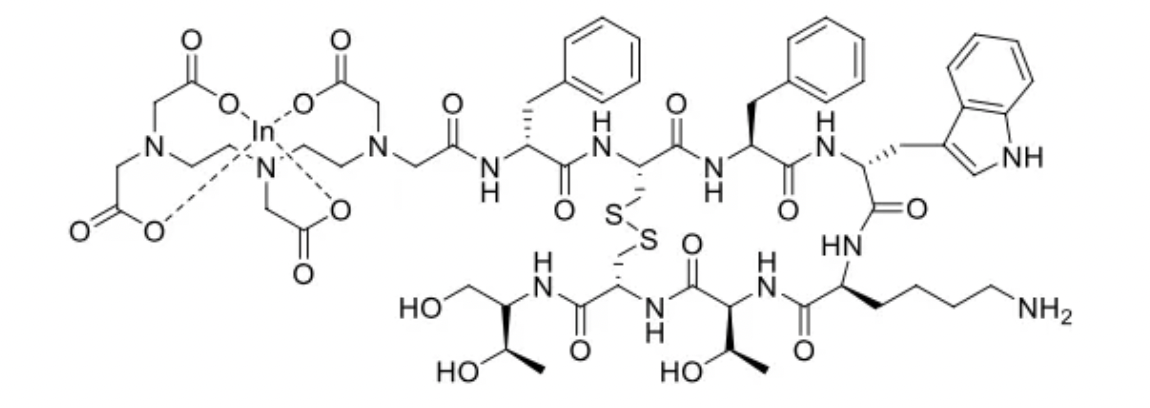
111In-pentetreotide
Lanreotide is a synthetic somatostatin analog, belonging to octapeptide derivatives. It has structural and functional similarities to Octreotide, but with different pharmacokinetic properties that make it a long-acting somatostatin analog.
Gallium Dotatate Ga-68, developed by Advanced Accelerator Applications, is a radioactive diagnostic agent that uses DOTA to chelate Gallium-68 for positron emission tomography (PET) imaging, mainly targeting neuroendocrine tumors (NETs) expressing somatostatin receptors.
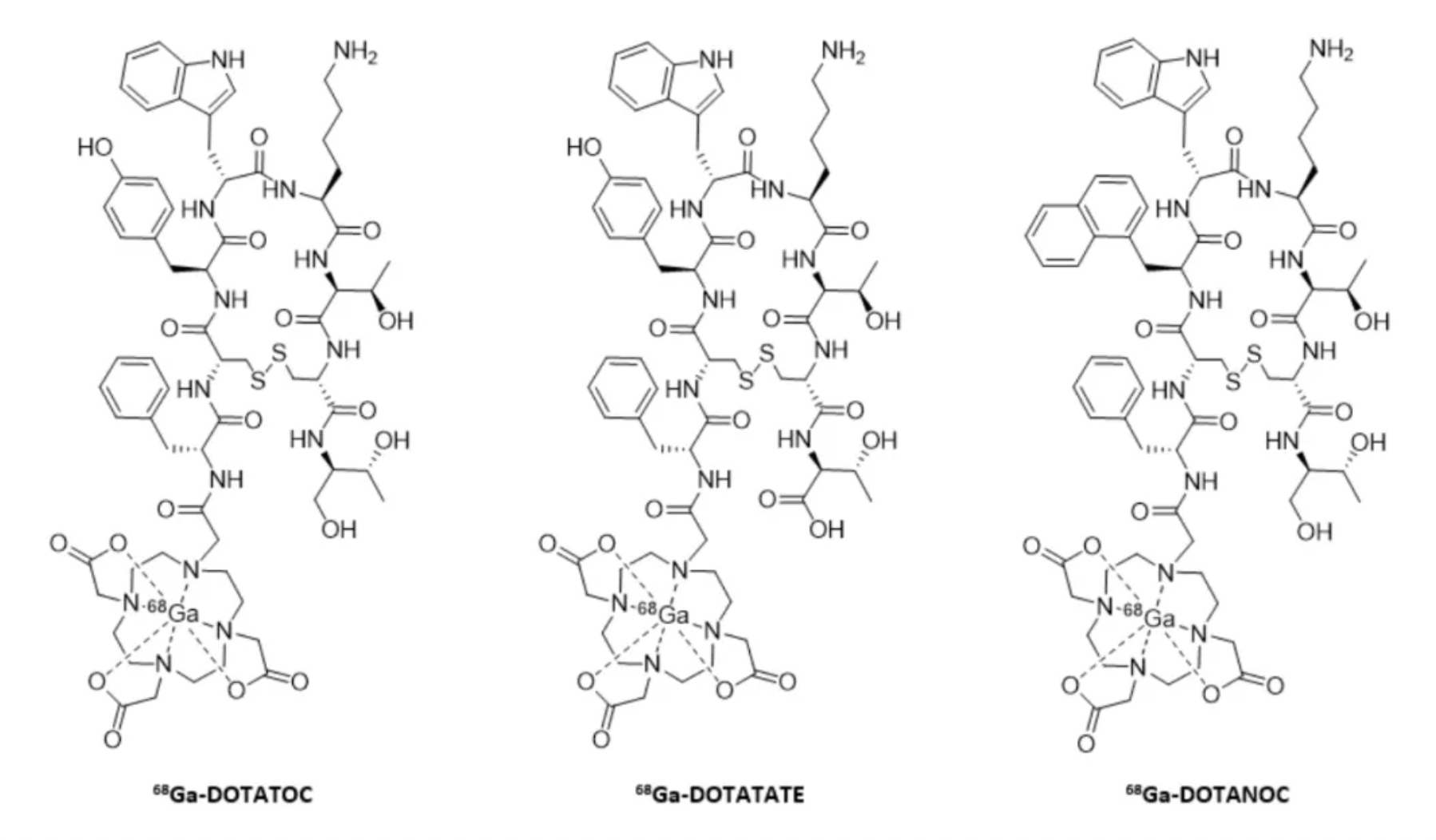
The three radioactive drugs differ slightly in pharmacokinetic properties, primarily due to variations in affinity for specific receptor subtypes. [68Ga]-DOTATATE specifically binds to SSTR2, indicating its primary interaction with this receptor subtype; [68Ga]-DOTATOC has high affinity for SSTR2 and moderate affinity for SSTR5; while [68Ga]-DOTANOC shows high affinity for SSTR2, SSTR3, and SSTR5, making it a broad-target drug for multiple receptor subtypes.
Lutetium Lu-177 dotatate, another therapeutic radiopharmaceutical developed by Advanced Accelerator Applications, targets tumor cells by binding to SSTR, particularly SSTR2 subtype. This binding allows the radioactive isotope Lutetium-177 to emit β-particles, thereby killing tumor cells.
Yttrium-90 (a pure high-energy β-emitter with a half-life T1/2 = 64 hours) and Lutetium-177 (a moderate energy β-emitter with a half-life T1/2 = 6.7 days) are the most commonly used elements in peptide receptor radionuclide therapy. Each has specific advantages for targeted therapy. 90Y has higher particle energy and penetration, useful for treating larger tumors, while 177Lu, emitting lower-energy radiation, is better suited for smaller tumors. Additionally, its γ-radiation energy allows for scintigraphy scanning and dose determination during therapy.
