Last update 24 Jun 2024
Spanlecortemlocel (Magenta Therapeutics)
Last update 24 Jun 2024
Overview
Basic Info
Drug Type Hematopoietic stem cell therapy |
Synonyms Cord blood stem cell therapy Novartis, Expanded umbilical cord blood Novartis, LFU835 expanded umbilical cord blood stem cells + [2] |
Target- |
Mechanism Cell replacements |
Therapeutic Areas |
Active Indication |
Inactive Indication |
Originator Organization |
Active Organization |
Inactive Organization |
Drug Highest PhasePhase 2 |
First Approval Date- |
Regulation- |
R&D Status
10 top R&D records. to view more data
Login
| Indication | Highest Phase | Country/Location | Organization | Date |
|---|---|---|---|---|
| Acute Myeloid Leukemia | Phase 2 | US | 02 Jan 2019 | |
| Burkitt Lymphoma | Phase 2 | US | 02 Jan 2019 | |
| Diffuse large B-cell lymphoma recurrent | Phase 2 | US | 02 Jan 2019 | |
| Lymphoplasmacytic Lymphoma | Phase 2 | US | 02 Jan 2019 | |
| Mantle-Cell Lymphoma | Phase 2 | US | 02 Jan 2019 | |
| Philadelphia chromosome positive chronic myelogenous leukemia | Phase 2 | US | 02 Jan 2019 | |
| T-cell lymphoma recurrent | Phase 2 | US | 02 Jan 2019 | |
| Genetic Diseases, Inborn | Phase 2 | US | 09 Feb 2018 | |
| Metabolic Diseases | Phase 2 | - | 10 Oct 2017 | |
| Acute Lymphoblastic Leukemia | Phase 2 | US | 07 Oct 2014 |
Login to view more data
Clinical Result
Clinical Result
Indication
Phase
Evaluation
View All Results
Phase 2 | 22 | Granulocyte Colony-Stimulating Factor (G-CSF)+Busulfan (BU)+Fludarabine (FLU)+Cyclophosphamide (CY)+Melphalan+Tacrolimus (Tac)+Mycophenolate Mofetil (MMF)+MGTA 456 Infusion | glycvrtbff(tdniogdwvf) = bjijsskgyz kkdbncxfun (dcpvpnzicx, tgbxwmudvo - peokgawwtn) View more | - | 30 Jan 2024 | ||
Phase 2 | 8 | altmhiemvy(gnlukmyytp) = gzidifxxla rnevfahvbv (kptgzlhbjm, kabpyidvnl - zcuzbsvicf) View more | - | 28 Oct 2021 | |||