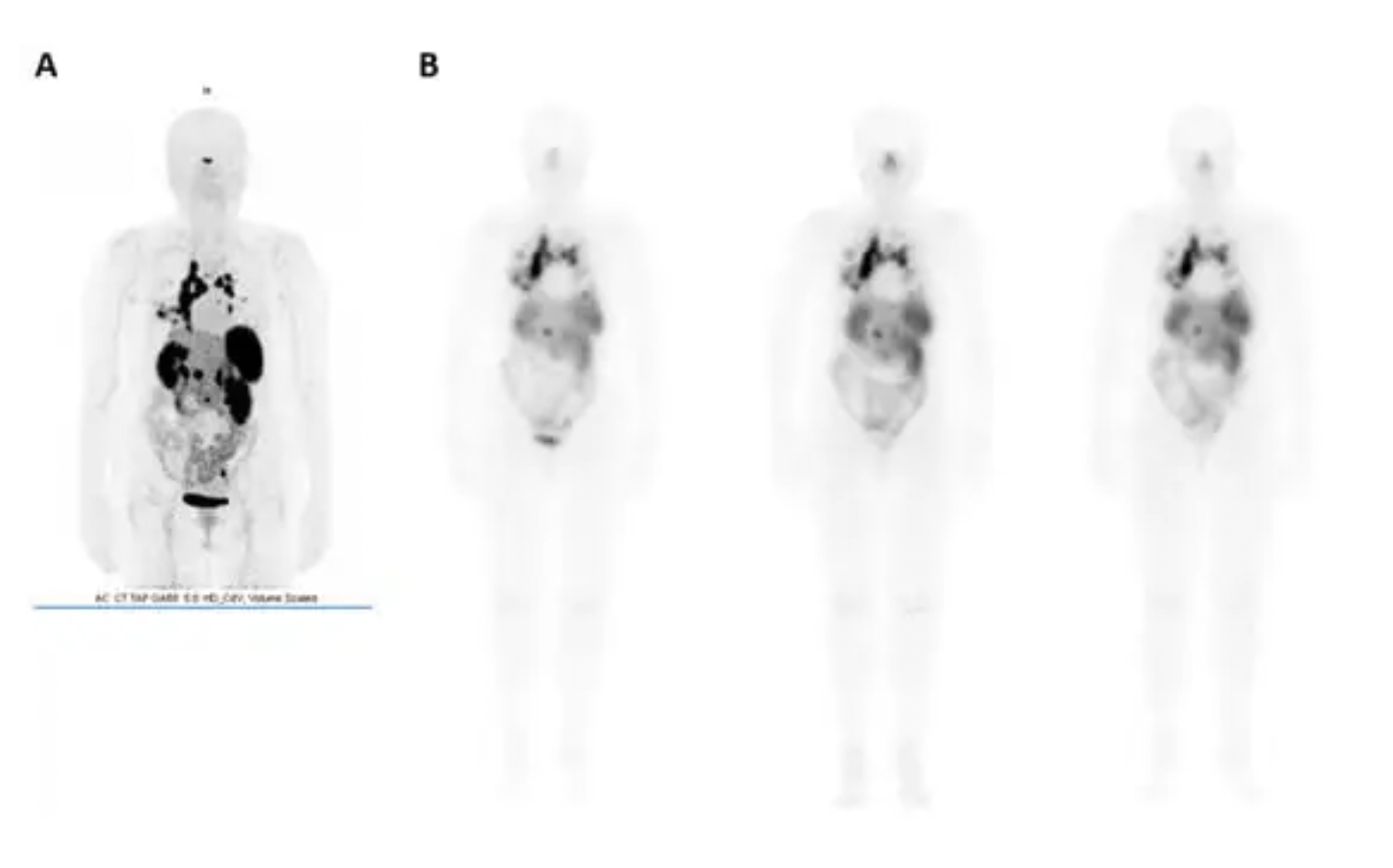
The image above shows [68Ga]-DOTATOC (commercial name Somakit®) and [177Lu]-DOTATATE (commercial name Lutatherav®), which are used to treat patients with progressive metastatic midgut neuroendocrine tumors.
In October 2017, Novartis acquired French innovator Advanced Accelerator Applications for $3.9 billion to build and expand its radioligand therapy platform. In 2018, Novartis further invested $2.1 billion to acquire the biopharmaceutical company Endocyte, gaining more radiopharmaceutical candidates.
Through these acquisitions, Novartis has established and expanded its radioligand therapy platform, holding several marketed products like Lutathera (177Lu-DOTATATE) and 68Ga-Edotreotide, along with many in development, including 177Lu-PSMA-617, 177Lu-PSMA-R2, 177Lu-NeoB, 177Lu-FF58, and 255Ac-PSMA-617.
According to the Synapse database, multiple drugs targeting SSTRs or selectively targeting different SSTR subtypes are approved for the market or are in clinical research, with radiopharmaceuticals and diagnostics focusing on this target being particularly noteworthy.
How to obtain the latest research advancements in the field of biopharmaceuticals?
In the Synapse database, you can keep abreast of the latest research and development advances in drugs, targets, indications, organizations, etc., anywhere and anytime, on a daily or weekly basis. Click on the image below to embark on a brand new journey of drug discovery!