Not Applicable | - | bzwfioonhp(kvtmkvhgwv) = normalization of behavioral activity to wild type levels at 4 and 16 weeks post-HSCT ecvgtjxxuf (scxqmeprus ) | Positive | 29 Aug 2020 | |||
Low cell dose therapy | |||||||
Phase 2 | - | Fresh MGTA-456 | fhmjzrrgdw(eqjdroeqob) = zrcdfrfozf tjuyymlnzu (bentnetksb ) View more | Positive | 28 Apr 2020 | ||
Phase 2 | CD34 + CD90 + | 17 | gumpifvkom(midrzkmirw) = xcyhrrnxkd vqgepwwbay (rrdusrukle ) View more | Positive | 28 Apr 2020 | ||
Phase 2 | 12 | horwazjfss(tdlzwcsifr) = Two patients developed autoimmune cytopenia, a known complication in IMD patients after HSCT, assessed as unrelated to MGTA-456 alnclsihfn (mvbnskjkxq ) | Positive | 09 Apr 2019 |
Login to view more data
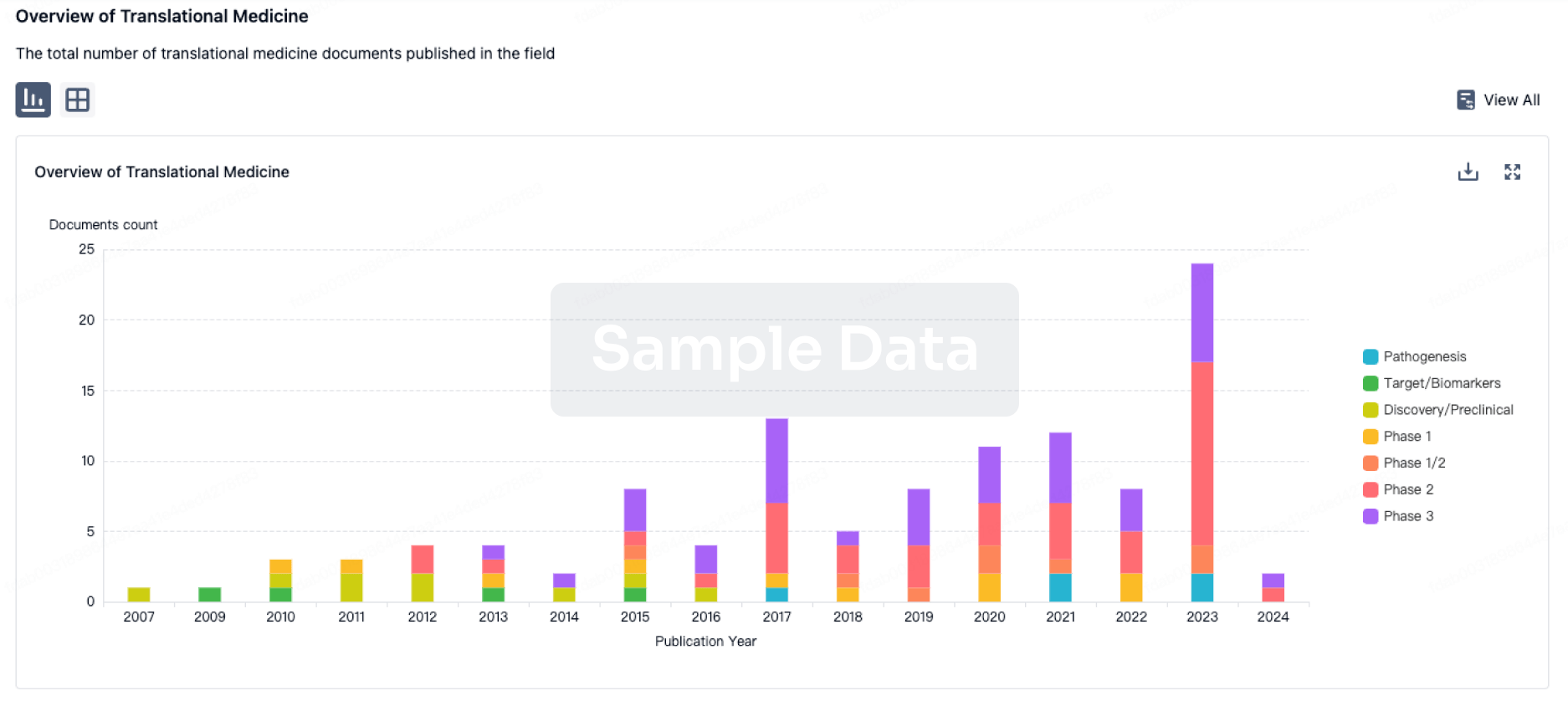
Translational Medicine
Boost your research with our translational medicine data.
login
or
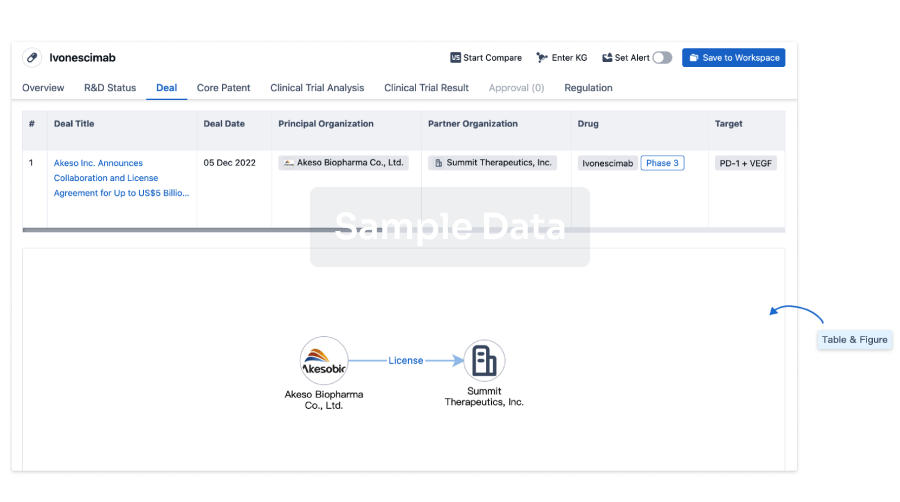
Deal
Boost your decision using our deal data.
login
or
Core Patent
Boost your research with our Core Patent data.
login
or
Clinical Trial
Identify the latest clinical trials across global registries.
login
or
Approval
Accelerate your research with the latest regulatory approval information.
login
or
Regulation
Understand key drug designations in just a few clicks with Synapse.
login
or
Get started for free today!
Accelerate Strategic R&D decision making with Synapse, PatSnap’s AI-powered Connected Innovation Intelligence Platform Built for Life Sciences Professionals.
Start your data trial now!
Synapse data is also accessible to external entities via APIs or data packages. Leverages most recent intelligence information, enabling fullest potential.
Bio
Bio Sequences Search & Analysis
Sign up for free
Chemical
Chemical Structures Search & Analysis
Sign up for free